![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
20 Cards in this Set
- Front
- Back
|
Regulation of protein synthesis |
Highly energy demanding so coordinated Uses 90% of chemical energy of cell Number of copies made corresponds to number of copies needed Proteins are targeted to cellular locations Degradation keeps pace with synthesis |
|
|
Proteins synthesis is complex |
In eukaryotes: Requires >300 bio molecules 70 > ribosomal 20 AA And so on |
|
|
3 early advances in proteins synthesis |
Proteins synthesized at ribosomes AAs activated for synthesis by attachment to tRNA via Aa-tRNA syhthetases TRNA acts as an adapted to translate mRNA into protein |
|
|
Genetic code for proteins consist of triplets of nucleotides |
20 common AAs 4 letter code in groups of 2 is insufficient but a 4 letter code in groups of 3 is sufficient Use overlapping mRNA code |
|
|
tRNA brings AA to mRNA |
Adaptor |
|
|
Overlapping v no overlapping |
Non: do not share nucleotides Provides much more flexibility and is used in all living systems |
|
|
Genetic code features |
Written in 5’-3’ direction First codon establishes reading frame 61/64 codons code for AAs There are 3 termination codes: UAA, UGA, UAG AIG is start code and Met |
|
|
Most AAs have more than one code |
20 AAs with 61 possible codons Only Met and Trp have a single codon Some codons are less subject to causes mutation |
|
|
Some exceptions with code |
Mitochondria encode their own DNA and use a slightly different code |
|
|
Wobble |
Weak H bonds formed Contain inosinate |
|
|
Molecular recognition |

The codon sequence is complementary with the anticodon sequence Codon base pies with anticodon in MRNA via h bonds The alignment of 2 RNA segments is antiparallel |
|
|
Resistant to mutations |
Degenerates (have same meaning) allows certain mutations to still code for the same AA Silent mutations-different nucleotide but same AA in protein Mutation in first base produces a conservative substitution (a change in AA) GUU-Val but AUU-leu |
|
|
Some mRNAs are edited before protein synthesis |
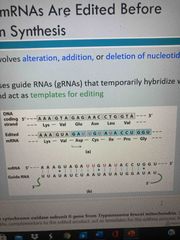
Alteration, addition, deletion of nucleotides Editing uses guide RNAs (gRNA) that hybridize with mRNAabd act as a template for editing |
|
|
5 stages of protein synthesis |

1. Activation of AAs: AA is attached to tRNA 2. Initiation: mRNA and tRNA bind to small ribosomal subunit, then large unit binds 3. Elongation: cycles of peptide binding occur until stop codon 4. Termination: translation stops when stop codon is reached. MRNA and protein detach and ribosome is recycled 5. Protein folding: posttranslational processing |
|
|
Ribosome |
Make up 25 % of weight in bacteria 65% rRNA (forms core and does peptide bond formation) 35% protein Made of 2 subunits |
|
|
Ribosome bacteria v eukaryotic |

Similar in 2 subunits with mRNA in between them Euk: larger, more complex contain >80 proteins Chloroplasts and mitochondria have simpler ribos than bacteria |
|
|
Characteristics of tRNAs |

Amino acid arm Anticodon arm D arm (contributes to folding) T psi c arm (binding btw base and ribose, helps in folding) |
|
|
tRNA synthetases |
Specific for both AA and tRNA (second genetic code) Specific tRNA molecule for a specific synthetase Anticodon region |
|
|
During intiation. Stage |

First is unique. AUG (met) Initiation factors bind to ribosome then met joins complex EPA: exit, peptide, accept AA |
|
|
During elongation |

Binding to amino site GTP hydrolysis |

