![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
53 Cards in this Set
- Front
- Back
|
elements
|
-a substance that can't be broken down into a simpler chemical substance
-25 are essential for living organisms |
|
|
symbols
|
general symbols- C=carbon, H=hydrogen, He=helium, etc.
|
|
|
trace elements
|
-elements that are present in very small amounts
(ex. I=iodine, Zn=zinc, Co=cobalt, Cu=copper, Fe=iron) -know 3 -% in human body=trace |
|
|
Table 6.1, Elements that make up the human body
|
Oxygen-O-65%,
Carbon-C-18.5%, Hydrogen-H-9.5%, Nitrogen-N-3.3%, Calcium-Ca-1.5%, Phosphorus-P-1.0% |
|
|
atoms
|
-the basic building blocks of all matter
-the smallest part of an element that has characteristics of that element |
|
|
Nucleus
|
center of the atom
-positively charged |
|
|
protons
|
positively charged particles
|
|
|
neutrons
|
no charge
|
|
|
electrons
|
negatively charged
|
|
|
electron energy levels
|
1st energy level----2
2nd level----8 3rd level----8 4th level----10 |
|
|
isotopes
|
atoms of the same substance that have different #'s of neutrons (more)
|
|
|
covalent bond
|
-atoms share electrons
-not always shared equally EXAMPLE: |
|
|
water
|
-makes up 70-95% of most organisms
-water is polar |
|
|
polar molecule
|
molecule with an unequal distribution of charge
ex- water |
|
|
Label an atom, structure
|
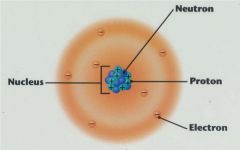
This is a labled atom.
|
|
|
MOLECULE
|
a group of atoms held together by covalent bond
|
|
|
ION
|
a charged particle made of atoms
|
|
|
ionic bond + example
|
The attractive force between two ions of opposite charge
EXAMPLE: chlorine and sodium |
|
|
COMPOUND
|
a substance that is composed of 2 or more different elements that are chemically combined
|
|
|
Electron cloud
|
the region of space surrounding the nucleus/ELECTRON
|
|
|
Hydrogen bonds
|
The attraction of opposite charges between hydrogen and oxygen which makes a weak bond. HELPS HOLD PROTEINS TOGETHER
|
|
|
capillary action
|
water can climb up tubes
|
|
|
WATER TEMPERATURE CHANGES
|
'heats up slowly ---> requires a lot of heat to increase temp.
'cools off quikly 'good insulator (ex. wetsuits) "Important for cells "Helps maintain homeostasis |
|
|
Ice
|
'expands when it freezes
'ice is less dense than water--->floats LiQUID- MOVES AROUND ICE- ORGANIZED |
|
|
Diffusion
|
the net movement of particles from an area of higher concentration to an area of lower concentration
|
|
|
Brownian motion
|
Diffusion results because of the random movement of particles
|
|
|
Dynamic equilibrium
|
the molecules continue to move randomly and collide with one another; however, no further charge in concentration will occur
|
|
|
rate of diffusion chart
-affects of temperature, pressure and concentration |

title- Rate of Diffusion
see Question |
|
|
Single Bond
|
C-C Carbon can bond with other carbon atoms
SHARE 1 e- |
|
|
Double Bond
|
C-C When each atom shares two electrons
SHARE 2 e- |
|
|
Triple Bond
|
C-C When each atom shares three electrons
SHARE 3 e- |
|
|
Isomers
GLUCOSE/FRUCTOSE |

--> C 6 H 12 O 6
Isomers- compounds w/ the same chemical formula but different physical structure |
|
|
Isomers
Glucose/Fructose |
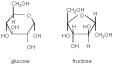
--> C 6 H 12 0 6
ISOMERS- compounds w/ the same chemical formula but different physical structure |
|
|
Biomolecules
|
Large organic proteins that have Carbon
ex- proteins |
|
|
Carbon--->Organic
|
Organic molecuels contain Carbon and Hydrogen
|
|
|
Polymers
-condensation -hydrolysis |
polymers: a large molecule formed when many smaller molecules bond together
-formed by condensation (1 molecule joins larger chain and forms bigger polymer) -breaks polymers apart (takes ine part of polymer off) |
|
|
Carbohydrates
|
a biomolecule composed of carbon, hydrogen and oxygen, USED BY CELLS TO PROVIDE ENERGY
|
|
|
-Monosaccharide
|
simplest type of carb, simple sugar
ex- fructose, glucose |
|
|
Disaccharide
|
2 monosaccharides combined
ex- Glucose + Fructose = Sucrose |
|
|
Polysaccharide
|
More than 2 monosac's
ex- starch, glycogen, cellulose |
|
|
Lipids
|
-C, H or small amount of O
EX. FATS, OILS, WAXES -non-polar = insoluble in water -3 Fatty acids and 1 glycerol Fatty acid=long chain of C + H Glycerol=3 C molecuel which is backbone for lipid |
|
|
1 Saturated
2 Unsaturated 3 Polyunsaturated |
1- Carbon have single bonds
2- Carbon have double bonds 3- Carbon have triple bonds |
|
|
Protiens
|
carry out metabolism + give structure to tissue
-C,H,N,O + sometimes S |
|
|
1- amino acids
2- Peptide bond 3- Importance of Shape 4- Enzymes |
1- basic building blocks of proteins
- some acidic, some basic, some no charge -->causes them to attract/repel eachother 2- covalent bond b/w amino acids - diff shapes and sizes 3- b/c if an amino acid sequence changes the protein may fold diff and won't be able to do its job 4- a protein that changes the rate of a chemical reaction--> speeds them up |
|
|
Nucleic ACIDS
|
COMPLEX biomolecule that stores cellular information in the form of a code --> blueprint
|
|
|
1- Nucleotides
2- 3 groups: Nitrogen base, simple sugar and phosphate group 3- DNA and RNA |
1- smaller sub-units that make up polymers
2-3 groups: nitrogenous base, simple sugar, phosphate group 3- Deoxyribonucleic acid and ribonucleic acid DNA- MASTER COPY OF ORGANISMS INFORMATION CODE, DETERMINES HOW AN ORGANISM LOOKS AND ACTS RNA- copy of DNA used for making proteins |
|
|
Cells
|
basic units of living organisms
|
|
|
Cell Theory
|
1- all organisms are composed of 1 or more cells
2- basic units of structure and function of organisms 3- All cells come from preexisting cells |
|
|
pOKARYOTIC VRS. EUKARYOTIC
|
prokaryotic- (bacteria)
1- do not contain membrane bound organelles (small specialized structures w/in cells Eukaryotic (animal and plant cells) - 1- membrane bound organelles 2- most multicellular organisms 3- can be unicellular 4- yeast, amoeba and some algae |
|
|
Organelles
1- Cell Wall 2- Nucleus 3- Neucleolus 4- ribosomes 5- cytoplasm 6- nuclear envelope 7- ER 8- Golgi Apparatus 9-Vacuole 10- Lysosomes 11- Choloroplasts 12-Mitochondria 13- Cytoskeleton |
1- provides additional support and protection outside membrane, non-selective
2- brain of the cell, directions 4 proteins 3- where chromosomes are made 4- where cells produce proteins -->directions from DNA 5- clear fluid inside cell 6- seperates the nucleus from the cytoplasm 7- site of cellular chemical reactions 8- sorts proteins and packs them into membrane bound structures and sends them away 9- stores food,enzymes and waste 10- contain digestive enzymes 11- captures sunlight/energy and converts it to chemical energy 12- transform energy 13- used for support and structure -constantly changing shape -microtubles (cylinder) and microfilaments (protein fibers) |
|
|
Plasma Membrane
|
flexible boundary b/w cell and its environment that allows a steady supply of nutrients to come into the cell despite the outer conditions
-glucose, amino acids + lipids |
|
|
Phospholipid
Phospholipid Bilayer |
-lipids-->3 fatty acids and 1 glycerol
-if u replace a fatty acid w/ a phosphate you get a phospholipid -->2 fatty acids, 1 phosphate, 1 glycerol SEE PICTURE IN NOTES head is polar--> soluble in water tail is non polar--> insoluble in water |
|
|
proteins
|
throughout entire membrane
-help regulate which molecules enter and leave the cell |

