![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
65 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
What effects does Acetaminophen possess?
|
Analgesic
Antipyretic **no anti-inflammatory activity |
|
|
|
Acetaminophen (Tylenol) T or F:
1. Affects platelets fxn 2. Does not affect uric acid levels 3. causes gastric irritation 4. should not be given to children |
1. False
2. True 3. False 4. False |
|
|
|
What is the most serious adverse effect of acute overdose of Acetaminophen?
|
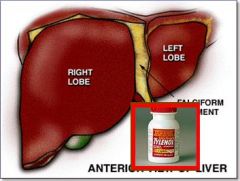
Hepatic necrosis
|
|
|
|
There is some evidence that Acetaminophen exerts its action on _______ in the CNS
|
COX-3
|
|
|
|
What CYP causes bioactivation of Acetaminophen?
What 2 things induces this CYP? |
CYP 2E1
Alcohol Isoniazid |
|
|
|
Acetaminophen can cause __1__, especially in __2__-sensitive individuals
|
1. acute hemolytic anemia
2. Primaquine |
|
|
|
Acetaminophen-induced __1__ is synergistic with __2__
|
1. Hepatotoxicity
2. Alcohol consumption or Isoniazid |
|
|
|
What are 3 rare adverse effects of Acetaminophen?
|
1. Neutropenia
2. Pancytopenia 3. Leukopenia |
|
|
|
What are the 3 mechanisms of Acetaminophen metabolism that are safe?
|
1. Sulfation
2. Glucuronidation 3. Glutathione |
|
|
|
What is the down-side of Sulfation and Glucuronidation in Acetaminophen metabolism?
|
They run at a relatively full capacity = co-substrates needed for the rxn run out relatively quickly
|
|
|
|
What is the antidote for Acetaminophen overdose?
Within what time period should the antidote be given to yield maximal effects? |
N-acetlycysteine = regenerates Glutathione
8 hours |
|
|
|
What are the first line drugs used in Rheumatoid Arthritis treatment?
|
NSAIDs
|
|
|
|
Drugs that cause dramatic improvement in Rheumatoid arthritis; do not arrest the disease process itself; adverse effects limit their long-term use
|
Corticosteroids
|
|
|
|
RA drugs that have a slow onset of action (6 wks to 6 months) and their mechanism of action is not well understood
|
Disease Modifying Antirheumatic Drugs (DMARDs)
|
|
|
|
What are the 2 antimalarial drugs used for Rheumatoid Arthritis treatment?
|
Hydroxychloroquine
Chloroquine |
|
|
|
Which antimalarial is used more commonly to treat RA and SLE?
|
Hydroxychloroquine
|
|
|
|
What is the contraindication for Hydroxychloroquine/Chloroquine?
|
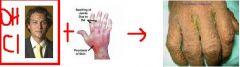
Psoriatic Arthritis b/c of possible development of Exfoliative Dermatitis
|
|
|
|
Antimalarial drugs can cause irreversible __1__ damage.
Which one causes this more? |

1. Retinal
2. Chloroquine |
|
|
|
What should gold compounds not be given with? Why?
|
Penicillamine
B/c it chelates gold |
|
|
|
What are 4 contraindications for Gold compounds?
|
1. Previous gold toxicity
2. Pregnancy 3. Liver or Kidney impairment 4. Blood dyscrasia |
|
|
|
What are the 4 adverse effects of Gold Compounds?
|
1. Dermatitis and diarrhea
2. Eosinophelia & other blood disturbances 3. Proteinuria -> nephrotic syndrome 4. Aplastic anemia (rare but fatal) |
|
|
|
What are 2 gold compuonds used in RA?
|
1. Aurothioglucose
2. Auranofin |
|
|
|
Administered IM and is an adjunctive treatment of early active cases of adult and juvenile RA
|
Aurothioglucose
|
|
|
|
Adverse effects of this DMARD are listed below:
1. Erythema 2. Exfoliative dermatitis 3. Kidney toxicity 4. Blood dyscrasias |
Aurothioglucose
|
|
|
|
This is the only gold compound available for Oral administration
|
Auranofin
|
|
|
|
What is Penicillamine's mechanism of action?
|
Oral chelating agent
|
|
|
|
What is Penicillamine used for? (3)
|
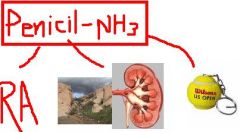
1. Wilson's disease = copper accumulation disease
2. Cystinuria = urinary stones 3. resistant cases of RA |
|
|
|
What is Sulfasalazine used to treat? (3)
|

1. Rheumatoid arthritis
2. Ankylosing spondylitis 3. Ulcerative colitis |
|
|
|
Leflunomide:
-Administration: __1__ -converted to an active metabolite that inhibits __2__, an enzyme involved in __3__ -leads to a translocation of __4__ to the nucleus -stimulated cells are arrested in __5__ phase |
1. Oral
2. Dihydroorotic acid dehydrogenase 3. Ribonucleotide synthesis 4. P53 5. G1 |
|
|
|
DMARD that is converted to an active metabolite that inhibits Dihydroorotic acid Dehydrogenase, an enzyme involved in ribonucleotide synthesis.
Inhibits autoimmune T cell proliferation and production of auto-antibodies by B cells |
Leflunomide
|
|
|
|
This was the 1st agent that was indicated for both symptomatic improvement and retardation of structural joint damage in RA
|
Leflunomide
|
|
|
|
What are 3 possible adverse effects of Leflunomide?
|
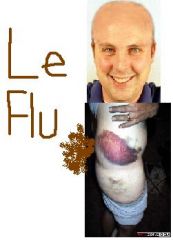
1. Alopecia
2. Rash 3. Diarrhea |
|
|
|
List the 3 Anti-TNF-alpha compounds used to treat RA
|

1. Infliximab = monoclonal Ab to TNF-alpha
2. Etanercept = recombinant form of TNF receptor that binds TNF-alpha 3. Adalimumab = monoclonal TNF-alpha Ab |
|
|
|
IL-1 receptor antagonist that reduces the signs and symptoms and slows the progression of structural damage of moderate to severe active RA in patients who failed one or more DMARDS
|

Anakinra
|
|
|
|
How is Anakinra administered?
|
Subcutaneously once per day
**anaKINra = SKIN = Subcutaneously |
|
|
|
This drug is a co-stimulatory or second-signal blocker of T cell activation
|
Abatacept
|
|
|
|
How is Abatacept administered?
|
IV infusion
- day 0, week 2, & week 4 followed by monthly injections |
|
|
|
When is Abatacept used?
|
In patients who are unresponsive to DMARDs of TNF antagonists
|
|
|
|
What is there an increased risk of when using Abatacept?
|
Infections
|
|
|
|
What causes Gout?
|
increase in the amount of Uric Acid in the body
|
|
|
|
What are the primary causes of Gout?
|
Overproduction or under-excretion of Uric Acid
-Under excretion = diabetes and in starvation -Over production = de novo synthesis |
|
|
|
What could be a secondary cause of Gout?
|
Accumulation of Uric Acid due to Leukemia
-diseases with alot of Purine synthesis and degredation |
|
|
|
List 6 drugs or groups of drugs that may cause under-excretion and/or accumulation of Uric Acid (hint)
|
1. Diuretics = Thiazides, Furosemide, Ethacrynic acid
2. Anti-TB drugs = Pyrazinamide, Ethambutol 3. Nicotinic acid 4. Ethanol 5. Cytotoxic drugs 6. Salicylates |
Dr. Gouty takes Diuretics, has TB, Smokes and Drinks (which are Cytotoxic) and treats everything with Aspirin
|
|
|
What are the symptoms of Gout?
|
Inflammatory reaction mediated prostaglandins and immune cells due to phagocytosis by synoviocytes of uric acid crystals deposited in peripheral joints -> inflammation -> severe pain
|
|
|
|
List the pathway leading to Gout
|

-
|
|
|
|
Why are Diuretics and Low-dose Salicylates contraindicated in Gout?
|
they block tubular secretion of Uric Acid
|
|
|
|
List the pathway of Uric Acid synthesis?
|
Purines -> Hypoxanthine -> Xanthine -> Uric acid
|
|
|
|
3 drugs used in the treatment of Acute Gout
|
1. Colchicine
2. NSAIDs -> Indomethacin 3. Corticosteroids |
|
|
|
Explain Colchicines method of action in treating acute attacks of gouty arthritis
|
Inhibits microtubule polymerization = inhibits Leukocyte migration and phagocytosis = anti-inflammatory
|
|
|
|
What are the adverse effects of Colchicine?
|
NVD
**Colchicine makes you feel bad in NeVaDa |
|
|
|
What is the primary NSAID used to treat severe gout?
|
Indomethacin
|
|
|
|
What other NSAIDs can be used to treat gout?
|
Ibuprofen
Naproxen Sulindac "I SIN" with Gout |
|
|
|
What NSAID should not be used to treat gout? Why?
|
Aspirin b/c of its dual effect on the kidney
|
|
|
|
What makes Corticosteroids useful in treatment of gout?
|
Anti-inflammatory properties
Side effects associated are significant with prolonged use |
|
|
|
What are the 2 prophylactic drugs used in Gout?
|
1. Uricosuric agents
-Sulfinpyrazone - Probenicid 2. Allopurinol |
|
|
|
What is the mechanism of action of the Uricosuric agents?
|

increase urinary excretion of Uric Acid by blocking the Active Reabsorption in proximal tubule
|
|
|
|
What should be maintained when using Uricosuric agents? Why?
|
Large urine volume to minimize possibility of stone formation
|
|
|
|
What may Sulfinpyrazone increase the serum concentration of?
|
1. Sulfonamides
2. Salicylates **displaces them from plasma binding proteins |
|
|
|
Oral uricosuric agent used to treat hyperuricemia associated with CHRONIC gout or secondary to other causes (drug-induced hyperuricemia)
|
Probenicid
*also inhibits secretion of Penicillin |
|
|
|
Gout drug that should not be used for ACUTE attacks b/c it can aggravate inflammation if administered during initial stages
|
Probenicid
|
|
|
|
What is Allopurinol's mechanism of action?
|
Inhibits Xanthine oxidase and thus inhibits Uric Acid synthesis
|
|
|
|
When is Allopurinol used?
|
In both primary and secondary forms of gout
|
|
|
|
What are the side effects of Allopurinol?
|
1. Vasculitis
2. Agranulocytosis 3. Hypersensitivity rxns |
|
|
|
Allopurinol inhibits the conversion of __1__ and __2__ to their inactive metabolites.
If allopurinol needs to be used with these 2 antimetabolites, their doses must be __3__ |
1. Mercaptopurine
2. Azathioprine 3. reduced |
|
|
|
When should Uricosuric agents not be used?
|
if GFR < 50 mL/min
|
|

