![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
203 Cards in this Set
- Front
- Back
|
Uses for general anesthetics
|
Unconsciousness: opioid (morphine), inhaled (ether), anticholinergic (atropine)
Analgesia: opioids Amnesia: ether, atropine No movement: ether (sort of, only spinal reflexes), succinyl-choline (NMJ blocker of ACh receptors) Autonomic control: opioids for ↓HR, ether for ↓BP, sometimes atropine |
|
|
Routes of administration of general anesthetics
|
Inhaled:
- ex: desflurane, sevoflurane, isoflurane, NO (only when administer in hyperbaric chamber) - typically smaller molecules, lipophilic to cross membranes/BBB - potency determined by minimum alveolar concentration (MAC) (50% immobile to noxious stimuli) - when administered vessel-rich tissues have majority then drop off quickly after cessation. Muscle/fat slowly accumulate then leech back after cessation Intravenous: - tend to be larger molecules, also lipophilic to cross BBB - generally large volume of distribution (brain, liver, kidneys) where drugs work effect. Non-brain tissues may then leech back drug to central compartment - cessation of effect generally due to redistribution not metabolism/elimination |
|
|
4 Stages of general anesthesia
|
Induction: period initial administration before loss of consciousness. Patient progresses from analgesia without to with amnesia. Patients can converse.
Excitement: period after loss of consciousness marked by excited and delirious activity. May have irregular HR/RR, uncontrolled movements, vomiting, breath-holding leading to respiratory compromise. Rapid acting drugs used to move through this stage ASAP Surgical Anesthesia: relaxed skeletal muscles, regular breathing, slowing/stopping of eye movements (rolling→fixed, loss of corneal & laryngeal reflexes, pupil dilation and no light reflex, intercostal paralysis). Surgery may begin. Overdose: too much medication given relative to amount of surgical stimulation and patient has severe brainstem or medullary depression (cessation or respiration, potential CV collapse). Lethal w/o cardiovascular and respiratory support. |
|
|
Mechanisms of action of general anesthetics
|
General: tip NT balance toward glutamatergic signaling over glutamate, mediated by interactions w/ membrane bound proteins
GABA-A receptors: - generally inhibitory, involved in precise timing of neural circuits. - Augmentation disrupts coordinated patterns of neuronal activity preventing accomplishment of complex cognitive tasks (like conscious awareness) producing unconsciousness - Drugs: propofol, midazolam, etomidate, isoflurane, desflurane NMDA Receptors: - activated in conditions of repetitive firing, thought to mediate working memory, recall, attention. - Inhibition disrupts higher order cognitive function (working memory, attention) - drugs: ketamine, NO Glycine receptors: contributes immobility. Drugs: propofol, etomidate, thiopental Voltage gated Na channels: ketamine, propofol Adrenergic receptors: dexmedetomidine |
|
|
Adverse effects of general anesthetics
|
Cardopulmonary: ↓BP (↓CO, ↓SVR), ↓HR, ↓tidal volume (dose dependent), ↑apneic threshold, ↓hypoxic response, ↑RR
CNS: ↑cerebral vasodilation (↑flow, pressure) Biotransformation/toxicity: halothane hepatitis, fluoride tox (isoflurane), CO toxicity (desflurane) Malignant HTN: due to mutated ryanodine receptor (regulates Ca in SR of muscles). Causes hypercarbia, metabolic acidosis, tachycardia, rhabdomyolysis. Mostly inhaled agents. Tx: dantrolene (Ca blocker), ↓temp |
|
|
Local anesthetics kinetics
|
LA’s have hydrophobic/aromatic group separated from tertiary N (ester or amide), N can be protonated (weak base)
Distribution: all tissues, highest in skeletal muscle, also brain, liver, lungs, kidneys, spleen Mechanism: unprotonated form diffuses into neurons, is protonated, then blocks the cytoplasmic mouth of Na channels, stabilizing the channel in the inactivated state. - mechanism is shared w/ class 1 anti-arrythmics and AEDs and so can cause synergistic effects Metabolism: amides metabolized by liver (more systemic toxicity), esters quickly hydrolyzed by plasma esterases to PABA (less toxic) |
|
|
Adverse effects of local anesthetics
|
CNS toxicity: usually early, drowsiness may progress to excitation/seizures (due to high susceptibility of GABAergic CNS interneurons to LA’s: thin, unmyelinated axons), leading to coma and death
Cardiotoxicity: depressed cardiac activity and arrhythmias due to inhibition of ventricular and pace-maker cell action potentials (carried by NA channels). Synergistic effect w/ anti-arryhthimics (esp. Amides), potentiated by hyperkalemia (more Na channels inactivated) Hypotension: inhibition of perivascular smooth muscle Na channels, resulting in vasodilation and ↓BP PABA allergy: esters can be hydrolyzed to PABA, a known allergen |
|
|
Administration of epinephrine w/ local anesthetics
|
Benefits: used as a local vasoconstrictor to reduce blood flow (though might haves some other mechanism)
- prolongation of effect - reduced plasma concentration of LA - reduced surgical bleeding - intensified analgesia and anesthesia Risks: - in appendages w/o collateral circulation (ear lobe, distal extremities, nose, etc) can cause ischemic conditions |
|
|
Factors that influence effectiveness of local anesthetics
|
Diameter of nerve fiber: small and unmelinated nerves are more sensitive. C fibers (unmelinated) are most sensitive (A least), found in dorsal root ganglion meditating primarily pain. Order of block: pain> temp> touch> deep pressure> motor function
Firing rate, AP duration (use-dependent block): faster firing neurons are inactivated longer (better target). C fibers are higher frequency w/ long APs than A so are more sensitive Position of fiber in nerve bundle: central fibers (for distal structures) are less exposed, so Las effect more proximal structures first Local blood flow (value of epinephrine): particularly used w/ esters, not good in structures w/o collateral blood flow Liver function (for amides): hepatic dysfunction will reduce clearance of amides so must reduce dose Tissue pH (from inflammation): at acidic pH drug less drug is uncharged so less crosses the membrane so less nerve block |
|
|
Local anesthetics by duration of block
|
Short (20-45min): procaine (ester), benzocaine (ester)
Medium (1-2h): lidocaine (amide), tetracaine (ester) Long (3.5h in adult, 8 child): bupivacaine (amide) |
|
|
Local anesthetics by application/use
|
Topical: benzocaine, proparacaine (eye drops)
Local: procaine, lidocaine Regional: tetracaine, bupivacaine |
|
|
Procaine
|
= ester local anesthetic
Duration: short, 20-45min Use: local Tox: converted to PABA causing allergies (block action of sulfonamide) |
|
|
Lidocaine
|
= amide local anesthetic
Duration: medium (1-2h), 2h for spinal anesthesia, .5 for topical, 1h for local Use: local (spinal epidural, local topical anesthetic) Tox: systemic tox due to liver metabolism, synergistic w/ amiodarone |
|
|
Tetracaine
|
= ester local anesthetic
Duration: medium (slowly hydrolyzed), 2h for spinal, 30-60min for topical Uses: regional (spinal), topical anesthesia of nose and throat Side effects: mucous membrane irritation, urticarial, burning |
|
|
Bupivacaine
|
= amide local anesthetic
Duration: long (3.5 adult, 8 child) Use: regional (sometimes epidural for labor to reduce opioids) Side effects: arrhythmias (higher cardio tox b/c dissociates more slowly. New analogue has less), highly synergistic w/ anti-arrythics |
|
|
Common vestibular symptoms
|
Dysequilibrium (imbalance or unsteadiness while standing/walking): loss of vestibulospinal, proprioreceptive, visual, motor function, joint pain or instability, or psychological factors
Lightheadedness/presyncope: decrease blood flow to the brain Sense of rocking or sway in ship (Mal de Debarquement): vestibular system adapts to continuous, passive motion and most readapt to stable environment, or Anxiety Motion sickness: visual-vestibular mismatch Nausea/vomiting: stimulation of the medulla Oscillopsia (illusion of visual movement): spontaneous (acquired nystagmus), head-induced (severe, bilateral loss of vestibulo-ocular reflex) Floating/swimming/rocking/spinning inside head: anxiety, depression, somatiform disorders Vertical diplopia: skew eye deviation Vertigo (rotation, linear movement, or tilt): imbalance of tonic neural activity to vestibular cerebral cortex |
|
|
Causes of acute dizziness
|
All are spontaneous, exacerbated by head movements:
- Vestibular neuritis: vertigo, disequilibrium, N/V, oscillopsia - Labyrinthitis: vertigo, disequilibrium, N/V, oscillopsia, hearing loss, tinnitius - Wallenberg Infarct (lateral medullary syndrome): vertigo, disequilibrium, N/V, oscillopsia, tilt, lateropulsion, ataxia, crossed sensory loss |
|
|
Causes of chronic dizziness
|
Bilateral vestibular defect or >3d from unilateral vestibular defect:
- disequilibrium, occasionally oscillopsia - induced by head movements or walking (esp. in the dark or on uneven surfaces) Mal de Debarquement: - rocking or swaying as if on a boat - spontaneous while lying or sitting, rarely occurs while in motion Oscillopsia: - subjective illusion of visual motion - spontaneous w/ eyes open Anxiety/depression: - lightheadedness, floating or rocking - induced by eye movements w/ head still |
|
|
Causes of vertigo spells
|
Benign paroxysmal positional vertigo: vertigo, lightheadedness, nausea. Positional (lying down, sitting up or turning over in bed, bending forward)
Orthostatic hypotension: lightheadedness. Positional (standing up) Transient ischemic attack: vertigo, lightheadedness, disequilibrium. Spontaneous Migraine: vertigo, dizziness, motion sickness. Usually movement induced Panic attack: dizziness, nausea, diaphoresis, fear, palpitations, paresthesias. Spontaneous or situational Motion sickness: nausea, diaphoresis, dizziness. Movement induced, usually visual-vestibular mismatch Meniere’s disease: vertigo, disequilibrium, ear fullness from hearing loss and tinnitus. Spontaneous, exacerbated by head movements. |
|
|
Acute vestibular loss
|
Causes: unilateral vestibular neuritis (HSV reactivation in CN8), labyrinthitis, acoustic neuroma, multiple sclerosis, infarct to vestibular system, gentamicin ototoxicity
Symptoms: spontaneous vertigo, disequilibrium, N/V, oscillopsia aggravated by head movements Signs: slow phase nystagmus towards lesion, quick phase nystagmus away from lesion, torsional nystagmus away from lesion, deviation to left when walking straight, slow gait, horizontal and mild torsional nystagmus enhanced when fixation blocked, abnormal VOR on lesion side when head thrust performed, normal Romberg test Management: - first 3 days: bed rest, vestibular suppressants (Phenergan), prednisone and acyclovir (if viral) - >3d: vestibular rehabilitation |
|
|
Benign Paroxysmal Positional Vertigo (BPPV)
|
= peripheral vestibular disorder causing spells of vertigo that last <1min when patient moves head
Cause: spontaneous, trauma, labyrinthitis, ischemia to the labyrinth Symptoms: poor balance, vertigo, lightheadedness that last for a few seconds upon head movement. Commonly getting up from bed, turning over in bed, lying down, moving head back (dentist, bookshelf). Less commonly: trouble walking, nausea, blurred vision Mechanism: displacement of calcium carbonate crystals (otoconia) in the semicircular canals (they have broken free of the macula of the utricle). Otoconia in posterior SCC (canalithiasis) or cupola of posterior SCC (cupolithiasis) Tx: reposition otoconia via head maneuvers (canalith repositioning therapy) |
|
|
Vestibular migraine
|
= type of migraine
Symptoms: tinnitus (high pitch), ear fullness (ache), phonophobia, photophobia, spontaneous vertigo (rare, <1h), visual auras, motion sickness. Tend to improve w/ short nap Mangement: - avoid tyramine (chocolate, cheese, nuts), nitrates, aspartame, MSG, aged alcohol - meds: daily aspirin, beta-blockers, tricyclics |
|
|
Meniere’s syndrome
|
= idiopathic disorder of the inner ear, likely related to excess endolymphatic fluid
Symptoms: tinnitus (low pitch, roar), ear fullness or hearing loss, spontaneous vertigo (hours). Naps do not help, no visual auras or motion sickness Management: - Na restriction (<2g/d), reduced caffeine and alcohol - meds: diuretics, acetazolamide - surgical: endolymphatic shunt, transtympanic gentamicin injection |
|
|
Physical therapy for chronic imbalance
|
Good for:
- disuse equilibrium: functionally intact vestibular apparatus, fear of fall and limited movement cause the problem Not good for: - vestibular hypofunction: permanent neural damage (neuritis, ototoxicity) |
|
|
Genetic variations related to neurodevelopmental disorders
|
Chromosome aneuploidy: trisomy 13, 18, or 21
Structural chromosome defects: - partial trisomy - translocation: Robertsonian translocation (partial t21,14) - deletion (LIS1): Miller-Dieker syndrome (large d17 from unbalanced translocation: severe lissencephaly (smooth brain, unformed gyri), microcephaly, bitemporal narrowing, vertical furrows on head, clinodactyly, cardiac defects), Isolated lissencephaly syndrome (small d17: less severe lissencephaly) Subchromosomal abnormalities: - genomic disorders (contiguous deletion syndrome): recurrent imbalances (duplication/deletion) due to segmental duplications - subtelomere rearrangements: often unbalanced translocations Monogenic disorders: AR, AD, X-Linked (fragile X mental retardation: CFF expansion causing hypermethylation of FMR1 gene and reduced transcription) Complex (polygenic) disorders |
|
|
Spina bifida
|
= non-fusion of the embryonic halves of the vertebral arches
- a/w neural tube defects: incomplete development of the brain, spinal cord, and/or meninges - most common neural tube defect in US (1.5-2K/4mil), first trimester folate reduces risk by 70%, valproate increases risk 3x |
|
|
Neural tube defects
|
- all a/w spina bifida
Cranial NTDs: anencephaly, exencephaly (results when neuropore does not close). Spinal NTDs: open spina bifida, occurs when posterior neuropore doesn’t close, usually involves tethering - myelocele: protrusion of spinal cord through defect in vertebral arch - myelomeningocele: hernia containing both meninges and spinal cord, may have inappropriate tethering of neural structures to adjacent structures - myeloschisis: open spinal cord (plate-like mass of nervous tissue w/o overlying membrane). Child is very prone to infection (meningitis) |
|
|
DSM IV Axes of Mental Disorders
|
Axis I: Psychiatric Disorders
- depression, anxiety, bipolar, OCD Axis II: Personality Disorders and Mental Retardation - obsessive-compulsive personality Axis III: Medical Disorders - general medical problems that the psych patient has (hypothyroidism, lupus, HTN, etc) Axis IV: Global Assessment of Function |
|
|
Psychiatric Mental Status Exam (MSE)
|
ABC STAMP LICKER
Appearance: grooming, clothing, hygiene, accessories (tattoos, piercings) Behavior: focus, distraction, appropriate response, interaction Cooperation: answering questions, truthful, evasive, manipulative Speech: spontantous, fluent/broken, volume, emotional tone (prosody) Thought: form (pattern of language, organized, neologisms, word approximation, clang associations, word salad), content (appropriate, congruent w/ mood, delusions (bizarre/non-bizarre)) Affect: full/limited, quality (euthymic, irritable, restricted, blunted, euphoric), congruent to situation Mood: (according to patient) Perception: hallucinations (type) Level of Consciousness: alert, intoxicated, oriented (time, place, person), stable/fluctuating Insight: aware of current situation, understanding of symptoms/treatment Cognition: attention, judgment, problem solving, speed of processing, verbal/spatial ability Knowledge: word meanings, information, recognition of face/objects Endings: suicidal, homicidal, violent Reliability: statements consistent w/ observations, family info, plausible |
|
|
Monoamine oxidase inhibitors (MAOIs)
|
Mechanism: inhibition of MAO receptors preventing NE and DA breakdown. (A receptors: CNS, liver, gut, skin. B receptors: CNS, liver, platelets)
- phenelzine/tranylcypromine: irreversible, non-specific (A &B) - Selegiline: irreversible, selective (B) - Moclobemide: reversible, selective (B) Side Effects: - serotonin syndrome via drug interactions (↑serotonin): flushing, diaphoresis, N/V, diarrhea - require tyramine free diet (avoid excess DA production). Avoid: cheese (except cream/cottage), meats (beef, liver, cured meats), fish, vegetables (avocados, fava beans, sauerkraut), yeast extracts, wine/beers, chocolates, coffee |
|
|
Tricyclic antidepressants (TCAs)
|
= imipramine, amitriptyline, nortryptyline, desipramine, clomipramine
Mechanism: selectively inhibit NE reuptake (despipramine) OR inhibit 5-HT reuptake (clomipramine) OR both (imipramine) Side Effects: - anti-adrenergic effects: dizziness, sexual dysfunction, orthostasis - anti-muscarinic/cholinergic effects: dry mouth, constipation, confusion or delirium in the eldery - Anti-histaminergic effects: sedation, weight gain - Quinidine-like effects on cardiac muscle: prolonged QT interval, risk for torsades de pointes w/ ventricular tachycardia, fibrillation, death - High lethality in overdose |
|
|
Selective serotonin reuptake inhibitors (SSRIs)
|
= fluoxetine, sertraline, paroxetine, citalopram, escitalopram, fluvoxamine
Mechanism: inhibit reuptake of 5-HT without significant effects on reuptake of NE or DA (serotonin specific) Side effects: - generally much less severe than TCAs or MOIs - GI disturbances - sexual dysfunction |
|
|
Serotonin-norepinephrine reuptake inhibitors (SNRIs)
|
= venlafaxine, desvenlafaxine, duloxetine
Mechanism: inhibits NE and 5-HT reuptake by binding 5-HT transport at low concentrations and NE transporter at higher concentrations Side effects: - sexual dysfunction - ↑BP - potential lethality in overdose (but not as risky as TCAs) |
|
|
Atypical antidepressants
|
Mirtazapine:
- 5-HT2 and α2 receptor antagonist (5-HT and NE) - somnolence Trazadone: - 5-HT2A receptor antagonist - somnolence, priaprism Vilazodone: - 5-HT1A receptor partial agonist and modest inhibitor of 5-HT reuptake - SSRI-like effects: anorgasmia, nausea, diarrhea, insomnia Bupropion: - weak inhibitor of NE, 5-HT, and DA reuptake - seizures (especially in bulimic patients) |
|
|
Benzodiazepines
|
Indications: anxiety (panic attacks, PTSD), sedation or pre/co-anesthesia , insomnia, hypnosis, muscle relaxation (spasticity), anti-convulsant, withdrawal of sedative-hypnotics
Adverse effects: bizarre dreams, ↓respiratory drive, potentiate respiratory/CNS depression from other drugs (ETOH, barbituates), cross-tolerance to other sedative-hypnotics Mechanism: forms complex that potentiates the inhibitor action of GABA at the GABA-A receptor (Cl- ion gate, causing hyperpolarization). Does not work w/o GABA (unlike barbituates, so more specific) Classes: - long-acting/stead: diazepam (Valium): T1/2 of 30h, converted to intermediates w/ long T1/2, glucoronidated then excreted. Good for anxiety - short acting (triazolam): T1/2 of 3h (avoids daytime hangover), converted to active metabolite (short T1/2), glucoronidated and excreted. Good for sleep disorders. Tolerance/dependence: - Less tolerance (Valium): longer acting active/metabolites so greater time to re-adapt to drug absence. Used to aid withdrawal from alcohol or barbituates - More tolerance: short acting may cause more physical dependence due to quick loss of activity - withdrawal can be precipitated by administration of flumazenil (benzodiazepine receptor antagonist) |
|
|
GABA receptor modulators
|
Agonists (diazepam, triazolam): enhance GABA mediated Cl- conductance and neuronal inhibition
Antagnoist (Flumazenil): binds benzo receptor, competitively inhibits benzos (reducing benzo effect, inducing withdrawal). No effect in isolation Inverse agonist (β-carboline carboxyethyl ester): binds benzo receptor at same site but reduces GABA mediated CL- conductance and neuronal inhibition. Causes anxiety, muscle spasms, pro-convulsive state. |
|
|
Non-benzodiazepine benzodiazepine receptor antagonists (NBRAs)
|
= short-acting hypnotic agents
Indications: acute or chronic Tx of sleep disorders Mechanism: bind to subtypes of the benzo receptor and facilitate GABA-mediated Cl- conductance and neuronal inhibition - rapidly absorbed with no active metabolites: generally few anti-anxiety effects at low hypnotic doses Types: - Zolipidem (Ambien): relatively selective for Type 1 benzo receptor found in brain areas a/w sleep. Rapidly absorbed/eliminated, T1/2 2.6h. Loses effect when taken chronically - Eszopiclone (Lunesta): binds all 3 benzo receptor types (selectivity mechanism unclear). Rapid onset, T1/2 of 6h, no reduction in effect at 6mo |
|
|
Neural mechanism of action for Ethanol
|
- dissolves in lipid bilayer of plasma membranes, reducing membrane viscosity and disruption protein function
- ↑ GABA-mediated Cl- conductance through GABA-A receptor - ↓ glutamate-mediated cation conductance through NMDA receptor subtypes - ↑ serotonin-mediated cation conductance through 5HT3 receptors on inhibitory neurons Pharmokinetics: - max [blood] time: 30-90min - elimination: 90-98% by oxidation - follows zero-order kinetics: 7-10g/h, 5h to metabolize 4oz - oxidation by: NAD+ dependent alcohol DH, NADPH-dependent microsomal ethanol oxidizing system. Both form acetaldehyde, which is converted to acetate by aldehyde DH Effects: - dose-related progression of CNS depression (similar to other sedative-hypnotics) - most sensitive structure: polysynaptic reticular activating system and cerebral cortex: depression of these areas results in euphoria, disorganized thought, dulling of performance |
|
|
Therapeutic uses of sedative-hypnotic drugs
|
Have dose-related progressive effects on:
- sedation (anti-anxiety effects) - behavioral disinhibition - ataxia/nystagmus - sleep (hypnosis) - anesthesia - eventually: coma, respiratory depression, CV depression |
|
|
Alcohol Tolerance and Dependence
|
Pharmacokinetic tolerance:
- related to induction of metabolic enzymes (variable as overall liver function decreases, leading to reverse tolerance): ↑Alcohol DH, ↑NAD+ synthesis, ↑MEOS activity Pharmacodynamic tolerance: - involves adaptation of the brain to ETOH-related neurological changes (poorly understood mechanism): ↓sensitivty to membrane fluidizing effects of alcohol, ↓GABA-A receptors (via down regulation), ↑NMDA/glutamate receptors leading to enhance excitatory drive - chronic ingestion leads to physical dependence; withdrawal symptoms occur upon cessation no reduction (can lead to convulsions) |
|
|
Neurological consequences of alcohol ingestion
|
Peripheral neuropathy, parasthesia, tremor
CNS deficits leading to dementia: - ventricular enlargement (brain shrinkage), ↑CSF volume - ↓white matter, especially in corpus callosum - neuronal loss, esp in frontal and entorhinal cortex, hypothalamus, and cerebellum - shrinkage of neuronal nuclei Ethanol neurotoxicity: ethanol is directly toxic to neurons Wernicke-Korsakoff syndrome (thiamine deficiency from poor nutrition/transporter activity/utilization, ↑dephosphorylation): thiamine triphosphate needed for NT synthesis, nucleic acids, co-enzymes, steroids, glutathione, glycolysis, TCA cycle, AA’s, others Hepatic encephalopathy: liver failure causing urea accumulation in the brain Other (non-neuro): skeletal and cardiomyopathies, acute/chronic pancreatitis, induction of liver enzymes, cirrhosis, cancer (oral, esophageal, splenic), teratogenicity/fetal alcohol syndrome |
|
|
Treatment for alcoholism
|
Disulfiram (anatabuse): irreversible inhibitor of Ald DH. Increase acetaldehyde (hangover) causeing immediate, severe negative reaction to alcohol intake. In 5-10min: flushing, ↑HR, SOB, N/V, headache, visual disturbance, mental confusion, postural syncope, circulatory collapse
Naltrexone: opiate receptor antagonist: Decreases rewarding effect of alcohol by release of endogenous opioids Acamprosate (Campral): reduces glutamate neurotransmission (may ↓relapse in detoxed patients) Baclofen: GABA-B agonist that decrease withdrawal symptoms and promotes abstinence by reducing alcohol-related anxiety and cravings Benzos: if patient is tolerant to alcohol, substitute to taper off CNS dependency ALL should be used in conjunction with cognitive/behavioral therapy |
|
|
Lithium
|
Indications: bipolar I/II/NOS (treats all phases: mania, depression, maintenance), unipolar depression (used w/ traditional antidepressant, synergistic)
Mechanisms (hypothesized): interferes w/ recycling of phosphatidylinositol bisphosphate PIP2 pathway (resulting in inhibition of signaling from pathogenic neurons), inhibits GSK-3β and activates Wnt pathat causing inositol depletion (impt for brain development and remodeling), inhibits adenylyl cyclase (side effect: diabetes insipidus) Side effects: polyuria, polydipsia, nausea, slowed cardiac conduction, tremor, slowed cognition (esp. in pts w/ significant anxiety), acne, psoriasis, rash Adverse effects: over dose (low therapeutic index, very lethal), nephropathy, thyroiditis/hypothyroidism, Ebstein’s anomaly (teratogenic malformation of the tricuspid valve) Excretion: renal (avoid in kidney failure). Inhibited by NSAIDs, thiazide diuretics, ACE inihibtors, Ca channel blockers. Halve does for elderly |
|
|
Indications for antipsychotics
|
Psychotic disorders: schizophrenia, schizophreniform disorder, schizoaffective disorder, brief reactive psychosis
Mood disorders: major depression (w/ psychotic features or as augmentation), bipolar disorder (all phases) Delirium and dementia: helps w/ agitation, hallucinations, impulsivity Stimulant-related psychosis: cocaine and meth Autism spectrum disorders Developmental disorders w/ impulsive aggression Eating disorders: newer ones can aid weight gain Personality disorders: borderline and narcissistic types Psychosis due to general medical condition |
|
|
Mechanism of antipsychotics
|
= “neuroleptics,” “major tranquilizers”
General: domapine blockade Types: D2 receptor antagonists (“typical”), D2/5-HT2a receptor antagonists (“atypical”) Target areas: caudate, putamen, nucleus accumbens, amygdala, hippocampus, cerebral cortex Effects: anti-psychotic, anti-manic, anti-agitation, anti-emetic Pharmokinetics: large volume of distribution (lipophilic), significant plasma protein binding, metabolized by Cyt P450 system w/ variable activity of metabolites. Excreted as glucuronides |
|
|
Classes of antipsychotics
|
Structural classes: phenothiazines, thioxanthenes, butyrophones
Potency classes: - low (200mg/d): chlorpromazine - mid (20-50mg/d): perphenizine - high (2-4mg/d): haloperidol Atypical (block 5-HT2a receptor in addition to D2): quetiapine, clozapine, olanzapine, ziprasidone, risperidone Aripiprazole: partial D2 and 5-HT1a agonist, and 5-HT2a antagonis |
|
|
Side/Adverse effects of antipsychotics
|
Side effects:
- parkinsonian affects (extrapyramidal D2 blockade): bradykinesia, tremor, dystonia - Sedation (from histamine antagonism) - postural hypotension (from α1 blockade) - dry mouth, urinary retention, cognitive impairment (muscarinic/cholinergic blockade) - hyperprolactinemia (D2 blockade) Toxicity/Adverse effects - metabolic syndrome: weight gain (hyperglycemia, hyperlipidemia, HTN), DM and progressive CV dz - Cardiotoxicity: torsades des pointes (more risk w/ electrolyte abns) - Seizure: lowers seizure threshold (esp Clozapine) - Agranulocytosis: low absolute neutrophil count - Tardive Dyskinesia (orofacial and extremity dyskinesias): from longterm use (4-5% risk/y), potential from DA receptor upregulation/sensitization - Neuroleptic malignant syndrome (classically high potency in dehydrated pt): muscular rigidity, fever, leukocytosis, altered mental status, myoglobinemia. Potentially fatal if untreated (dantrolene and bromocriptine, or ECT) |
|
|
Mania
|
= period of abnormality and excessively elevated, expansive or irritable mood lasting one week (or less if hospitalization required)
DIG FAST Distractability Insomnia Flight of ideas Activity: goal directed Speech: pressured Thoughtlessness: impulsivity |
|
|
Psychosis
|
= loss of contact with reality resulting in severe functional and social impairment
Symptoms: - Hallucinations - Delusions - Formal thought disorder |
|
|
Schizophrenia
|
Dx:
-Characteristic symtopms: 2+ symptoms for >1mo (or less if treated. >1mo if autism present): delusions, hallucinations, disorganized speech, grossly disorganized or catatonic behavior, negative symptoms (affective flattening, alogia, avolition) - social/occupational dysfunction - signs of disturbance >6mo - Exclusion of schizoaffective and mood disorders, also substance abuse and general medical disorders Mechanism (hypothesized): excessive dopamine action. Supported by drug tests (illicit: PCP, amphetamines, cocaine), treatment correlates to parkinsonian symptoms Tx: antipsychotic |
|
|
Opiates
|
= morphine and codeine (derived directly from poppy plant)
Indications: pain management, pulmonary edema, cough suppression, constipation, detoxification (naloxone/naltrexone used to reverse opioid analgesia) Mechanism: agonize G-protein coupled opioid receptors → post-synaptic K+ efflux or presynaptic Ca+ influx → inhibition of firing/NT release within the anterolateral system (targets the periaquductal grey w/ many μu type opioid receptors) Side effects: respiratory depression, sedative effect |
|
|
Marijuana
|
= contains cannaboids (THC)
Mechanism: agonize endocannaboid receptors → post-synaptic K+ efflux or presynaptic Ca+ influx → inhibition of neuronal firing/NT release Effects: euphoria and mild hallucinogenic effects |
|
|
Dissociative anesthetics
|
= ketamine and phencyclidine
Indications: used as anesthetics in humans and animals Mechanism: NMDA receptor antagonists, facilitates dopamine release Effects: high pain tolerance (aware but not bothered by pain), psychosis, confusion, agitation, vertical nystagmus, hyperthermia, heat intolerance |
|
|
Cathinone Analogs
|
= mephedrone, methylone, “bath salts”
Mechanism: effects on serotonin and dopamine Effect: moderate euphoria to psychosis and extreme agitation/aggression |
|
|
MDMA
|
= ecstasy
Mechanism: modified amphetamine that modifies the 5-HT system Effect: euphoria, empathogenicity, hallucinations. Interacts w/ SSRIs and MAOI |
|
|
Methylxanthines
|
= theophylline, theobromine, caffeine
Mechanism: inhibit phsophodiesterase from breaking down cAMP Effects: increased alertness, inotropic effects, mild diuresis, increased acid secretion in gut Side effect: increase heart rate and contractility |
|
|
Nicotine
|
Mechanism: stimulates nicotinic cholinergic receptor, affects autonomic ganglia as well as CNS
Effects: Low doses: euphoria, alertness High doses: respiratory paralysis Rapid development of tolerance and dependency |
|
|
Cocaine
|
Indications: illicit, topical anesthetic for eye surgery
Mechanism: blockade of monoamine transporters leading to prolonged effect of catecholamine (DA, NE). Peripherally blocks Na+ channels, causes vasoconstriction Effects: pressor (stroke/MI even in young/healthy individuals), increased alertness, psychosis, damage to nasal septum |
|
|
Amphetamines/methylphenidate
|
Indications: ADHD, narcolepsy
Mechanism: indirect agonists that enhance the effect of catecholamines Effects: increased euphoria, alertness, psychosis, cardiac problems, neurotoxicity |
|
|
Major Depressive Episode
|
= 5+ symptoms for >2 wks, representing a change in functioning. Must include depressed mood and/or anhedonia (loss of interest/pleasure)
9 cardinap symptoms (D SIG E CAPS): depression, sleep disturbance (+/-), Interest (anhedonia), Guilt (worthlessness), Energy (fatigue), Concentration (or decision making capacity), Appetite (+/-), Psychomotor agitation or retardation, Suicide (or recurrent ideations) Additional criteria: - symptoms must cause clinically significant impairment - symptoms not due to direct physiologic effects of a substance or medical condition - symptoms not better accounted for bereavement (more than 2mo after loss) Subtypes: chronic, catatonic features, melancholic features, atypical onset, postpartum onset |
|
|
Specifiers/Subtypes of Major Depressive episodes
|
- chronic: full criteria met continuously for >2 years
- catatonic features (2+): motoric immobility (cataplexy, waxy flexibility, stupor), excessive motor activity (purposeless), extreme negativism or mutism, peculiarities of voluntary movements (posturing, stereotypes, grimacing), echolalia or echopraxia - melancholic features: (1+) loss pleasure in all activities, lack of reactivity to usually pleasurable stimuli -OR- (3+): distinct quality of depressed mood, worse in morning, early morning awakening, psychomotor agitation or retardation, anorexia or weight loss, excess or inappropriate guilt - atypical onset: A. mood reactivity. B. (2+) weight gain/increased appetite, hypersomnia, leaden paralysis, interpersonal rejection sensitivity. C. Not melancholic or catatonic - postpartum onset: <4wks post-partum |
|
|
Major Depressive Disorder
|
Criteria:
- presence of major depressive episode: single or recurrent - not better accounted for by another psychiatric disorder - no history of manic episode Associated symptoms: - anxiety (very common): obsessive rumination, panic attacks - somatization (fairly common): body aches/pains, headaches, GI distress - Psychosis (uncommon): hallucinations/delusions/catatonia, a/w severe illness, often requires specific treatment |
|
|
Dysthymia
|
= chronic, low grade depression
Criteria: depressed mood for most of the day, for more days than not, for a period of 2 years. Symptoms are not absent for more than 2mo. 2+ symptoms are present: - appetite (+/-), sleep (+/-), low energy, low self-esteem, poor concentration/difficulty making decisions, feelings of hopelessness Potential Causes: - unresolved major depression - personality disorder (depressive personality) - non-mood disorders |
|
|
Adjustment disorder
|
Criteria:
- Development of emotional or behavioral symptoms in response to an identifiable stressor occurring within 3mo of the onset of the stressor - Symptoms are clinically significant: marked distress in excess of what would be expected -OR- significant impairment of social or occupational functioning - Disturbance does not meet criteria for another axis I or II disorder - Symptoms do not represent bereavement - Once stressor has ended symptoms do not persist for >6mo May be comorbid w/ anxiety (symptoms of worry, nervousness, jitteriness) or mix with anxiety and depressed mood (anxiety, sad mood, tearfulness, hopelessness) |
|
|
Mood disorder due to general medical condition
|
Criteria:
- prominent and persisitent disturbance in mood: depressed/anhedonic -OR- elevated/expansive - Evidence due to direct physiological consequence of medical condition - not better accounted for by other mental illness - not in delirium - clinically significant distress or impairment Common medical conditions a/w depression: - CNS disease: stroke, dementia (AD, HD, PD, BE), traumatic brain injury, temporal lobe epilepsy, tumor, infections (encephalitis, HepC, HIV) - endocrine disease: hypothyroidism, cushing’s, hyper/hypo-parathyroidism - cancer: pancreatic carcinoma - Cardiovascular disease - rheumatologic disease: SLE, MS, psoriasis, RA, osteoarthritis |
|
|
Substance induced mood disorder
|
Criteria:
- prominent and persistent disturbance in mood: depressed/anhedonic or elevated/expansive - evidence that symptoms developed: during/within 1mo of substance use, or substance etiologically related to mood symptoms - not better accounted for by other diagnoses - not delirium - clinically significant distress or impairment Common pharm cause of depression: - interferon/immunomodulators, corticosteroids/hormones, anti-neoplastic agents, neurologic agents (levodopa, AChE-I, anti-epileptics), antipsychotics, sedative-hypnotics, analgesics (opioids, NSAIDs), anti-bacterials (sulfonamides), antifungals, CV drugs (β-blockers, digoxin) Common intoxicants causing depression: - chronic alcohol abuse, cocaine/methamphetamine/MDMA (due to monamine depletion), sedative-hypnotics, opiates |
|
|
Evaluation of depressive symptoms
|
1. Assess for SIGE CAPS
2. R/o medical causes (hypothyroid) 3. Assess for recent death (grief) 4. Assess for major life changes/new medical diagnosis (grief) 5. Assess for recent births (post-partum) 6. Assess for recent medication changes (substance induced) 7. Assess for illicit drug use and hx of drug abuse (substance induced) 8. Assess for manic episodes (bipolar) |
|
|
Risk Factors for major depression
|
Family history of depressive disorders
Prior personal history of a depressive disorder Female gender Certain personality traits: high neuroticism, low extroversion Trauma: loss of parents at early age, childhood abuse, life stressor (bereavement, chronic financial problems) Disorders: - other psychiatric disorders: alcohol or drug abuse, anxiety - neurologic disorders: parkinson’s, alzheimer’s, epilepsy, stroke - other medical disorders: diabetes, heart disease, hypothyroidism, immune deficiencies |
|
|
Theory of Depression
|
Biological sensitivity makes patients more susceptible to exogenous stressors
- genes: multiple susceptibility alleles, each of small effect (polygenic and heterogeneous) - cells: subtle molecular abnormalities - systems: abnormal mood regulation and information processing - behavioral endophenotypes: complex functional interactions and emergent phenomena Environmental factors: - proximal risk factors: stressful life events - distal risk factors: relatively severe stressors experienced early in life (child abuse) may up-regulate the stress vulnerability of individuals to depressogenic events for decades |
|
|
Monoamine deficiency hypotheses of depression
|
1: NE deficiency is causally related to MDD symptoms
- depletion of reserpine can precipitate depression, MDD patients have ↓[NE metabolites] in urine/CSF, ↑cortex β-adrenergic receptors in suicide patients, NE reuptake inhibitors are effective treatment 2: Dopamine plays a role in physiological and behavioral areas of depression - high rate of Parkinson’s comorbidity (low DA), psychostimulant withdrawal a/w mood change/depression sx, depressed pts have ↓DA transporters, MAO inhibitors effective 3: Relative 5-HT deficiency linked to MDD - low 5HT metabolites in CSF of MDD pts, ↓5-HT transporters in MDD pts, ↑5-HT2 receptors in suicide pts, SSRIs effective Overall: monoamine alteration contributes but does not fully explain depression (67% do not remit w/ single antidepressant, 33% not w/ multiple) Other disturbances of transmitter hormone systems identified: cortocotrophin-releasing factor , thryroid, glutamate |
|
|
Structural/functional neural change in depressed patients
|
Regional atrophy (rather than brain death in neurodegerative dzs)
- ↓volume of frontal lobe and hippocampus - ↓neuropils, dendritic spines (↑dendritic spines in amygdala, esp w/ chronic stress - ↓glial cells (oligos) Similar effects seen in patients under chronic stress or taking glucocorticoids: social/environmental stress interferes/inhibits the maturation of neuronal cells from their primitive form to more expansive/connected forms. Brain patterns reversed by antidepressant treatment On PET: (not uniform: like due to variable genetic, biochem, social factors) - hypoactive frontal lobe and cingulate gyrus |
|
|
Behavioral Activation (BA) therapy
|
= brief, solution-focued intervention for depression
Aim: change behaviors that maintain or exacerbate depression, reduce avoidance and withdrawal Intervention: - get pt moving and doing things that make them feel competent. - Use graded task assignments (break into mini-steps to allow for sense of accomplishment after each) - have pt create hierarchy of reinforcing activities, ranked by difficulty - track goals independently along w/ clinician Assessment: - Beck Depression Inventory (before and after) |
|
|
Cognitive Behavioral Therapy (CBT)
|
= psychotherapeutic approach to psych disorders that addresses dysfunctional emotions, maladaptive behaviors, cognitive processes and contents through goal-oriented, explicit systematic procedures
Indications: mood, anxiety, personality, eating, substance abuse, tic, and psychotic disorders Intervention: replace maladaptive coping skills, cognitions, emotions and behaviors with more adaptive ones by challenging and individuals way of thinking/reacting /habits/behaviors. - The more severe the patient’s disorder is the less likely they will be able to engage in CBT. Start with behavioral activation (change) |
|
|
Interpersonal Therapy
|
= therapy for depression that focuses on the interpersonal relationships of the patient
Aim: improve communication patterns and how people relate to each other Indicated for patients that don’t like the dynamic approaches or are daunted by the ‘homework’ demands of CBT |
|
|
5 Phases of FDA drug approval
|
Preclincial: drug is studied in animals or chemically in lab. Toxicology and bioavailability studies are done.
Phase I (Safety and bioavailability): first human subjects, usually normal controls (except chemo studied in end-stage patients) Phase II (dosing and treatment strategies): safety data continues to be gathered Phase III (efficacy): - must demonstrate efficacy in 2 pivotal trials - usually placebo controlled unless placebo would pose risk to patients (then use current standard of care) Phase IV: post-market surveillance FDA approval: package inserts mandated by FDA contain all information. Does not defined standards of treatment |
|
|
Clinical milestones in treatment of depression
|
Goals: looking for response, remission, and recovery
Response: - clinically significant reduction of symptoms: 50% reduction in baseline measure on Ham-D and MADRS - not fully symptomatic but with more than minimal symptoms Remission: goal of acute treatment - pt no longer meets syndromal criteria (back to where they were before depression) - no or minimal symptoms: Ham-D <8 or MADRS <10 - functional improvement in employment and relationships - on first exposure to antidepressant 60% remiss, after past exposure only 40% - treatment failure: inadequate medication trials, pt genetics, pt early life trauma, medical comorbidities, comorbidity w/ personality disorder Recovery: - >2mo full remission (symptomatic and functional): if <2mo, then have relapsed - Defined as fully return to previous level of function Treatment resistance: <50% reduction in baseline function. Treatment non-response: <25% improvement. Typically will fail 2 adequate full-dose trials (2 SSRIs or 2 classes of meds). Refer at this point. Pseudo-resistance: resistance to treatment due to incorrect diagnosis or wrong expectations of patient/provider, or inadequate compliance/dose/effect |
|
|
Clinical Anxiety
|
= anxiety that interferes with functioning in family, work, or social situations or causes marked distress
Epi: often comorbid w/ depression/substance abuse, most have multiple anxiety disorders, 18% of US population, increased by 1SD since 1950’s (increased mobility, reduction in social support, increased perception of danger), 75% manifest by age 22, don’t see treatment (“part of personality”) - Symptoms: diffuse, unpleasant, vague sense of apprehension occurring in anticipation of internal/external danger. Must be distinguished from Fear (state of intense apprehension/agitation in face of imminent danger/threat) and Worry (excess negative thinking of inaccurate predictions that catastrophe is likely to occur). May also look like mania. |
|
|
Specific phobia
|
= excessive fear of an object or situation every time it is confronted
Epi: lifetime prevalence of 12.5%, generally chronic, often not treated, increase risk for other mental disorders (depression, bipolar, etc) - Animal/environmental/injury phobias tend to arise in childhood, while situational ones are more bimodal Physiology: increased autonomic activation upon approach of stimulus (↑HR, skin conductance) except for BII phobias which involve biphasic physiologic response (sympathetic arousal followed by overcompensation by parasympathetic hypotension and bradycardia → fainting) Differential: share characteristics of panic disorder, agoraphobia. Key aspect is limitation to specific, circumscribed situations DSM criteria: - persistent fear that is excessive or unreliable, cued by the presence of anticipation of a specific object or situation - exposure to phobic stimulus almost invariably invokes an immediate anxiety response: situationally bound or situiationally predisposed Panic Attack - person recognizes that the fear is excessive or unreasonable (may be absent in children) - phobic situation is avoided or endured with intense anxiety or distress |
|
|
Social Phobia
|
= excessive fear of embarrassment in social situations
Age of onset: mid-teens for generalized, early 20’s for non-generalized, possibly bimodal (~5, and mid-teens) Course: low natural recovery rate (37% over 12y), low rates of treatment-seeking (though highly treatable), often comorbid w/ depression DSM criteria: - Marked, persistent fear of one or more social or performance situations in which the person is exposed to unfamiliar people or to possible scrutiny by others - Exposure to the feared social situation almost always provokes anxiety - The person recognizes the fear is excessive or unreasonable - Feared social or performance situations are avoided or endured w/ intense distress - Avoidance, anxiety, or distress significantly intereferes w/ the persons general functioning or quality of life - Specific (phobic of one type of situation, like public speaking) vs. generalized subtypes (phobic across multiple situations) |
|
|
Generalized anxiety disorder
|
= excessive worry about potential future events (car, relationship, school, savings) causing significant distress for at least 6mo. (brief worry about stressor would be adjustment disorder + anxiety)
Symptoms (3+ present more days than not): restlessness or feeling keyed up, easily fatigability, difficulty concentrating or mind blanking, irritability, muscle tension, sleep disurtbance Dx: GAD-7 scale |
|
|
Panic Attack
|
= intense bout of anxiety developing abruptly and peaking within 10min, usually lasts <30min
A/w: Panic disorder, OCD, PTSD, specific and social phobias Symptoms (PANIC): Palpitation, Abdominal pain, Nausea, Increased perspiration, Chest pain/Choking/Chill |
|
|
Panic disorder
|
= Recurrent (2+) unexpected panic attacks
Symptoms (4+, STUDENTS FEAR 3C’s): Sweating, Trembling, unsteadiness/dizziness, Derealization/depersonalization, Elevated HR, Nausea, Tingling (paresthesias), Shortness of breath, Fear of dying, Fear of losing control, Fear of going crazy, Choking, Chest pain, chills - at least 1 attack is followed by 1mo of: concern about additional attacks, worry about implication of attacks/consequences, or change in behavior related to attacks (typically agoraphobia) Categories: panic disorder +/- agoraphobia, or agoraphobia w/o panic attacks don’t get full attacks but avoid situations that might trigger symptoms |
|
|
Agoraphobia
|
= anxiety about being in places in which escape might be difficult or embarrassing, or where help might not be available, due to fear of having a panic attack or panic-like symptoms
Typical fears: outside home alone, on a bridge, in a crowd, travel in bus/train/plane DSM subtypes: panic disorder +/- agoraphobia, agoraphobia w/o panic disorder |
|
|
Treatment of Anxiety
|
CBT: addresses cognitive errors causing inability to cope with future events
Behavioral training: relaxation, breathing exercises, confrontation of fears Pharmacologic tx: - anxiologics: benzodiazepines (alprazolam, lorazepam, clonazepam), buspirone (5-HT partial agonist) - antidepressants: SSRIs (fluoxetine, paroxetine, sertraline), SNRIs (venlafaxine, duloxetine), TCAs (imipramine, amitriptyline, desipramine), MAOIs (selegiline patch, phenelzine) - others: hydroxyzine, beta-blockers, anti-epileptics/mood stabilizers (valproic acid), antipsychotics (olanzapine, thioridazine) |
|
|
Obsessive Compulsive disorder
|
DSM:
- presence of either obsessions or compulsions (usually both) - person is aware that obsessions/compulsions are unreasonable - obsessions/compulsions cause: marked distress, significant impairment in normal function, are time consuming (require >1hr/d) - context of the obsession/compulsion is not restricted to another disorder (trichotillomania, body dysmorphic disorder, anorexia nervosa, hypochondriasis) - not caused by a substance or medical condition Other Phenomenology: - functional relationship between obsession/compulsions usually present - content can be highly unusual: image of classmate from yearbook, fear of turning into another person - range of insight from complete to poor - cycle: obsessions → anxiety → compulsions → temporary relief Epi: 1mo 1.3%, lifetime 2.5%, male onset peaks 13-15 female 20-24, often comorbid MDD/anxiety/other psych disorder Screening: ask about washing/cleaning, checking, bothersome thoughts, time consuming daily activities, concern about orderliness/symmetr |
|
|
Obsessions
|
= persistent thoughts, impulses, or images that are experienced as intrusive, inappropriate and distressing
- the obsessions are not simply excessive worries about real life problems - the person attempts to ignore or suppress the obsessions or neutralize them with other thought or action - the person recognizes that the obsession are a product of their own mind Most common: contamination, harm, symmetry |
|
|
Compulsions
|
= repetitive behaviors (e.g. handwashing) or mental acts (e.g. praying silently) that the person feels driven to perform in response to an obsession or according to rigid rules
- compulsions are aimed at reducing distress or preventing a dreaded situation: may be either realistic or completely excessive - most common: checking, cleaning/washing, other, repeating, mental rituals, ordering, hoarding/collecting, counting Mental compulsions: special words, images, numbers recreated mentally to reduce anxiety. Ex: special prayers, mental counting/list making/reviewing |
|
|
Obsessive compulsive Personality Disorder
|
Characterized by: preoccupation with details, perfectionism, excessive devotion to work/productivity, overconscientiousness and inflexibility, inability to discard worthless objects, reluctant to delegate, miserly spending style, indecisiveness, rigidity and stubbornness
|
|
|
Psychobiology a/w OCD and tic disorders
|
- problems are a/w the circuit between the orbitofrontal cortex, the striatum, and the thalamus.
- OCD risk is higher in pts w/ FHx of Tourette’s - OCD can be a/w Pediatric Autoimmune Neuropsychiatric Disorder Associated with Strep (PANDAS): group B strep infection causes production of Abs against the basal ganglia (overlaps w/ Sydenham’s chorea) |
|
|
Treatment of OCD
|
CBT:
- exposure in vivo: prolonged confrontation w/ anxiety provoking stimuli (e.g. contact w/ contamination) - imaginal exposure: prolonged imaginal confronation with feared disasters - ritual prevention: blocking of compulsions (leaving kitchen w/o checking stove) - cognitive interventions: correcting erroneous cognitions (anxiety decreases w/o ritualizing) Pharm: (serotonin reuptake inhibitors are only class consistently effective in OCD) - anti-depressants: clomipramine (TCA selective for serotonin transporter), fluvoxamine (SSRI) |
|
|
Tic Disorders
|
Tic = sudden, repetitive, non-rhythmic, involuntary movement or sound involving a discrete group of muscles
Types: Tourette’s ( chronic motor + vocal), Chronic motor tic disorder, Chronic vocal tic disorder, transient tic disorder (duration 1-12mo) Features of tics: premonitory sensation, suppressible, unidirectional, sudden, not rhythmic. |
|
|
Tourette’s Disorder
|
= multiform, frequently changing motor and phonic tics
- onset before age 18 - multiple recurrent, involuntary, rapid, purposeless motor movements affecting multiple muscle groups - one or more vocal tics - duration of more than 1 year Epi: 5-30/10K in children, 0.4%-0.7% of general population, 3:1 M:F, Coprolalia <15% of Tourette’s pts - tics increase as a result of tension or stress and decrease with sleep, relaxation, or when focusing on an absorbing task - syndrome visible by age 7, but early signs may begin by age 5 - a/w visual/motor learning disabilities (stuttering), social-emotional effects Common tics: eye-blinking, neck jerking, shoulder shrugging, facial grimacing Complex tics: tapping, touching (people/things), squatting, retracing steps, deep knee bends, twirling when walking, jumping, smelling, rarely self-injurious actions (hitting, biting onself) |
|
|
Treatment of tic disorders
|
Modification of DA signaling:
- typical antipsychotics (D2 antagonists: haloperidol, pimozide), atypical antipsychotics (D2/5HT antagonists: risperidone, olanzapine) Behavioral Modification: - relaxation techniques, habit reversal Deep Brain Stimulation: - thalamus: centromedian-parafascicular complex - globus pallidus |
|
|
Bipolar disorder
|
= disorder consisting of episodes of depression and mania (or hypomania)
Types: I (mania), II (hypomania), NOS, cyclothymia (2y in adults/1 in adolescents: hypomania + subthreshold depression) Epi: 1% mania/2-5% hypomania, M=F, AoF 19 (rare after 50), depressive episodes > manic, 10-15% rapid cycling, 60% substance abuse comorbidity, >50% anxiety comorbidity, 19% suicide rate (↓ if never hospitalized) 50% attempt Symptoms: - Manic (>1w, severe impairment)/hypomanic episodes (4d, w/o severe impairment): (DIG FAST) Distractability, Insomnia, Grandiosity, Fast thoughts (racing)/flight of ideas, Activities (increased, goal-driven), Speech (pressured), Thoughtless behaviors (impulse, reckless) - Depressive Episodes (>2wks): (SIGECAPS) Sleep changes (+/-), loss of Interest, Guilty feelings/worthlessness, Energy low, Concentration (poor), Appetite changes, psychomotor changes (usually retardation), suicidal ideation Mixed episodes: mood episode including symptoms of both depression and mania occurring simultaneously Dx: - if present w/ depressive episode: r/o other medical/psych illness, search for hx of mania/hypomania - if presents w/ mania/hypomania: r/o other medical/psych causes |
|
|
Mania
|
= a period of elevate or euphoric or irritable mood lasing >1 week (or less if hospitalized)
- must cause severe impairment Symptoms (3 or 4 if irritable): characterized by accelerated cognitive and behavioral activity with occur simultaneously with the mood change - DIG FAST: Distractability, Insomnia, Grandiosity, Fast thoughts (racing)/flight of ideas, Activities (increased, goal-driven), Speech (pressured), Thoughtless behaviors (impulse, reckless) |
|
|
Hypomania
|
= a period of elevated, euphoric or irritable mood lasting at least 4 days
- must not cause severe impairment Symptoms (3 or 4 if irritable): characterized by accelerated cognitive and behavioral activity with occur simultaneously with the mood change - DIG FAST: Distractability, Insomnia, Grandiosity, Fast thoughts (racing)/flight of ideas, Activities (increased, goal-driven), Speech (pressured), Thoughtless behaviors (impulse, reckless |
|
|
Pathophysiologic theories of Bipolar Disorder
|
Genetics: susceptibility inherited in non-Mendelian fashion, additive effect of multiple small genes
Environment: specific environment, random events, epigenetics, intrauterine/perinatal events Environment and genetics interactions: individuals w/ family members w/ BP are 8-10x more likely to develop it. 7% risk w/ first degree, 40-75% for monozygotic and 6-11% for dizygotic twins Neurobiology: cortisol changes in HPA axis, ↓ventromedial prefrontal cortex activity and ↑amygdala/ventral striatum activity, ↓grey and white matter mass Etiologic theories: - stress sensitivity/emotional dysregulation mediated by brain derived neurotrophic factors, oxidative stress, HPA axis disruption - dysregulated circadian cycles (shortened or lengthened): when cycles stressed, pts are more irritable/manic the next day. Possible correlation btwn various gene polymorphisms involved in circadian rhythms and BPD - Kindling: over stimulation of neurons sensitize them to the point where they start spontaneously firing. Though to be mechanism behind rapid cycling, involving neurons in deep limbic areas. Kindling is more clinically demonstrated by the fact that over time episodes become more frequent, well periods shorter, psychosocial triggers decrease, rapid cycling increases, and treatment responsiveness decreases |
|
|
Pharm treatment for bipolar disorder
|
First line: mood stabilizer monotherapy
- Lithium, valproic acid (Depakote), carbamazepine (tegretol), lamotrigine (lamictal) - optimize dose while minimizing side effects. Additional meds may be needed to treat side effects: propranolol for lithium tremor, zinc or selenium for valproate alopecia Second line: - add second mood stabilizer or antipsychotic if patient is only partially responsive to initial treatment - use atypical antipsyhotic if patient has psychotic symptoms or particularly severe mania presentation: olanzapine, quetiapine, aripiprazole Refractory: - reassess diagnosis, drug interactions, therapeutic levels/dose, lack of medication adherence, concomitant substance use - consider adding another mood stabilizer (not previously used) or ECT (may cause short term memory loss) General: avoid antidepressants, can precipitate rapid cycling. Exception if patient is severely depressed and suicidal (use w/ mood stabilizers, taper off quickly) |
|
|
Psychological treatments for bipolar disorder
|
Interpersonal and social rhythm therapy:
- based on circadian rhythms, lifestyle issues, and interpersonal stressors affecting pt’s mood - identify relationships btwn social routines and mood episodes, self-monitoring of timing of daily activities and mood episodes - pt encourage to keep stable social rhythms, anticipate disruptions, adaptively deal w/ social stressors. Use role-play to prevent future interpersonal problems/social disruption Family focused therapy: - for families w/ high conflict/expression of negative emotions and low problem solving abilities (pts have more episodes in these families) - family taught about BD, medication adherence, triggers. Together develop relapse plan, ways to de-escalate stressful verbal exchanges - communication enhancement exercise (pt +family) Cognitive behavior therapy: - interrupts cycle between cognition, feelings and behavior. - involves psychoeducation (BD course, meds, stress management), cognitive restructuring (challenge pts distortions/negative thoughts), live events scheduling (alleviate inactivity, reduce overstimulation) - equips pt w/ better coping skills, strategies for early mood detection and control, treatment for comorbidities Integrated Group Therapy (Dual Diagnosis Treatment) - pts engage in weekly group therapy for 20wks - encourage med adherence, avoidance of other substances, identification of triggers, address denial, recovery vs relapse thinking |
|
|
Diagnostic features of PTSD
|
Signs/symptoms: ERASE for >1mo, causing significant social or occupational impairment
- Exposure to trauma evoking strong fear, helplessness, horror: violent personal assault, military combat, natural disasters, severe motor vehicle accident. Worse if human mediated (assault/rape) - Re-experience of traumatic events through nightmares and flashbacks, causing psychological stress (resembles a panic attack): ↑HR, chest discomfort, chills, hot flashes, sweating, nausea/abd distress, lightheadedness, shortness of breath, choking sensation, cortical interpretation and expressions of fear - Avoidance of stimuli: thoughts, feelings, people, activities - Sympathetic Excitation (state of hyper-arousal): decreased sleep, irritability/anger, difficulty concentrating, hypervigilence Epidemiology: - lifetime prevalence 8-13%, higher among those w/ significant lifetime exposures (40-75%). Many prisoners have PTSD - usually begins as acute stress disorder (presents 3-6mo after trauma). Signs may disappear if pt is engaged and busy, but relapse in less active mind - risks for suicide: refugee, combat exposure, war-related internal displacement, early childhood trauma, family hx of disease/genetics (FKBP5) or antisocial behavior, sudden death of loved one, poorer coping strategies, pre-existing psych disorders, reduced hippocampus size |
|
|
Neural centers controlling fear response
|
Lateral hypothalamus: increased HR/BP
Dorsal vagal nerve: bradycardia, ulcers Parabrachial nerve: panting, respiratory distress Basal forebrain: vigilance, increased attention Reticular Pontis caudalis: increased startle response Central gray area: freezing, social interaction Paraventricular nerve: corticosteroid release |
|
|
Model of Fear disorders
|
- Pre-existing sensitivity: genetic (30-40%), environmental (early c
childhood trauma) - Learning of fear: traumatic event - Consolidation of fear: memory/storage of the fear in which the emotional memory regulated by the amygdala outweighs the explicit memory regulated by hippocampus and cortex. May occur hours to days after event, depends on numerous features: sympathetic arousal, HPA axis activation, social support, substance use, sleep - Expression of fear: clinical manifestation of disorder. May be generalized (initial discrete fear broadens to many places/situations), or discrete to a specific cue. - Sensitization or extinction: fear response may increase or decrease with time and exposure |
|
|
Treatment of PTSD
|
Pharm: 30-40% not responsive, others only partially responsive
- SSRIs (paroxetine, sertraline): increase serotonin release from the raphe nucleus to act on the amygdala - SNRIs/NRIs: decrease NE and locus coeruleus firing - Other antidepressants and benzodiazepines Alternative therapy: - ECT: hippocampus, dorsal/ventral prefrontal cortices - psychotherapy (modification of prefrontal cortices): CBT (exposure and anxiety management), eye movement desensitization and reprocessing when recalling - D-cycloserine: improves glutamate neurotransmitter activity at NMDA receptor, at the time of emotional learning can help improve extinction |
|
|
Factors that aid mucosal attachment, damage and invasion in bacterial meningitis
|
Secretion of enzymes/toxins:
- S. pneumo secretes neuramidase (NanA): ↓viscosity of local mucin in upper respiratory tract - IgA protease secretion: cleaves mucosal IgA, aids immune evasion - N. meningitides contains LPS or endotoxin in outer membrane: w/ other cellular enzymes damages mucosal cells Environmental Factors: - Arid conditions (Sub-Saharan Africa): dryness reduces mucosal integrity/clearance aiding colonization - smoking, crowding, precedent viral infections (influenza) increase risk |
|
|
Bacterial Meningitis Pathogens
|
- most are encapsulated (usually polysaccharide, gram+/-), extracellular pathogens that colonize the nasopharyngeal or GI mucosal surfaces, then invade the blood stream and penetrate the BBB
- less often, bacteria spread direct directly from contiguous site: middle ear, maxillary sinuses, neurosurgical site or trauma Nasopharyngeal: - Neisseria meningitides (meningococcus), Streptococcus pneumoniae (pneumococcus), Haemophilus influenzae GI: - E. coli K1, Group B strep (S. agalactiae): most common in neonates, tx w/ ampicillin plus either cefotaxime or gentamicin Alternative: - intracellular: Listeria moncytogenes |
|
|
Factors that aid blood stream survival in bacterial meningitis
|
Encapsulation: most pathogens are encapsulated, extracellular
- capsule (usually polysaccharide), increases resistance to phagocytosis (still sensitive to complement opsonization) Evasion of alternative complement system: - N. Meningitidis: Factor H binding protein (surface lipoprotein) binds factor H to inhibit alt complement pathway - S. pneumoniae: C3b binds inefficiently to Factor B on bacterial capsule surface - H. Influenzae: PRP capsule blocks acceptance of C3b on surface |
|
|
Inflammation and end organ damage in bacterial meningitis
|
- invasion of bacteria into the BBB stimulates TLR on astrocytes and microglia (phagocytes) → cytokine secretion, inflammation. TLR4 is most important for recognition of LPS (esp. N. meningitides)
- S. pneumonae lyses itself once it enters the CNS, but still elicits immune response from toxin release - Local immunodeficiency in the CNS space allows for uncontrolled bacterial replication: no lymphatics, minimum complement in CSF, low immunoglobin levels - Much of the damage of meningitis is due to inflammation in a contained space: increased vascular permeability (leukocyte invasion), edema, vasculitis (ischemia), release of toxic elements all damage neurons - arachnoid space is classic location of meningitis |
|
|
Time course of bacterial meningitis
|
0Hrs: initial colonization of CNS space
- bacteria and bacterial products accumulate in CSF - no symptoms 4hrs: bacterial replication, initial immune response - release of inflammatory mediators and cytokines - fever 8-24h: full immune response - BBB disruption, cerebral edema, transendothelial migration of leukocytes, release of proinflammatory and toxic mediators, impaired cerebral blood flow, elevated ICP, vasculitis - elevated CSF protein, meningmus/neck stiffness, elevated CSF leukocytosis, possible systemic complications, altered mental status, focal symptoms, seizures 24-48h: secondary effects - neuronal damage - focal symptoms, hearing loss, paralysis, cognitive impairment, death |
|
|
Clinical presentation of bacterial meningitis
|
Symptom triad (2/3 of pts have all 3): fever, nuchal rigidity (pain on neck flexion), altered mental status
Other symptoms: headache, seizures, cranial nerve abnormalities (esp. CN 3, 4, 6,7), sepsis Signs (<50%): Kernig’s (pain upon hip flexion and knee extension: causes neck flexion), Brudzinski’s (hip flexion elicited upon flexion of neck: avoiding flexion) Diagnosis: - head CT (r/o risks for LP): AMS, focal neurologic findings, Hx of CNS lesion (mass/stroke), seizure w/in 1wk of presentation, immunocompromised, papilledema - LP (L3-L5): ↑WBC (>>100, neutrophil predominant, viral would be lymph predominant), ↑protein (100-500), ↓glucose (<50), positive cultures/gram stain (neg viral PCR) - counterindications of LP: ↑ICP, bleeding risk, paraspinal infection near site, respiratory distress w/ uncontrolled airway |
|
|
Streptococcus pneumonia meningitis
|
Characteristics: gram positive diplococcus (lancet shaped), polysaccharide capsule, α-hemolytic
Epi: most common cause of bacterial meningitis in the US (esp in pts >6mo), mortality 20% Risk factors: any impairment in PMN function, complement action, or antibody production: asplenia, HIV, primary immunodeficiency (esp. agammaglobulinemia), cirrhosis of liver, end stage renal dz, DM, hematologic malignancies, CSF leak Pathogenesis: - direct spread from contiguous structures (sinuses, middle ear) or bacteremia - capsule inhibits C3b opsonization Clinical presentation: classic triad Treatment: - IV abx: PCN, ceftriaxone/cefotaxime, or vacomycin if resistant to other abx - IV dexamethasone: 1st four days, adults only Prevention: pneumococcal polysaccharide vaccine indicated for susceptible patients |
|
|
Neisseria meningitides
|
Characteristics: gram negative diplococcus (coffee bean shaped), polysaccharide capsule
Pathogenesis: - attaches to upper airway epithelium -LPS/endotoxins, factor H binding protein (inhibits alt. complement ptwy) Risk factors: crowded conditions (military, college), travel to endemic areas (sub-Saharan Africa) Clinical presentation: focal neurologic signs, DIC, purpura fulminans, adrenal infarction (Waterhouse-Friderichsen syndrome) Poor prognosticators: age >60, lack of meningitis, septic shock, thrombocytopenia, purpura. Mortality 8-10% Treatment: - IV abx: PCN or ceftriaxone/cefotaxime - no dexamethasone Prevention: vaccination (not available for serogroup B) for people with risk factors |
|
|
Haemophilus Influenza meningitis
|
Characteristics: gram negative rod, serotype B is encapsulated
Clinical presentation: otitis media, sinusitis Treatment: ceftriaxone/cefotaxime, or ampicillin Prevention: rifampin for 4d if household exposure |
|
|
Listeria monocytogenes
|
Characteristics: gram positive rod, facultative intracellular, flagellated (tumbling motility)
Risk factors: dairy/deli meat ingestion (infects GI mucosa), iron overload (hemochromatosis, transfusions), AIDS, immunosuppression, malignancies, pregnancy, extremes of age Clinical presentation: nuchal rigidity less common, tends to affect the brainstem→ rhomboencephalitis (causes cranial nerve and cerebellar defects) Treatment: IV ampicillin or PCN G (cannot use cephalosporins) +/- gentamicin (aminoglycoside) |
|
|
Brain abscess
|
Clinical presentation: headache, stiff neck, fever, neurological deficits, seizures. Fever/systemic signs often absent
Regions infected: frontotemporal > parietal > cerebellum > occipital Pathogensis: - direct seeding from contiguous site: subacute/chronic otitis media, mastoiditis, frontal or ethmoid sinusitis (to frontal lobes), dental infection (to frontal lobes) - hematogenous: bacteremia, endocarditis, congenital heart dz, chronic pulmonary infections, pulmonary AV malformations - cryptogenic Organisms: typically polymicrobial - bacterial: streptococcus, anaerobes (bacteroides, prevotella, propionibacterium), S. aureus (head trauma, surgery), klebsiella pneumonia, nocardia (acid fast stain) - non-bacterial: toxoplasmosis (immunocompromised), fungal (aspergillosis, zygomycosis) Diagnosis: - MRI w/ contrast: early (hypointense area), late/breakdown of BBB (ring enhancing lesion) - LP usually not helpful Tx: - bacterial: drainage/excision of lesions >2.5cm, empiric IV abx: ceftriaxone/cefotaxime + metronidazole + vancomycin. If dental: PCN + metronidazole. If post-surgical: vancomycin + ceftazidime. If endocarditis: vancomycin + gentamicin. Follow up 4-6 for resolution - non-bacterial: If toxo: pyrimethamine + either sulfadiazine or clindamycin/leucovorin. If aspergillosis: voriconazole. If zygomycosis: amphotericin B |
|
|
Types of CNS inflammation
|
Meningitis: inflammatory process affecting the meninges, characterized by CSF pleocytosis, typically infectious can be non-infectious
Aseptic meningitis: acute meningitis syndrome w/ negative CSF bacterial culture (commonly used interchangeably w/ viral meningitis) Encephalitis: inflammatory process of the brain parenchyma w/ neurologic dysfunction, typically viral infection but can have non-viral infections or non-infectious causes Myelitis: inflammatory process of the spinal cord Meningoencephalitis: CNS infection /w clinical features of meningitis and parenchymal involvement |
|
|
Non-polio enteroviruses
|
Family: picornaviridae
Syndromes: exanthema/enanthems, myopericarditis, pleurodynia, ocular infection, fulminant neonatal infection, CNS infections (mening, enceph) CSF infection: most common cause of viral meningitis, also cause encephalitis. Both are self-limited, occur post viremia. Can cause severe illness in neonates, chronic meningoencephalitis in pts w/ B-lymphocyte defects (X-linked agammaglobulinemia). Rarely causes flaccid motor paralysis. Dx: CSF enteroviral PCR. Rx: supportive Transmission: fecal-oral (direct contact w/ feces or infected food, water, fomites). Also respiratory secretions: esp children <4y (59%) in summer/fall (common cold pathogen). |
|
|
Lymphocytic choriomeningitis virus (LCMV)
|
Risk factors: exposure to rodent droppings
Syndromes: mostly asymptomatic, but flu-like illness w/ viral meningitis or encephalitis can occur, usually self-limiting. During pregnancy can cause congenital defects of miscarriage Dx: paired serologies Rx: supportive |
|
|
HSV encephalitis
|
Epi: most common cause of sporadic viral encephalitis (20-30% of US cases). Not common (2.3/mil/yr). 90% of adults are seropositive w/ HSV1 by 4th decade.
Transmission: primary CNS infection via CN5 or olfactory tract or via latent reactivation of infection. Neonatal encephalitis caused by HSV-2 Presentation: fever + focal neurolical signs (altered mental status, CN defects, hemiparesis, aphasia, ataxia, focal seizures, hypomania, Kluver-Bucy Syndrome) Pathology: focal temporal lobe hemorrhagic necrosis Dx: MRI, LP:↑WBC (lymphocyte predominant), RBC (hemorrhagic necrosis), protein, HSV PCR Tx: acyclovir Prognosis: 70% fatality if untreated, 20-30% fatality if treated. Survivors w/ high rates of neurologic deficits |
|
|
HSV aseptic meningitis
|
Syndrome: usually a/w primary HSV-2 genital infections. Generally benign and self-limited. Rarely recurrent meningitis (Mollaret’s: recurrent inflammation of the meninges)
Symptoms: resolving headache/stiff neck, generally mild, tx not sought Dx: LP (CSF w/ HSV PCR) Tx: acyclovir (not clear if changes natural history) |
|
|
JC virus encephalopathy
|
Family: polyomaviridae
Epi: seroprevalence 50-90% of adults Risk factors: immunosuppression Syndrome: progressive multifocal leukoencephalopathy Dx: CSF JC PCR Tx: immune reconstitution Prognosis: poor |
|
|
CMV encephalopathy
|
Family: Herpesviridae
Epi: seroprevalence 40-100% of adults Risk Factors: immunosuppression Syndrome: progressive dementia (encephalitis), acute change in mental status (ventriculoencephalitis), myelitis, polyradiculitis Dx: CSF CMV PCR Rx: ganciclovir, foscarnet Prognosis: poor |
|
|
HIV encephalitis
|
Family: retroviridae
Epi: 20-30% of HIV cases (pre-HAART era) Risk factors: AIDS Syndrome: HIV-associated dementia Dx: clinical Tx: HAART Prognosis: can improve with therapy |
|
|
Measles virus
|
Family: paramyxoviridae
Syndromes: acute encephalitis, acute disseminated encephalomyelitis (ADEM), subacute sclerosing panencephalitis (SSPE) Dx: IgG/IgM Tx: none |
|
|
West Nile Virus
|
Family: Flavivirus
Vector: mosquito (culex species) Syndrome: - asymptomatic (80%) - mild (20%)/non-specific flu-like sx: fever, headache, body aches, N/V, truncal rash - severe (1/150 cases): meningitis (25%), encephalitis (60-75%), poliomyelitis. Elderly at highest risk for neuroinvasive disease and death Dx: IgM in serum or CSF Tx: none |
|
|
Rabies encephalitis
|
- Highest case mortality for any encephalitis
Transmission: inoculated through skin, migrates to brain via motor/sensory neurons (50-100mm/day, can take a while depending on location of bite) Presentation: - prodomic symptoms: parathesias at wound, fever, chills, N/V, headache - Encephalitis (“furious”): hydrophobia, hyperactivity, coma, autonomic instability, dysarthria, agitation - Paralytic (“dumb”): ascending paralysis Dx: PCR of CSF/saliva/serum., skin biopsy, serum/CSF antibodies Rx: palliative Pathology: negri bodies (look like bullets) |
|
|
HIV neuropathology
|
- HIV is neurovirulent not neurotropic: does not directly infect neuronal cells (free virus not found in neural tissue). Can infect some neural cells (acutely: meningeal macrophages, chronically perivascular macrophages, microglia, some glial cells)
- pathology results from immune activation of astrocytes, microglia, macrophages causing cytokine/chemokine release, inflammation, neural injury. 3 Main syndromes: peripheral neuropathy, myelopathy, HIV-associated Neurocognitive disorder (HAND) |
|
|
HIV peripheral neuropathy
|
= most common neurological complication of HIV. Generally a/w advanced immunodeficiency
Types: HIV-associated peripheral neuropathy, antiretroviral toxic neuropathy (caused by didanosine/ddI, dideoxycytidine/ddC, stavudine d4T. 18% of pts asymptomatic, 4% symptomatic) Risk factors: - host related: Age, karnosfsky score (performance status), substance abuse (EtOH, IVDA), medical comorbidities (DM, IGT, ESRD), nutritional status, genetics - Disease related: exposure to d-drugs (ddC, ddI, D4T), low CD4 count, high viral load, AIDS-related complications (dMAC, vaculolar myelopathy, AIDS dementia complex) Presentation: neuropathic symptoms ( pain, distal parasthesias, numbness, muscle cramps, impaired sensation, relfexes), impairment of QoL, unemployment, inability to perform ADLs Dx: neurologic exam. Typically proximal extremities, band around costal margin/sternum - small fiber neuropathy: sensory symptoms (mostly pain) +/- loss of temp sensation. Normal reflexes - large fiber neuropathy: sensory symptoms + signs (loss of light touch, temp, vibration, and/or proprioception). Reduced/absent reflexes. Work-up: - r/o B12, thyroid, DM. Assess for EtOH/IVDA - consider nerve conduction study, skin biopsy for confirmation - tx w/ analgesic |
|
|
Dx of neuropathic pain
|
Combination of positive and negative sensory symptoms
- parathesia: any abnormal sensation, evoked or spontaneous - dysethesia: unpleasant abnormal sensation, evoked or spontaneous - allodynia: evoked pain from stimulus not normally painful - hyperalgesia: exaggerated response to painful stimuli - hypalgesia: reduced response to painful stimulus |
|
|
HIV Myelopathy
|
Epi: 5-10% of AIDS patients.
Presentation: slowly progressive - painless leg weakness and spasticity, + Babinski, posterior column signs. Paresthesisas often asymptomatic, lacks discrete sensory loss level (similar to B12) - Urinary symptoms: urgency, frequency, erectile dysfunction Dx (exclusion) - MRI: generally normal, may so mild atrophy of thoracic cord - neuroimaging (CT/MRI): r/o mass lesion, myelitis - LP: assess inflammation - serum HTLV, B12, syphilis serology Tx: antiretrovirals, antispasticity meds, oxybutynin for neurogenic bladder, PT/OT |
|
|
HIV associated Neurocognitive Disorder (HAND)
|
Symptoms:
- cognitive: decreased concentration, forgetfulness, mental slowing, loss of mental flexibility. Subcortical: no aphasia, recall<recogn, impaired visuospatial, frontal/exec norm, slow early mental speed. Cortical: early aphasia, recall=recogn, impaired visuospacial, reduced frontal/exec, slow late mental speed. - behavioral: apathy, depression, personality change, withdrawal, irritability - motor: gait incoordination, clumsiness, tremor, increased reflexes Risk factors: - host: age, weight, gender, high viral set point, low nadir CD4 count, drug abuse, anemia, thrombocytopenia, insulin resistance, genetics (ApoE4, MCP-1, CCR-2, TNF-R) - viral: some viral strains more pathogenic, co-pathogens present (ex. HCV) Dx: exclusion - r/o: B12, hypothyroidism, major depression, syphilis, chronic meningitis (LP), CNS lesions (MRI w/ contrast), substance abuse, HCV co-infection Tx: - HAART induction: 1/3 of patients may improve. No other disease modifying therapy available |
|
|
AIDS opportunistic infections
|
AIDS = CD4 <200 or opportunistic infection:
Fungal: candidiasis (esophageal, LRT), coccidiodomycosis, Cryptococcus, histoplasmosis, pneumocystis carinii Bacterial: salmonellosis (recurrent), pneumonia (recurrent), tuberculosis (recurrent), disseminated MOTT Malignancy: cervical cancer (invasive), Burkitt’s lymphoma, CNS lymphoma, Kaposi’s Sarcoma Parasitic: cryptosporidiosis, Isosporiasis, CNS toxoplasmosis Viral: CMV, progressive multifocal leukoencephalopathy (JC virus), HIV encephalopathy Lesions types: - focal vs. diffuse (dx w/ head CT/MRI) - focal w/ mass effect: toxo, primary CNS lymphoma (get serum toxo IgG) - focal w/o mass effect: PML, stroke, HSV/VZV encephalitis, CMV encephalitis |
|
|
Toxoplamosis gondi encephalitis
|
Epi: 15% of adults latently infected w/ protozoa.
Transmission: consuming undercooked meat w/ tissues cysts, ingestion of oocyts (shed in cat feces which have sporulated). AIDS defining illness: virtually all pts have CD4<200, most common etiology for focal brain masses in AIDS Presentation: (mass effect) subacute onset of headache, confusion, focal weakness, seizure Dx: most made clinically, definitive only w/ biopsy - Serum: T. gondi abs - CT/MRI: 1+ mass lesions in the brain - LP: r/o other causes of lesion (EBV PCR) Tx: pyrimethamine + sulfadiazine + folinic acid for 3-6wks. Secondary prophylaxis w/ pyrimethamine+sulfadiazine until CD4>200 for 6mo Prognosis: 85% improve in 7-10d (confirms dx) |
|
|
Primary CNS lymphoma
|
= diffuse, high grade B cell lymphoma a/w EBV
Epi: most commonly in immunocompromised (HIV, transplant) patients and the elderly (70+) Histo: grow concentrically around vessels. Stain w/ CD20 (identifies B cells) Dx: (r/o toxo, PML) - Serum: negative toxo IgG - LP: positive EBV PCR - SPECT-Thallium 201 imaging: appears as a hot lesion (has active membrane pumps that uptake marker. Toxo/JC would appear as cold/dead). Prognosis: 5mo on HAART, 2mo w/o |
|
|
Progressive Multifocal Leukoencephalopathy (PML)
|
= infection of JC virus (polyomavirus). Only known cause of viral demyelinating disease
Epi: 3-5% of HIV patients, generally CD4<100. 85% of cases a/w HIV, 80% of adults seropositive for JC (likely latent in kidneys, spleen, or marrow) Presentation: slowly progressive focal neurological deficits, occasionally w/ seizures Pathogenesis: demyelinating disease of CNS due to infection of the oligodendroglia. Dx: - LP: non-inflammatory, +JCV PCR (80% sensitive, 95% specific) Prognosis: w/ HAART survival to 6y for 66% (vs. 1.5-4mo), 44% of survivors showing neurologic improvement |
|
|
Cryptococcal Meningitis
|
= opportunistic infection of Cryptococcus neoformans
Epi: 5-8% of HIV pts (before HAART) Presentation: (non-specific) headache, fever, malaise, meningismus (25%), photophobia, mental status change (↑ICP) Dx: - Serum: CrAg - LP: ↑opening pressure, ↑WBC (lymphocytic), ↑protein, ↓glucose, CrAg, culture (india ink stain) Tx: amphotericin B + flucytosine for 2 weeks, then fluconazole for 10wks, then half dose fluconazole until CD4>200 for 6mo. Repeat LP to manage ICP |
|
|
CMV disease
|
Epi: most adults seropositive, so disease may be reactivation or new infection. Infection in 40% of HIV pts.
Clinical course: - may be disseminated - end-organ disease (retinitis, colitis, esophagitis): occurs w/ severe immunosuppression: (CD4<50), represents compartmentalized infection, may be preceded by viremia. - neurologic syndromes: microglial nodular encephalitis, ventriculitis, polyradiculomyelopathy, mononeuritis multiplex Dx: - CSF: variable pleocytosis, ↑protein, normal/↓glucose, + CMV PCR Tx: gancyclovir + foscarnet + cidofovir |
|
|
Glioma
|
= most common primary brain tumor
Types: astrocytoma, oligodendroglioma, ependymoma Presentations (depends on site involved): seizures, motor/sensory deficits, changes in consciousness Path: infiltrative so normal elements of brain tissue are seen in background |
|
|
Brain stem gliomas
|
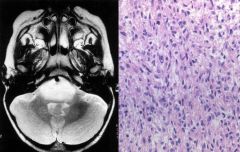
= tumor specific to the pediatric population, arising the posterior fossa
- look like infiltrating astrocytoma, generally grade 2 or 3 but will progress to glioblastoma - tend to be rapidly fatal: growing tumor impinges on the brainstem, lose cranial nerve function as it grows |
|
|
Low grade, circumscribed brain tumors a/w seizures
|
- tend to be localized to the superior tentoral compartment
- generally curable - tend to have both infiltrative and cystic component Types: - glanglioblastoma: glial background w/ dysmorphic mature neurons (large nuclei, prominent nucleoli, , Nissl substance, triangular shape) - dysembryoplastic neuroepithelial tumor (DNT) - pleomorphic xanthoastrocytoma (PXA) |
|
|
Primitive neuroextodermal tumor
|
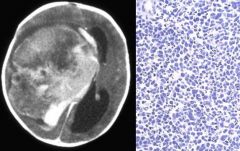
= malignant embryonal tumors found supratentorally
- similary to medulloblastomas (which are only in the cerebellum) Histo: small, round, blue cells |
|
|
Pineal tumor
|
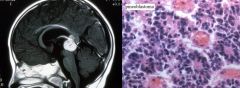
Types:
- pineoblastoma: characterized by Flener-Wintersteiner rosettes, very poor prognosis (high grade, age 1-5) - germinoma: characterized by alkaline phostphatase production or c-Kit expression. More treatable |
|
|
WHO Grading of pediatric tumors
|
Grade I: can be treated by surgical resection
- pilocytic astrocytoma - gangliogioma - subependymal giant cell astrocytoma Grade II: - infiltrating “diffuse” astrocytoma Grade III: - anaplastic astrocytoma Grade IV: - glioblastoma |
|
|
Neurofibromatosis 1
|
Genetics: AD mutation in neurofribonin gene on chr 17. 300+ known mutations leading to alteration of Ras-GAP function
Prevalence: 1/4000 Clinical presentation: - neurofibromatomas in subcutaneous tissue at an early age, café au lait patches - may also have plexiform neurofibromas (pathognomonic for NF1): benign but can transform to malignant peripheral sheath tumors - optic gliomas (second most common NF1 tumor): cause blindness, endocrine abnormalities. Can involve component of the visual system (nerve, chiasm, tract). Histo: low grade, look like pilocytic astrocytomas |
|
|
Neurofibromatosis 2
|
Genetics: AD mutations of NF2 and merlin genes (membrane-cytoskeleton linking protein family, involved in cell motility, proliferation, Rac signaling/invasion)
Prevalence: 1/40,000 (rare than NF1) Clinical: - schwannomas: bilateral acoustic neuromas (pathognomonic) - meningiomas: often multiple - spinal ependymomas - others: meningioangiomatosis, glial hamartomas, cerebral calcifications |
|
|
Tuberous sclerosis
|
Genetics: AD mutations in hamartin and tuberin
Prevalence: 1/5000 Pathogenesis: hamartin/tuberin proteins normally complex to form mTor inhibitor. Absence of mTor inhibition allows for cell proliferation Clinical: “tuberous sclerosis complex” - CNS tumors: cortical tubers (push on brain structures, obstruct ventricles), subependymal giant cell astrocytoma (SEGA. Non-proliferating/invasive. 1-20% of cases), subependymal nodules - Non-CNS tumors: cardial rhabdomyomas, renal angiomyolipoma, rectal polyps, retinal and liver hamartomas - other features: developmental delay, seizures |
|
|
Von Hippel-Lindau Disease
|
Genetics: AD mutation of VHL (chr 3)
Prevalence: 1/36,000 Pathogenesis: normally VHL degrades HIF-1a, but when lost a “hypoxic” response is upregulated: VEGF secretion, glycolysis, tumorigenesis Clinical: - hemangioblastomas: in cerebellum and spinal cord, presenting in 2nd/3rd decades. Highly vascular and polycythemic (produce red cells as part of hypoxic response) Histo: clear cells w/ benign nuclei and vascular structures. - Also: renal cell carcinoma, pancreatic tumors, pheochromocytoma |
|
|
Astrocytomas
|
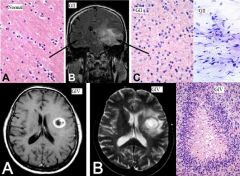
Non-infiltrating:
- I (pilocytic): relatively benign, do not progress to higher grades, generally found in children Infiltrating: - II (diffuse): prognosis 4-8y w/ treatment. Appears as hyperintensity on T2 MRI w/ edema. Histologically brighter/less pink neuropil (diluted from edema), hypercellular, neoplastic astrocytes w/ long, irregular processes, hyperchromatic nuclei - III (anaplastic): prognosis 2-5y w/ treatment. Appears as even more hyperintensity in T2. Histologically higher mitotic activity and cell density - IV (glioblastoma): most common (often de novo), prognosis <1y w/ treatment. Appear as ring around central necrosis w/ periedema on T2. May infiltrate corpus callosum to contralateral hemisphere (butterfly glioma) Histo: pseudopalisades along necrosis, microvascular hyperplasia (VEGF secretion) |
|
|
Oliodendrogliomas
|
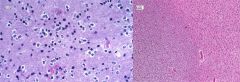
= relatively rare (5-10% of gliomas), slow growing tumor most commonly present in people 40-50y
Presentation: commonly frontal lobe, cannot be distinguished from astrocytoma by symptoms (seizure) or imaging Histo: - grade II: appear as sheets of nuclei that are arounder and smaller, surrounded by halos of clear cytoplasm (fried eggs). Calcifications common. - grade III (anaplastic): further increased cell density, nodules w/ increased mitotic activity, necrosis Prognosis: - deletions of 1p and 19q are favorable (60-80% of patients) and have long-lasting response to chemo/radiation - overall are infiltrative but less aggressive, more responsive than astrocytomas |
|
|
Metastatic CNS neoplasms
|
= most common kind of brain tumors, mostly spread hematogenously from lung, breast, melanoma (lesser kidneys, GI)
Characteristics: -often found in gray-white junctions (cortical-subcortical) b/c arteries line this area - often multifocal (unlike primary) - often sharply demarcated masses (except some melanomas) - histologic features depend on site of origin (ex: pigment in melanoma), often poorly differentiated (by the time it’s metastasized) so use immunohistochemical markers (thyroid transcription factor for lung, ER/PR/GCDFP for breast, S-100/HMB-45/Melan-A/MITF for melanoma) |
|
|
Meningioma
|
= benign, extra-axial tumor (contains no brain tissue, originates outside) of the arachnoid cells attached to the dura
Epi: F>M, 50-60y, second most common primary brain tumor Symptoms: vague, non-localizing. Grades: - I (most common): slow growing (allows brain to accommodate for mass). Usually solitary, consider neurofibromatosis 2 if multiple. Histo: form whorls (nodules) which become calcified as psammoma bodies, fibroblastic w/ elongated cells and collagen deposition, secretory w/ intracytoplasmic droplets. Extent of resection determines prognosis. - II (atypical): have brain invasion. Histo: >4 mitoses/10 hpf, increased cellularity, small cells, high nucleus/cytoplasm, prominent nucleoli, patternless growth, necrosis. Higher recurrence rate, tx w/ resection + radiation - III (anaplastic): sarcomatous growth patterns, >20 mitoses/ 10hpf |
|
|
Schwannomas
|
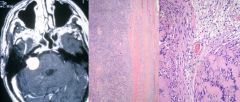
= benign, extra-axial tumor of the Schwann cells (neural crest derived). Third most common primary brain tumor
- schwann cells found at exit of cranial/spinal nerve roots leaving the brain, most commonly cerebello-pontine angle (CN8, internal acoustic meatus) - includes acoustic neuroma: presents w/ tinnitus and hearing loss - occur as part of neurofibromatosis 2, must be considered in Dx Path: - Gross: encapsulated tumors, well delineated from nerves - Micro: elongated nuclei, areas of hypercellularity (Antoni A) and hypocellularity (Antoni B), palisades of nuclei (Verocay bodies) Tx: can be resected w/o sacrificing the nerve (unlike neurofibromas). |
|
|
Neurofibromas
|
= tumor of the peripheral nerve sheath
- can present as cutaneous neurofibromas or solitary neurofibroma growing on a peripheral nerve - can progress to peripheral neural sheath malignant tumors Path: - gross: arise within the nerve, cannot be separated from the nerve (unlike schwannomas) - micro: collagen-rich, hyperchromatic and angulated nuclei |
|
|
Neurocytomas
|
= adult neuronal tumor of the lateral or third ventricles (near foramen Monroe)
- usually low grade lesion, can be resected Histo: stipulated cells w/o visible processes (unlike astrocytoma), nucleis are uniform and evenly spaced. Stained for glial fibrillary protein |
|
|
Spinal cord tumors
|
Locations: epidural, intra-dural extramedullary (btwn dura and cord), intramedullary (intra-axial)
Types: - ependymomas: intra-axial gliomas arising adjacent to ventricular system (esp 4th) and central canal of spinal cord (esp. in adults). Well circumscribed and contrast enhancing. Solid growth pattern (feasible for total resection). Histo: pervascular pseudorosettes (tumors around vessels) - asyrocytomas: intra-axial gliomas w/ diffuse infiltration. Appear as enlargements of the spinal cord, not contrast enhancing. - meningiomas: intramedullary tumors |
|
|
Ependymomas
|
= gliomas that arise next to the ventricular system.
Epi: most commonly form in the brain in first 20y, then in spinal cord in adults. - Most commonly grow along 4th ventricle in the brain or central canal in the spinal cord Path: - gross: well circumscribed, contrast enhancing, solid growth pattern - histo: pseudorosettes (growth around vessels) Tx: resection |
|
|
Pediatric brain tumors
|
- 2/3 in posterior fossa (unlike adults where 2/3 are in cerebral hemispheres)
Classic presentation: vomiting, unsteady gate (wide stance, ataxia), failure to thrive, nystagmus, headache Workup: MRI Types: - pilocytic astrocytoma: most common benign pediatric tumor - medulloblastoma: most common malignant brain tumor, only occurs in the cerebellum - large cell/anaplastic medulloblastoma: aggressive, age <5 - posterior fossa ependymoma: less common, arise within 4th ventricle - anaplastic ependymoma: worst prognosis if age <3 - Atypical teratoid/rhabdoid tumor (ATRT): often a/w genetic predisposition syndrome (dINI1) - brain stem gliomas: specific to pediatric population, rapidly fatal - pineal blastoma - germinoma - others (a/w seizures): gangliomas, dysembryoplastic neuroepithelial tumors, pleomorphic xanthoastrocytomas: generally curable |
|
|
Pilocytic astrocytoma
|
= most common benign tumor in pediatrics
Presentation: vomiting, unsteady gate +/- other posterior fossa symptoms (headache, failure to thrive, nystagmus) Gross: on MRI see cyst w/ contrast enhancing mural nodule Mico: astrocytes w/ long, hair-like processes (bland looking), solid proliferation and formation of microcysts (coalesce to form macrocysts),characteristic Rosthenthal fibers (pink staining deposits of proteinaceous material—not in infiltrative astrocytomas) Tx: surgical resection, good prognosis (recurrence is rare) |
|
|
Medulloblastoma
|
= most common malignant brain tumor in children
- occurs only in the cerebellum - high grade, malignant, w/ nat’l his of dissemination throughout CNS Histo: small, round, blue cells, rosette formation (indicates primitive neuroepithelium), small nodules of better differentiated neural cells (neural islands) expressing synoptophysin and chromogranin |
|
|
Large cell/anaplastic medulloblastoma
|
= aggressive medulloblastoma variant
- typically in children <5yo - present with dissemination at onset (will find cells in CSF) - have c-myc amplification leading to aggressive proliferation Histo: anaplastic cells, apoptotic lakes Prognosis: extremely poor |
|
|
Posterior fossa ependymoma
|
= glioma arising in the 4th ventricle in children (adults is in spine)
- cause vasogenic edema - generally low grade/resectable. MRI: patchy, contrast enhancement in 4th ventricle w/ flair signal in the cerebellum Histo: - low grade: pseudorosettes (radial arrangement of tumor cells around vessels) w/ fibrillar processes extending to central vessel, papillary conformation - high grade/anaplastic: mitotic figures, microvascular proliferation. Poor prognosis if <2y (goal to get to age3-4), long term only 50% survival |
|
|
Atypical teratoid/rhabdoid tumor (ATRT)
|
= malignant tumor of infancy (<2y)
- most commonly found in cerebellum, also supratentorial compartment - clinically aggressive, usually presents with dissemination - differentiation across glial, neuronal, epithelial, and muscular derivations: will stain for a variety of markers - characterized by loss of INI1 expression (monosomy 22q11.2): 1/3 a/w rhabdoid tumor predisposition syndrome (will also have kidney involvement). Should do germline testing Histo: large, rhabdoid cells (and other morphologies), immunophenotypically diverse (GFAP, Syn, actin, vim, cytokeratin) |
|
|
Autism Spectrum Disorder
|
Epi: 1% of US, but increasing
Risk factors: male gender, Caucasian, unknown genes/environmental exposures 4 categories: - autistic disorder: behaviorally defined developmental disorder - asperger’s syndrome: normal intelligence, but social difficulties and repetitive behaviors - pervasive developmental disorder NOS - Rett syndrome: rare X-linked syndrome involving MECP2 gene. Normal development until 2-4y, then skills regress, stereotypical hand wringing and repetitive behaviors arise, and inability to use language develops Domains of functional impairment: language and communication, social interaction (verbal/non-verbal), restricted interests/repetitive behaviors (stimming, flapping, toe walking) |
|
|
Autism disorder
|
= behaviorally defined developmental disorder
auses: most not identified, environment, genetics (chromosomal abnormalities) Characteristics: - functional domain deficits: language and communication, social interaction (verbal/non-verbal), restricted interests/repetitive behaviors, subnormal - other: subnormal intelligence (comorbid), unusual abilities (savants, prodigious capacity), high serotonin levels Comorbidities: intellectual disability (1/3), metabolic, GI (GERD, constipation), neurologic (tics, tuberous sclerosis), psych (anxiety, depression, OCD, psychosis) Dx: - genetic screening for copy number variants (CNVs) w/ genomic hybridization testing - earliest sign is missed milestones of language development: 64% have expressive delay, 18% receptive-expressive delay Therapy: - goals: improving self-care, social, and communication skills - most school systems only provide support until age 22 |
|
|
Allostatic overload
|
Type 1: energy demand exceeds energy supply leading to resource allocation (ex: delayed puberty in response to poor nutrition)
Type 2: energy supply is adequate, but stressors lead to chronic release of stress hormones and inflammatory mediators (ex: early life stress (<17y), abuse/trauma/illness (excess stimulation) or neglect) |
|
|
Neuroanatomy of traumatic stress
|
Cerebral cortex perceives stressor (where traumatic memories are stored long term), hippocampus provides context (more memory), amygdala (conditioned fear) and prefrontal cortex (extinction to fear, moderates amygdala) also involved.
This response stimulates: - the HPA axis: CRH release from hypothalamus → ↑ACTH →↑cortisol - the autonomic nervous system - the immune system (via sympathetic response, also ACTH)→ inflammation (via NE stimulation) |
|
|
Diagnostic feathers of ADHD
|
>6 symptoms of hyperactivity-impulsivity persisting >6mo to a degree that is maladaptive and inconsistent w/ developmental level (for adults >17, only 4sx required)
Inattention: “Often” - fails to give close attention to details/makes careless mistakes, has difficulty sustaining attention, does not seem to listen when spoken to directly, does not follow through on instructions/school work, difficulty organizing tasks, avoids tasks that require sustained mental effort, losses things, easily distracted, forgetful Hyperactivity: “often” - fidgets, is restless, runs about, excessively loud, “on the go,” talks excessively Impulsivity: “often” - blurts out answers, has difficulty waiting turn, interrupts or intrudes |
|
|
Epidemiology of ADHD
|
- 3-7% of school age children, onset usually before age 7
- Adult prevalence of 4.4% (8 million adults) - Continues into adulthood for 50%, can have remission and relapse w/ secondary environmental trigger (substance abuse, social adversity) - Associated conditions: learning disabilities, mood disorders (BP, MDD), anxiety (OCD, generalized), PTSD, substance abuse, behavioral disorders (conduct disorder, ODD), Tic disorders |
|
|
Factors that increase the rate of induction of inhaled anesthesia
|
1. Increased alveolar ventilation/increased respiratory rate
2. decreased solubility of the drug 3. decreased cardiac output Palveoli = Parterial = Pbrain |
|
|
Substance Abuse vs Dependence
|
Abuse = problematic use, involve 1+ symptom for >1y
- failure to fulfill role obligations - repeated use in physically hazardous situations - repeated illegal problems related to use - continued use despite social or occupational problems Dependence = a cluster of cognitive, behavioral, and physiologic symptoms indicating that the individual continues substance use despite significant substance-related problems. >=3 over 1y - tolerance - characteristic withdrawal - larger amounts than intended - persistent efforts to cut down or control use - a great deal of time spent getting substance, taking it, or recovering - important activities given up or reduced - continued use despite psychological or physical problem caused by or exacerbated by use |
|
|
Treatment for substance abuse
|
Initiate stabilization if needed, tailor intervention to the patient, refer to mutual help groups
Rx: (>3mo after abstinence goals met) Nicotine dependence: - nicotine replacement: transdermal, gum, spray - bupropion (Welbutrin): monocyclic antidepressant (DA/NE reuptake inhibitor, AChR antagonist), also treats underlying depression - varenicline (Chantix): partial nicotinic cholinergic receptor agonist. Avoid in depressed patients (suicidal ideation) Alcohol dependence: add thiamine to avoid Wernicke-Korsakoff encephalopathy - benzodiazepines - disulfiram (antabuse): irreversibly inhibits acetaldehyde DH → ↑acetaldehyde → acute discomfort upon consumption (5-10min): headache, flushing, N/V, SOB. Risk of cardiotoxicity - Naltrexone (Revia): antagonizes mu opioid receptor → ↓euphoria, ↓cravings - Acamprosate (Campral): unknown MoA, can be used in patients w/ cirrhosis (renal excretion) Opioid dependence - Methadone: gradual withdrawal, blocks euphoria and cravings - Buprenorphine (suboxone): gradual withdrawal - Naltrexone (Vivitrol): opioid antagonist, does not reduce cravings |
|
|
Epidemiology of substance abuse disorders
|
- SUDs are the leading cause of preventable premature death in the US (>500,000/y, 4:1 tobacco:alcohol)
- Patients are generally equal or more compliant to treatment as patients with other illnesses requiring long-term lifestyle alteration - 17.5% of US is addicted to nicotine, 6.5 to alcohol, 1.7 to illicit drugs - most commonly abused substances: caffeine > alcohol > nicotine > prescription opioids > cannabis - dependence diagnoses: nicotine > alcohol > cannabis > prescription opioids > cocaine/stimulants - nicotine and alcohol cause the most deaths because they are the most accessible |
|
|
Neurotransmission in substance abuse
|
- mesolimbic dopamine system a/w reward/desire, implicated in addiction, schizophrenia, and depression. Substance abuse involves increased dopamine transmission in limbic system
- mesolimbic pathway: begins at ventral tegmental area of the midbrain, connects via nucleus accumbens (reward center) to the limbic system, the amygdala, hippocampus, and medial prefrontal cortex - striatum can lead to quick/impulsive behavior (buying drugs) b/c premotor cortex is not involved - cocaine binds dopamine reuptake transporters, increasing concentration in synaptic cleft and signaling |
|
|
Alcohol use disorders
|
Intoxication: presents with 1+: slurred speech, incoordination, unsteady gait, nystagmus, impaired attention or memory, stupor or coma
Withdrawal (0-72h): occurs after cessation/reduction of use. 2+: autonomic hyperactivity (↑HR/BP/temp), ↑hand tremor, insomnia, N/V, transient visual/tactile/auditory hallucinations, psychomotor agitation, anxiety, grand mal seizure Alcohol withdrawl delirium (5% of untreated withdrawal cases can progress in 3-10d): delirium (confusion, agitation, disorientation), hallucinations, paranoia, autonomic hyperactivity. A/w 5-15% mortality (esp. in pts w/ cardiac arrhythmias or other underlying disease) |
|
|
Positive symptoms of schizophrenia
|
Hallucinations: auditory>visual, tactile>olfactory, gustatory. Gradiation of awareness and severity. When meds often become more internalized than external
Delusions (paranoid, bizarre): fixed false belief such as paranoia about an unlikely event. Commonly involve: religion, politics, media, aliens, thought control, solipsism, veiled messages, grandiosity, capgrass syndrome (thinking people around you are imposters) Formal thought disorder: derailment (discontinuity in flow), loose association (lots of derailments), tangentiality, circumstantiality (delay getting to point), neologism (made up words), word salad, poverty of content, incoherence Disorganized behavior: outgrowth of delusion, hallucinations, neglect. Stereotypic, bizarre behavior: actions w/o purpose that are strange and often repetitive. Can be confused w/ tardive dyskinesia. Extremes are catatonia and catatonic excitement. Ex: fascination w/ reflection, combing hair, developing frozen pout |
|
|
Negative symptoms of schizophrenia
|
Poverty of speech
Poverty of Content of Speech Anhedonia (not depression, more like apathy) Amotivation and social withdrawal Blunted or restricted affect: inappropriate emotional lability, appropriateness (giggling at funeral), range and intensity of emotions. Often conveyed by prosody of voice Primary negative symptoms: independent of positive symptoms, present all the time. No great treatment Secondary negative symptoms: due to positive symptoms, improve with control of positive symptoms |
|
|
Predictors of disease course in schizophrenia
|
More malignant course a/w:
- insidious onset - non-paranoid type - younger age of onset (childhood onset is worst) - poor premorbid function socially, educationally, sexually occupationally - worse negative symptoms - less depressive symptoms - “schneiderian type” : formal thought disorder, first rank symptoms Large ventricles/cortical atrophy - Substance abuse - medication non-compliance |
|
|
Diagnostic criteria of schizophrenia
|
A. 2+ characteristic symptoms: delusions, hallucinations, disorganized speech, grossly disorganized or catatonic behavior, negative symptoms (flat affect, alogia, avolition). If delusions are bizarre or voices keep running commentary or talk to eachother about the patient (3rd person) it is sufficient w/o other symptoms
B: Social/occupational dysfunction: for significant portion of illness, function is markedly below pre-morbid function in 1+ area: work, interpersonal relations, self-care C. Duration of illness: 6mo of continuous symptoms, 1mo of criteria A symptoms D. exclude schizoaffective disorder or mood disorder E. exclude substance abuse related or medical etiology F. exclude pervasive developmental disorder |
|
|
Subtypes of schizophrenia
|
Paranoid type: paranoid delusions, generally better prognosis
Catatonic type: marked psychomotor disturbance (catatonic stupor, negativism, rigidity, excitement, posturing) Disorganized type (hebephrenic): disorganized thought/behavior (giggling at one’s reflection) Undifferentiated type: doesn’t meet other subtype criteria Residual type: no prominent positive symptoms now, + residual or negative symptoms |
|
|
Brief reactive psychosis
|
Sudden onset psychosis lasting >1d but <1mo
Often w/ stressful precipitant: post-partum Mean age: 25-30y Dx: - requires full return to normal function - must have 1+: delusions, hallucinations, disorganized speech, disorganized or catatonic behavior, affect is intense NOT blunted |
|
|
Schizoaffective disorder
|
= a mix of schizophrenia and major mood disorder
- patients may shift between this dx and schizophrenia over lifetime - pts at increased risk of suicide - functional impairment not required but may be present Criteria: - period of concurrent schizophrenia (Criterion A >1mo) + major mood episode (manic >1wk, depressed >2wks, mixed) - delusions or hallucinations w/o mood symptoms for >2wks - symptoms of mood disorder are substantial part of the disease course - Drug/EtOH related or medical cause excluded |
|
|
Delusional disorder
|
Functionally generally better than schizophrenia (affect and thought preserved, can sometimes function for decades w/o treatment). Less reliably responsive to antipsychotics (delusions often intractable)
Criteria: - non-bizzare (plausible) delusions >1mo. Discrete and encapsulated - never met schizophrenia criterion A - functioning but not grossly impaired, behavior not bizarre - duration of mood episodes relatively short compared to delusional periods - Drug/EToH related or medical cause excluded Types: erotomanic, grandiose, jealous (best prognosis), persecutory (worst prognosis), somatic, mixed |
|
|
Schizophreniform disorder
|
= disorder in which the symptoms of schizophrenia are present for a significant portion of time w/in a one month period, but signs of disruption are not present for the full 6mo required for diagnosis of schizophrenia
|
|
|
Risk factors for schizophrenia
|
Genetic:
- inherited vulnerability to schizophrenia and to psychosis in general: lower stress threshold - possible inheritance patterns (polygenetic): copy number variants, innumberable rare variants, epigenetics (50% discordance in monozygotic twins) - advanced paternal age Biological stresses: - birth and pregnancy complications: 2nd trimester influenza, premature rupture of membranes/birth, resuscitation or incubator, gross physical abnormality - post-natal causes: toxins, starvation, infections Psychosocial stressors: contribute to relapse not initial eitiology - individual stress: objective (natural disaster, financial loss, sexual/physical abuse, trauma), subjective (idiosyncrasies, pet peeves, barriers to avocations), psychiatric illness (leads to spiral of worsening illness/social function) - sociocultural stress: affects all social classes but pts tend to drift down as dz becomes debilitating, equal prevalence in 1st and 3rd world countries, although availability of support affects course of disease |
|
|
Prodomal syndrome for psychosis
|
Defined by:
1. attenuated positive symptoms that are too brief for clinical diagnosis: unusual thoughts/delusional ideas, suspiciousness, grandiosity, perceptual abnormalities, conceptual disorganization 2. Brief, limited, or intermittent psychotic symptoms: still self-awareness and questioning of the psychotic events 3. 30% reduction in functioning on social occupational, and psychological scales in the past year, plus genetic risk Common presentation: - mild impairment at puberty, prodromal symptoms in adolescence, onset of psychosis as young adult - social and mood symptoms that later manifest as attenuated positive symptoms - often a/w mild impairments in motor activity, social adjustment, cognition - 30% will progress to frank psychosis (will have greater decrease in gray matter in 12mo), 70% recover/do not progress Preventive intervention: education about current symptoms and risk of substance abuse, school support, vocational rehabilitation, treatment of manic/depressive symptoms, neuroprotection w/ omega-3 FA’s, family group treatment |
|
|
Personality disorder
|
DSM Criteria:
- an enduring pattern of inner experience of behavior that deviates markedly from the expectations of the individuals culture. Pattern manifests in 2+ areas: cognition (perception of self, others, events), affectivity (range, lability, appropriateness of emotional response), interpersonal functioning, impulse control - is unflexible and pervasive across personal/social situations - leads to clinically significant distress or impairment in social, occupational, other areas of functioning - not better accounted for by another mental disorder - not due to substance abuse of a general medical condition Unlike axis 1 disorders these are: - permanent (or long lasting), generative (rather than reactive), dominated mainly by behavior and relationships w/ others (rather than symptoms), dx on basis of long-term functioning (rather than present state) |
|
|
Paranoid Personality Disorder
|
DSM-IV cluster A
Characteristics: - externalization and projection of internal anger and hostility - anger and hostility - feelings of victimization - difficulties understanding actions of others - hypersensitivity to slights - lack of close relationships - tendency for reasoning to become severely impaired under stress Epi: - more common in males - 10-30% in inpatient psych, .5-2.5% in general population - increased prevalence if relatives w/ schizophrenia and delusional disorder, persecutory type |
|
|
Schizoid Personality Disorder
|
DSM-IV cluster A
Characteristics: “Hermit” - neither enjoys nor desires close relationships or sexual experiences - takes pleasure in few activities - comes off as cold and detached (but may be more sensitive than appearing) |
|
|
Schizotypal Personality disorder
|
DSM-IV cluster A
Characteristics: “Schizo light” - don’t interact with others, when they do they’re ‘odd’ (odd beliefs or magical thinking, ideas of reference, unusual perceptual experiences, odd thinking/speech) - suspiciousness or paranoid ideation, inappropriate or constricted affect - experience considerable psychological pain and depression - interpersonal avoidance motivated by fear of embarrassment or humiliation, anxiety - feelings of inadequacy - inhibitions about pursing gratifications - empirically indistinguishable from schizoid personality disorder Course: relatively stable w/ only a small portion progressing to schizophrenia |
|
|
Antisocial Personality Disorder
|
DSM-IV cluster B
Characteristics: “Jade” - sociopath - often reckless and law breaking, disinhibited (disregard for safety of self or others), failure to conform to social norms - irritability and aggressiveness, repeated fights/assaults (more for men) - lack empathy/remorse/regard for others as human beings - tendency to manipulate other’s emotions, able to fake emotion, can be charming - not internal sense of morality, intellectually aware of right/wrong and socially established consequences - sadistic: get pleasure from manipulating of hurting others Chronic: no treatment, must be 18 to diagnose (conduct disorder <18) |
|
|
Borderline Personality Disorder
|
DSM-IV cluster B
Characteristics: “bipolar light” - lie on the line between neurotic and psychotic - chaotic personal relations - extreme emotional lability, moody, trouble controlling anger - splitting of good/bad - projection, denial - self-mutilating behavior (cutting, burning, suicide) - chronic feeling of emptiness Course: - can’t diagnose before 18 - variable course, commonly chronic instability in early adulthood w/ episodes of serious affective & impulsive dyscontrol (self harm) - risk of suicide highest in young adulthood |
|
|
Histrionic personality disorder
|
DSM-IV cluster B
Characteristics: “Hollywood” - self-dramatization - emotionality - seductiveness - dependency/helplessness - non-compliance - self-indulgence - suggestibility - somatization - in males: macho, bragging about athletic ability |
|
|
Narcissistic Personality
|
DSM-IV cluster B
Characteristic: “Kanye West” - grandiosity with internal sense of inadequacy - lack of empathy - entitlement - motivated by avoidance of shame/humiliation - envious, interpersonally manipulative - devaluation and idealization (if it’s not mine it’s not important) - can also manifest in shy/covert type: inhibited/shy or self-effacing, tendency for hypochondria, highly sensitive for praise/slights, responds to criticism w/ inner shame/anger and flattery w/ exaggerated feeling of pleasure/superiority, lacks empathy but can fake it, compensatory grandiose fantasies for real accomplishments Clinical Course : - don’t deal with natural process of aging well - pathological narcissism at the extreme is antisocial PD |
|
|
Avoidant Personality disorder
|
DSM-IV cluster C
Characteristics: “super shy” - wants to interact but is anxious about it resulting in isolation - unwilling to get involved w/ people unless certain of being liked - preoccupied w/ being criticized or rejected in social situations Course: - tends to remit with age |
|
|
Dependent Personality Disorder
|
DSM-IV cluster C
- needs others to assume responsibility for most major areas of his/her life - difficulty disagreeing with others - difficulty w/ leadership positions - dose to excessive lengths to obtain nurturance and support from others (including doing unpleasant things) - preoccupied w/ fears of being left to take care of him/herself |
|
|
Obsessive-compulsive personality disorder
|
DSM-IV cluster C
Characteristics: “med student” - difficulty experience feelings of warmth, lover, and tenderness - feels strong when angry/defiant and weak when fearful/guilty - indecisive - perfectionism, loves list making - excessive morality and preoccupation with rules (moral rigidity) - postponement of pleasure - preoccupation with self-sufficiency - feelings of contempt for others who appear helpless |
|
|
Melancholic Depression
|
5/9 of following criteria: (cannot be melancholic without major depression)
Anhedonia Early morning awakening (sleep disorder: wake up early and can’t go back to sleep) Poor appetite with weight loss Psychomotor retardation or agitation Lack of mood reactivity (ex: no reaction to jokes) Diurnal variation (feels better as the day goes on, but still depressed) Previous major depression with remission Full response to somatic therapy No significant personality disturbance |
|
|
Atypical Depression
|
Aka: Rip Van Winkle/Anti-Atkins depression
Overeating and carbohydrate craving Spending more time in bed (not necessarily sleeping; “leaden fatigue” – extremely fatigued) Low self-esteem and guilt Common in bipolar depression |
|
|
Somatization Disorder
|
Criteria:
- variety of chronic complaints in multiple organ systems over a period of years. - Must include: 4 pain sx, 2 GI sx, 1 sexual sx, 1 pseudoneurologic sx - this is an unconscious process Course: - patients give inconsistent, colorful and dramatic histories, lacking facts - increased risk of iatrogenic injury (need to limit tests/procedures to those clearly indicated) - 75% axis 1 comorbidity also a lot of other subtle diseases Epi: starts <30y, 1-5% of primary care |
|
|
Conversion disorder
|
= a sudden loss of sensory or motor function (paralysis, blindness, mutism, stocking/glove anesthesia, non-epileptic seizures, vomiting, urinary incontinence) often following an acute stressor (not related to sex or pain)
- unconscious production of symptoms for unconscious gain - gain can be primary (reduction of anxiety, avoidance of conflict) or secondary (assuming sick role, avoiding responsibility) - La Belle Indifference: patient may be aware of but indifferent toward symptoms Epi: - usually people from rural, low socioeconomic, low education areas - 67% abuse history, recurrent in 25% of patients - 5-12% actually have neurologic disorder |
|
|
Pain Disorder
|
= Prolonged pain w/o physical findings w/ impaired function
- pain is the predominant focus of the clinical presentation and psychological factors play an important role in the severity, exacerbation, or maintenance of the pain - not feigned, deliberate, or related sex D’s: disability, disuse and degeneration, drug misuse, doctor shopping, dependency (emotional), demoralization, depression, dramatic accounts - 10-15% of adults (lower back pain) |
|
|
Hypochondriasis
|
= preoccupation w/ and fear of having serious illness despite medical evaluation and reassurance
- not delusional or feigned - Intense preoccupation, part of OCD spectrum |
|
|
Body Dysmorphic Disorder
|
= preoccupation w/ minor or imagined defect in appearance leading to significant emotional distress or impairment of functioning
- patients often repeatedly seek plastic surgery - part of OCD spectrum - MDD is most common comorbidity |
|
|
Facticious disorder/Munchausen
|
= patient consciously creates physical and/or psychological symptoms to achieve unconscious gain (medical/social attention—primary gain)
- May be by proxy causing illness in childhood by caregiver |

