![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
12 Cards in this Set
- Front
- Back
|
Define the term structural formula. |
Shows the unique arrangement of atoms in a molecule in a simplified form, without showing all the bonds. |
|
|
Define the carbon bond in alkanes and alkenes? |
> -enes have a double carbon bond, |
|
|
Define how many carbons are in alkanes and alkenes? |
>MEPB |
|
|
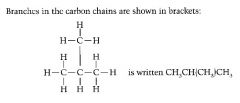
Define the prefixes used in organic chemistry to locate branches. |
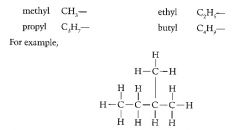
Prefixes are added to describe changes that have been made to root molecule. yl = the branch |
|
|
Define the 4 suffixes used in organic chemistry. |
>Alcohols (-OH) = -ol as in methanol, CH3OH |
|
|
What is a functional group? |
An atom or group of atoms in an organic molecule that are responsible for the characteristic of that molecule. |
|
|
a) What separates numbers? |
a) Commas |
|
|
Define an homologous series. |
A set of organic compounds with the same functional group. The compounds differ in the length of their hydrocarbon chains. |
|
|
What are structural isomers? |
Molecules that have the same molecular formula but whose atoms are arranged differently. |
|
|
What are the three ways in which structural isomerism can occur? |
>Positional isomerism - same functional group attached to main chain at different points |
|
|
How do you find how many hydrogens make up a branched or unbranched alkene? |
2 x C. |
|
|
Define the prefixes learnt in college. |
F - fluoro |

