![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
69 Cards in this Set
- Front
- Back
|
Totality of an organism's chemical processes. |
Metabolism
|
|
|
Metabolic pathways which release energy by breaking down complex molecules to simpler compounds.
|
Catabolic pathways
|
|
|
Metabolic pathways which consume energy to build complicated molecules from simpler ones.
|
Anabolic pathways
|
|
|
the capacity to do work - to move matter against opposing forces.
|
Energy
|
|
|
Energy in the process of doing work (energy of motion).
|
Kinetic energy
|
|
|
Energy that matter possesses because of its location or arrangement (energy of position).
|
Potential energy
|
|
|
Study of energy transformations
|
Thermodynamics
|
|
|
Collection of matter under study which is isolated from its surroundings
|
Closed system
|
|
|
System in which energy can be transferred between the system and its surroundings.
|
Open system
|
|
|
Energy can be transferred and transformed, but it cannot be created or destroyed (energy of the universe is constant).
|
First Law of Thermodynamics
|
|
|
Every energy transfer or transformation makes the universe more disordered (every process increases the entropy of the universe).
|
Second Law of Thermodynamics
|
|
|
Quantitative measure of disorder that is proportional to randomness (designated by the letter S).
|
Entropy
|
|
|
Those reactions that can occur without outside help.
|
Spontaneous processes
|
|
|
Those reactions that can only occur if energy is added to a system.
|
Nonspontaneous processes
|
|
|
Portion of a system's energy available to do work; is the difference between the total energy (enthalpy) and the energy not available for doing work (TS).
|
Free energy (G)
|
|
|
Formula for free energy
|
G = H - TS
|
|
|
Formula for free energy change
|
ΔG = ΔH - TΔS
|
|
|
In the formula for free energy change, what is ∆G?
|
change in Free energy
|
|
|
In the formula for free energy change, what is ∆H?
|
change in Enthalpy (total energy)
|
|
|
In the formula for free energy change, what is ∆S?
|
change in Entropy
|
|
|
In the formula for free energy change, what is T?
|
Temperature in Kelvin
|
|
|
A reaction that proceeds with a net loss of free energy.
|
Exergonic reaction
|
|
|
An ________reaction is one that releases free energy to its surroundings.
|
exergonic
|
|
|
Δ G of an exergonic reaction is _____ (positive/negative)
|
negative.
|
|
|
Amount of energy that reactant molecules must absorb to start a reaction.
|
Energy of activation
|
|
|
Unstable condition of reactant molecules that have absorbed sufficient free energy to react.
|
Transition state
|
|
|
An energy-requiring reaction that proceeds with a net gain of free energy.
|
Endergonic reaction |
|
|
An _____ reaction is one that requires free energy from its surroundings.
|
endergonic
|
|
|
Δ G of an endergonic reaction is ____ (positive/negative)
|
positive.
|
|
|
reaction in which the rates of forward and backward reactions are equal and there is no change in the concentration of products or reactants.
|
Chemical Equilibrium
|
|
|
The ΔG of a reaction at equilibrium is ______
|
Zero
|
|
|
To do work, cells manage energy resources by energy coupling, the use of an ___ process to drive an ____ one
|
exergonic/endergonic
|
|
|
Nucleoside triphosphate with unstable phosphate bonds that the cell hydrolyzes for energy to drive endergonic reactions.
|
ATP (adenosine triphosphate) |
|
|
The bonds between the phosphate groups of ATP’s tail can be broken by ____ reactions
|
hydrolysis
|
|
|
List the three main kinds of work of ATP.
|
1. Chemical work such as driving endergonic reactions such as the synthesis of polymers from monomers |
|
|
Chemical agent that accelerates a reaction without being permanently changed in the process, so it can be used over and over. |
Catalyst
|
|
|
Biological catalysts, which are usually proteins.
|
Enzymes
|
|
|
Enzymes catalyze reactions by lowering the _____
|
EA barrier |
|
|
The substance an enzyme acts on and makes more reactive.
|
Substrate
|
|
|
Change in the shape of an enzyme's active site, which is induced by the substrate.
|
Induced fit
|
|
|
Restricted region of an enzyme molecule which binds to the substrate.
|
Active site
|
|
|
List the 4 mechanisms that enzymes use to lower activation energy and speed a reaction |
1. orients substrates
2. put stress on bonds that must be broken, making it easier to reach the transition state. 3. favorable microenvironment for a specific reaction. 4. Enzymes may even bind covalently to substrate |
|
|
List the environmental factors that can effect enzyme rate. |
1.Temperature |
|
|
Small non-protein molecules that are required for proper enzyme catalysis. |
Cofactors
|
|
|
a non-protein organic cofactor.
|
Coenzyme
|
|
|
The protein portion of an enzyme.
|
Apoenzyme
|
|
|
The apoenzyme plus its coenzyme or cofactor.
|
Holoenzyme
|
|
|
molecules that bind to enzymes and prevent enzymes from catalyzing reactions. |
Inhibitors
|
|
|
Chemicals that resemble an enzyme's normal substrate and compete with it for the active site. |
Competitive inhibitors
|
|
|
Enzyme inhibitors that do not enter the enzyme's active site, but bind to another part of the enzyme molecule |
Noncompetitive inhibitors
|
|
|
Enzymes are proteins encoded by ___
|
genes
|
|
|
Mutations (changes) in genes lead to changes in ____ of an enzyme
|
amino acid composition
|
|
|
Altered amino acids in enzymes may alter their shape and affect ______.
|
substrate specificity
|
|
|
Specific receptor site on some part of the enzyme molecule other than the active site.
|
Allosteric site
|
|
|
stabilizes the active form of the enzyme
|
Activators
|
|
|
stabilizes the inactive form of the enzyme
|
Inhibitor
|
|
|
Regulation of a metabolic pathway by its end product, which inhibits an enzyme within the pathway.
|
Feedback inhibition
|
|
|
How can the localization of enzymes within a cell help order metabolism |
1.Metabolic pathway assembled in a multienzyme complex
2.Fixed locations within the cell as structural components of membranes 3.In solution, within an organelle with its own internal microenvironment |
|
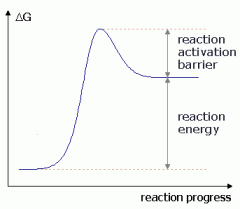
Endergonic or exergonic?
|
endergonic
|
|
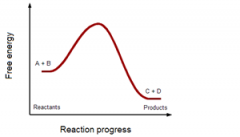
Endergonic or exergonic? |
exergonic
|
|
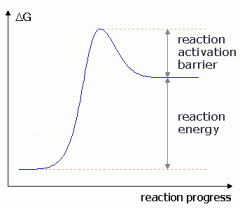
spontaneous or non-spontaneous? |
non-spontaneous
|
|
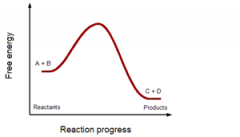
spontaneous or non-spontaneous?
|
spontaneous
|
|
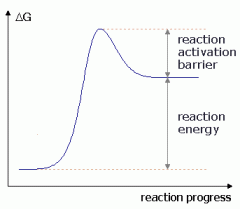
Substrates are ___energy and products are ____ energy. |
low
high |
|
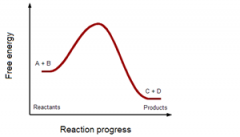
Substrates are ___energy and products are ____ energy. |
high
low |
|
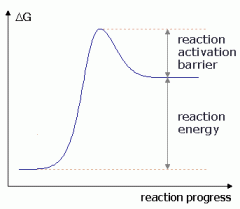
negative or positive ΔG? |
positive
|
|
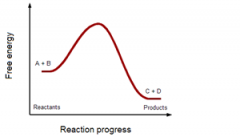
negative or positive ΔG?
|
negative
|
|
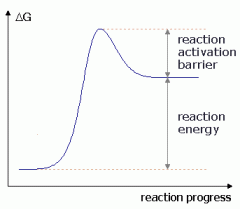
Does this reaction absorb or release energy (net)?
|
absorb
|
|
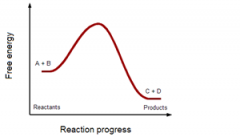
Does this reaction absorb or release energy (net)?
|
release
|
|
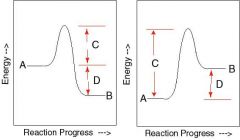
What are the sections of the graphs labeled A-D? |
A free energy of substrate |

