![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
18 Cards in this Set
- Front
- Back
|
What types of covalent bonds are there?
|
There are single bonds with one pair of electrons shared between atoms.
There are double bonds with two pairs of electrons shared between atoms. There are triple bonds with three pairs of electrons shared between atoms. |
|
|
What type of bond is shared in a single covalent bond?
|
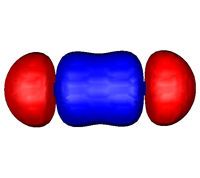
A sigma bond!
|
|
|
What type of bond is shared between atoms in a double covalent bond?
|
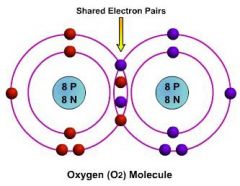
A sigma and a pi bond!
|
|
|
What type of bond is shared between the atoms of a triple covalent bond?
|
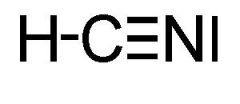
One sigma and two pi bonds!
|
|
|
What is electronegativity?
|
Electronegativity is a chemical property that determines the ability of an atom to attract electrons to itself in a covalent bond.
|
|
|
What is bond polarity?
|
Bond polarity is the dipole-dipole intermolecular forces between the positive end and negative end of a molecule.
|
|
|
How does bond polarity relate to electronegativity?
|
Bond polarity depends on the difference in electronegativities between atoms bonded together.
The greater the difference in electronegativity, the more polar the bond is. |
|
|
Sometimes a bond is polar but the molecule as a whole is not polar. Account for this and list the general rules of determining whether a molecule is polar.
|
Even if the bonds are polar, the positive and negative dipoles can cancel out and lead to a nonpolar molecule.
1. A symmetrical molecule is nonpolar regardless of the bonds being polar. 2. A nonsymmetrical molecule is polar if the bonds are polar. 3. A molecule with more than one type of atom attached to the central atom is often nonsymmetrical and polar. 4. A central atom with nonbonding electron pairs if often nonsymmetrical and polar. |
|
|
The formation of a covalent bond according to:
valence bond theory (VB) molecular orbital theory (MO) |
VB defines a covalent bond as the overlapping of two atomic orbitals when the electron spins are paired.
MO defines a molecule as similar to an atom with distinct energy levels that can be populated by electrons. |
|
|
What are Lewis Dot Molecular Structures (LDMS)?
|
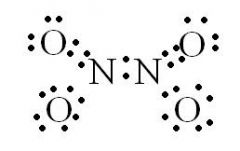
LDMS are diagrams that show the bonding between atoms of a molecule, and the lone pairs of electrons that may exist in the molecule.
|
|
|
How are Lewis structures formed?
(rules) |
1. Carbon is usually a central atom in the structure. In compounds with more than one C, the C atoms are chained together.
2. Hydrogen is never a central atom (only one electron!) 3. Halogens form a single covalent bond when oxygen is not present. 4. Oxygen forms two covalent bonds. 5. In simple molecules, the atom that appears once in the formula is usually the central atom. |
|
|
How are valence electrons arranged in LDMS?
|
1. The valence electrons are added together.
2. If the substance is polyatomic, the charge of the ion must be taken into account. 3. A pair of electrons is placed between each two atoms for a covalent bond (bonding pair). 4. The remaining electrons are used to complete the octets of the outer atoms (lone pairs). 5. If any electrons are left over, they are added in pairs to the central atom (lone pairs). 6. If the central atom does not have an octet, a double bond can be made. |
|
|
What is the octet rule?
|
A general rule that states each bonded atom should have a total of 8 electrons.
|
|
|
What are the exceptions to the octet rule?
|
Other atoms can have more than eight electrons.
For example: The central atom in PCl5 has a total of eight electrons. These exceptions usually occur with atoms in periods 3-7. |
|
|
How are charges taken into account for polyatomic ions and ions?
|
The - charge is interpreted as the adding of an electron.
For example in a SO4^2- ion, the 2- charge indicates that 2 extra electrons need to be added the to Lewis structure. |
|
|
What are resonance structures?
Give an example. |
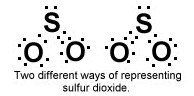
A resonance structure is an equally probable structure for a certain molecule. [Some molecules can have 2 or more resonance structures]
Ex: SO2 |
|
|
How can you tell if a compound has a resonance structure?
|
It is usually apparent when the central atom is bonded to 2 or more of the same type of atom.
For example in SO2, the the sulfur is double bonded to one O and single bonded to the other O. The resonance structure simply identifies the other scenario where the second O could have been double bonded instead of the first O. |
|
|
What about odd electron compounds?
Account for that. |
In a case where the total number of valence electrons is an odd number, then it would be impossible to follow the octet rule. These odd electron compounds are usually called free radicals. They have an unpaired electron that makes the molecule reactive.
An example would be NO2. |

