![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
25 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Crystalline solid |

|
What, mp, nature,cutting, solid, enthalpy of fusion , examples |
|
|
Amorphous solids |

|
What, shape or form, short range, mp, nature, cutting, so,id, enthalpy of fusion, ex |
|
|
On the basis of intermolecular forces |
Ionic Metallic Covalent network Molecular solids |
|
|
|
Ionic solids |

|
What, strength, conduction, enthalpy of vaporization, solubility, ex, |
|
|
Metallic solid |
Previous |
Constituents, contribution, forces, strengthn, enthalpy of fusion, fun of e- |
|
|
Covalent solid |
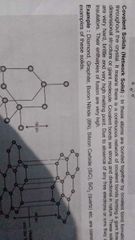
|
Other name, constitute, forces, what, strength, direction, conduction, enthapy of fusion, example, allotropes |
|
|
Diamond and graphite |

|
D what,strength, mp , conduction G what, conduction, forces, nature of forces |
|
|
Molecular solids |
Dispersion forces, London forces, dipole dipole force and hydrogen bond Non polar molecular solids , polar molecular solids, hydrogen bonded molecular solids. |
Forces 4, types |
|
|
Non polar molecular solids |

|
Constitute, forces , conduction , strength, mp, enthalpy of vaporization, volatile so state, example |
|
|
Polar molecular solids |

|
Constitute, forces, conduction, strength, mp bp, zenthalpy of vaporization, ex |
|
|
Hydrogen bonded molecular solids |

|
Constitute, room temp- volatile liquids or soft solids, non conductor example |
|
|
Lattice point |
In a crystalline lattice constituent particles are arranged in a definite three dimensional regular geometrical order along all the three axes in which each particle is depicted as in lattice point |
|
|
|
Crystal lattice |
Three dimensional regular arrangement of lattice points in in space or in a crystal is called a crystal lattice or space lattice |
|
|
|
Characteristics of crystal lattices |
First each point in lattice is called lattice site a lattice point second each lattice point represents one constituent particles that is atom Ion or molecule third we join lattice points by straight line to show geometry of the lattice |
Lattice site Lattice point Geometry of lattice |
|
|
Unit cell |
The smallest repeating unit in space lattice which one repeated over and over again in different directions produce complete crystal lattice |
|
|
|
Imp parameters of unit cell |
Edge length or axial distance- length or dimension along the three edges a b and c which may or may not be mutually perpendicular 2nd interaxial angle angle alpha beta and gamma between pair of edges are interaxial angle alpha represents angle between axis b and c beta represents angle between axis a and c and Gama represents angle between a and b |
2 |
|
|
Types of unit cell |
Fundamentally 2-primitive-it contains only 8 lattice point are constituent particles at 8 corners only and non primitive\ centers unit cell contain it contains one or more lattice points at other than it corner positions in a unit cell 1 what is body centred unit cell there are 8 lattice points at 8 corners and one additional lattice point at the centre of body secondface centered there are 8 points at corners and 6 more lattice points at the centre of each face endface centred unit cell there are lattice point at 8 corners and 1 constituent particles is present at the centre of any two opposite faces |
|
|
|
Crystal habits |

After taking the axial length a b and c and inter axial angle alpha beta and gamma into consideration various crystals are divided into 7 types of crystal systems also called crystal habits |
Zuca kl diamond pehan cafe Gaya Santi ca white tin PCaR KBK Epsont he MN PubG CHK CSK ZCG ECG CCS QK |
|
|
Bravais lattice |
The seven crystal systems can be further classified on the basis of unit cells present bravais a French mathematician in 1848 has established 14 different types of lattice called bravais lattice according to the arrangement of the points in different unit cells involved |
|
|
|
Constituent per unit cell primitive, bcc, fcc |
1, 2, 4 |
|
|
|
Relation between packing and stability |
Closer the packing more will be the stability of the crystal |
|
|
|
Close packing in 1d |
Arrangement of different atoms in a row touching each other forms one dimension or edge the coordination number is 2 |
|
|
|
Types of close packing in 2d |
Square close packing and hexagonal close packing |
|
|
|
Square close packing |
Spheres or constituent particles are packed in such a way that they are aligned together vertically as well as horizontally and centre of all spheres in are in a straight line here each sphere is in contact with four other spheres in same plane this is termed as square close packing and the coordination number is 4 |
|
|
|
Hexagonal close packing |
7 second row is arranged in the depression of the first row and all atoms aligned diagonal they are in contact with six others sphers in same plane it is known as hexagonal close packing it is more efficient packing method than square close packing hear coordination number is 6 and triangular voids are formed |
|

