![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
40 Cards in this Set
- Front
- Back
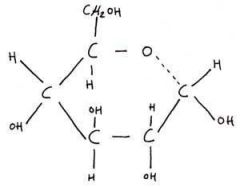
What molecule is this? |
alpha glucose |
|
|
What does ABBA stand for? |
OH- group Alpha Below Beta Above |
|
|
What is a condensation reaction? |
A chemical reaction that combines biological monomers Covalent bond is formed Water molecule is released |
|
|
What is a hydrolysis reaction? |
splitting larger molecules into smaller ones Covalent bond is broken Water molecule used |
|
|
What is metabolism? |
Sum total of all biochemical reactions taking place in the cells of an organism |
|

What are the similar properties of monosacchrides? |
1. Soluble in water 2.Sweet tasting 3. Form cyrstals |
|
|
What do two monosacchrides form? |
diasacchraide |
|
|
What is the general formula of monosaccharides? |
(CH2O)n |
|
|
Which two monosacchrides form lactose? |
glucose and galactose |
|
|
What is sucrose formed from? |
glucose and fructose |
|
|
What bond is formed in a condensation reaction between monosacchrides? |
glycosidic bond |
|
|
What is Maltose formed from? |
two glucose molecules |
|
|
Out of these sugars which is a non reducing sugar? Maltose, glucose, fructose, sucrose, lactose, galactose |
Sucrose, the rest are reducing, all monosacchrisdes are reducing sugars |
|
|
Why is it useful that starch is insoluble? |
It does not affect the water potential of the cell, water doesnt move in by osmosis |
|
|
Where is starch found? |
It is the main storage molecule in plants |
|
|
What is the difference between amylose and amylopectin? |
Amylose is alpha glucose monomers joind by 1,4 glycosidic bonds (forms long chains) Amylopectin has 1,4 glycosidic bonds as well as 1,4 glycosidic bonds. (branched) |
|
|
What kind of molecule is starch? |
polysaccharide |
|
|
Why are the glycosidic bonds in starch hydrolysed to form alpha glucose? |
Alpha glucose monomers can be easily transported for respiration |
|
|
Why can starch be stored in a small space? |
Small and compact |
|
|
Where is glycogen found? |
animals and bacteria- liver and muscles |
|
|
How is glycogen different to starch? |
more branches and shorter chains |
|
|
Why is glycogen highly branched? |
So the glycosidic bonds can be rapidly hydrolysed by enzymes to form alpha glucose monomers which can be easily transported and used in respiration |
|
|
Why is it important that glycogen is a quicker energy source than the equivalent amylopectin in plants? |

Animals are more active than plants and have a higher metabolic and respiration rate |
|
|
What characteristics of glycogen allow it to be stored in cells? |
insoluble- does not affect the water potential of the cell Compact- can be stored in a small space |
|
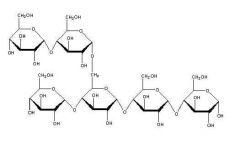
What molecule is this? |
Amylopectin |
|
|
How is cellulose different to glycogen and starch in terms of monomers? |
Cellulose is formed from beta glucose monomers whereas glycogen and starch are made of alpha glucose |
|
|
Where is cellulose formed? |
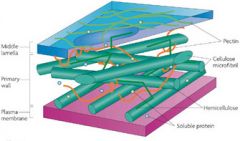
plant cell walls (cellulose cell wall) |
|
|
What are parallel chains of beta glucose linked together by? |
Hydrogen bonds between the many OH- groups |
|
|
What do many parallel chains of beta glucose held together by hydogen bonds between OH-groups form? |
microfibrils |
|
|
What bonds are beta glucose monomers held together by to form chains |
1,4 glycosidic bonds |
|
|
What results in alternate glycosidic bonds in cellulose? |
each monomer is rotaed 180 degrees to the next |
|
|
What do microfibrils do? |
They are very strong and support the cell wall and prevent it from bursting when cell becomes turgid |
|

Why can mammals not digest cellulose? |
They do not have cellulase |
|

Why can herbivores digest cellulose? |
Have symbiotic bacteria to produce cellulases |
|
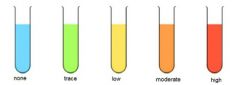
What is the test for reducing sugar? |
Benedicts: add an equal volume of benedicts to solution being tested; heat to 80C in waterbath; Solution changes colour from blue to a red precipitate forms |
|

What is the test for starch? |
Add iodine; changes colour from yellow/brown to black |
|
|
Why is NaOH added when testing for non reducing sugar? |
To neutralise acid |
|
|
Why is acid added (HCl) when testing for non-reducing sugar? |
to break glycosidic bonds |
|
|
What is the colour change when testing for for non reducing sugar? |
blue to red precipitate forming |
|
|
Describe how a test for non reducing sugar is carried out |
add acid (HCl) and heat to 80C in waterbath. Add NaOH and benedictssolution. Heat in water bath again for 5 mins |

