![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
49 Cards in this Set
- Front
- Back
|
Storage polysaccharides like ***(animals) and ***(plants) are broken down into *** which is metabolized via glycolysis.
|
glycogen
starch glucose 6-phosphate |
|
|
Cooperativity can be defined in terms of the ***.
|
Cooperativity Index (Rx)
|
|
|
--- Allosteric Constant ---
Is the equilibrium constant between L and R when no ligand is present. 1) Has the unit of ***. 2) Has a formula of... 3) dissociation constant for... 4) formation constant for... |
1) L
2) [T2]\[R2] 3) R2 4) T2 |
|
|
--- Simple Uncatalyzed Reaction ---
From the law of *** *** the reaction velocity is given by: v = k[S]. |
mass action
|
|
|
--- Lactose ---
1) Disaccharide composed of... 2) Reducing sugar? 3) α/β linked? 4) Broken down by... |
1) galactose --- glucose
2) Yes 3) β/α 4) lactase |
|
|
---M: monomeric subunit (termed protomer)
---M2 is the dimeric protein. 1) If 1st ligand causes a LARGE affinity increase for the 2nd it has *** Cooperativity. 2) There will be either NO ligand OR saturated w/ligand. |
1) Complete
|
|
|
--- General MWC Equation ---
1) The greater the value for L, the more *** the plot. 2) If L = 0 or n = 1, a *** curve is obtained. |
1) sigmoidal
2) hyperbolic |
|
|
--- Enzyme Kinetic Study ---
1) *** concentration is varied. 2) *** concentration is constant. 3) The effect on reaction *** is studied. |
1) Substrate
2) Enzyme 3) velocity |
|
|
--- Sucrose ---
1) Disaccharide composed of... 2) Reducing sugar? 3) α/β linked? 4) Broken down by... |
1) fructose --- glucose
2) No 3) α/β 4) sucrase (invertase) |
|
|
A *** is the structural unit of an oligomeric protein.
|
protomer
|
|
|
The *** model is consistent w/sigmoidal binding even though it's assumed there's no protomer cooperativity.
|
MWC
|
|
|
1) [E0] (free enzyme at *** ***) which is equal to [ET].
2) [ET] = [E]+[ES] --- is called the *** equation. 3) The *** assumption [ES] at any given time will be... k1[E][S] = k-1[ES] |
1) zero time
2) conservation 3) equilibrium 4) |
|
|
--- Trehalose ---
1) Disaccharide composed of... 2) Reducing sugar? 3) α/β linked? 4) Broken down by... |
1) glucose --- glucose
2) No 3) α/α 4) trehalase |
|
|
--- MWC Model ---
•In absence of ligand, equilibrium favors *** state. •Ligand binding shifts equilibrium toward *** state. |
T
R |
|
|
The *** relationship is the equilibrium constant for a particular enzyme reaction in terms of VMax and KM.
|
Haldane
|
|
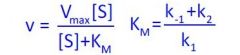
What equation is this?
|
Michaelis-Menten equation
|
|
|
--- Maltose ---
1) Produced by enzyme *** during breakdown of 2) grain starches *** and ***. In mammals, 3) it's then hydrolyzed into *** by enzyme ***. |
1) diastase
2) amylose and amylopectin 3) (2) glucose --- maltase |
|
|
The Adair equation describe binding under the *** model.
|
KNF
|
|
|
--- Cooperativity Index ---
1) If h = 1 then Rx = *** (*** type) 2) If h = 2 then Rx = *** 3) If h = 4 then Rx = *** |
1) 81 (Michaelis-Menten type)
2) 9 3) 3 81^(1/4) |
|
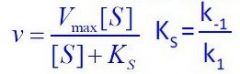
What equation is this?
|
Steady-State equation
|
|
|
--- Maltose ---
1) Amylose and amylopectin are similar to glycogen but.... 2) Other proteins uses to break down grains also include *** and ***... |
1) have no/little branching
2) proteases and lipases. |
|
|
1) The classical ligand/protein binding models used the proteins *** and ***.
2) One of these proteins has a ligand that w/*** effects rather than ***. |
1) myoglobin --- hemoglobin
2) homotropic --- heterotropic |
|
|
1) An alternative to the Michaelis-Menten
model is the.... 2) This model assumes that... |
1) steady-state assumption
2) over time ES levels reach a steady state |
|
|
--- Malting Process ---
1) # of steps 2) Which are...? 3) How long is each step? |
1) 4
2) soak - germinate - heat drying/curing 3) 2-3 --- 7-14 --- 4(2 & 2) |
|
|
Myoglobin --- Hemoglobin
1) Binding Sites? 2) Location? 3) Curve? |
1) 1 --- 4
2) muscle --- blood 3) hyperbolic --- sigmoidal |
|
|
A comparison of *** values may provide information about the amount of enzyme in different tissues.
|
Vmax
|
|
|
1) ***: process in which cereal grains
2) soak, germinate, and dry in a ***. 3) Heat Drying temp? 4) Heat Curing temp? |
1) kiln
2) Malting 3) 32-37 C 4) 78-100 C |
|
|
1) General binding curve of Langumir equation is...
2) *** is equivalent to the ligand concentration giving ½ fractional saturation. |
1) rectangular hyperbola
2) Kd |
|
|
Vmax depends on the amount of *** used whereas KM does not.
|
enzyme
|
|
|
--- Fructose ---
1) In skeletal muscle, it's metabolized by ***. 2a) In liver, acted on by *** and 2b) metabolized by *** and *** kinase. |
1) hexokinase
2a) fructokinase 2b) fructose 1-P --- triose |
|
|
Hemoglobin transitions between low and high affinity states due to *** w/in it. This results in a *** binding curve
|
1) cooperativity
2) sigmoidal |
|
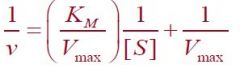
What equation is this?
|
Lineweaver-Burk Equation
|
|
|
Disaccharide Enzymes
1) Lactose 2) Sucrose 3) Trehalose 4) Maltose |
1) Lacase
2) sucrase (invertase) 3) trehalase 4) maltase |
|
|
--- pO2 ---
1) Lungs? 2) Peripheral tissues? |
1) 13.3 kPa
2) 2 kPa |
|
|
Briggs and Haldane came up with what?
|
Steady-State Assumption
|
|
|
--- Mannose ---
1) ***saccharide metabolized to *** which is 2) an intermediate in the *** pathway. 3) 1st --- *** Kinase 4) 2nd --- Phosphomannose *** |
1) mono --- fructose 6-P
2) glycolysis 3) Hexose Kinase 4) Phosphomannose isomerase |
|
|
--- Adair Equation ---
For the binding of a ligand to a protein w/2 binding sites for the ligand. 1) M2: two protomers are... 2) M2S 3) M2S2 |
1) both unbound
2) 1 bound protomer/1 unbound 3) both bound |
|
|
--- Mannose ---
1) Hexose Kinase Mannose + ATP ⇌ *** + ADP 2) Phosphomannose isomerase xxx ⇌ *** |
1) Mannose 6-P
2) Fructose 6-P |
|
|
--- Adair Equation ---
--Dimeric and NO Cooperativity-- 1) M + S ⇌ MS is the *** binding constant labeled K***. 2) M2 + S ⇌ M2S is the *** binding constant labeled K***. 3) M2S + S ⇌ M2S2 is the *** binding constant labeled K***. 4) Kb1 = ***Kb 5) Kb2 = ***Kb |
1) intrinsic --- Kb
2) apparent --- Kb1 3) apparent --- Kb2 4) Kb1 = 2Kb 5) Kb2 = ½Kb |
|
|
--- Ethanol Synthesis ---
1) Created when *** (generated during glycolysis) is re*** 2) with *** serving as final electron acceptor. |
1) NADH --- reoxidized
2) acetaldehyde |
|
|
--- LIVER Enzyme Locations ---
1) Alcohol DH 2) Cytochrome P-450 3) Catalase 4) ALdehyde DH |
1) cytosol (ADH)
2) microsomal (smooth ER) 3) peroxisomes 4) MITOCHONDRIA (ALDH) |
|
|
--- Ethanol Metabolism LIVER ---
1) 1st pathways ⇌ *** 2) mito ALDH ⇌ *** 3) then enzyme *** prepares it for 4) TCA cycle by ⇌ *** |
1) Acetaldehyde
2) Acetate 3) Acetyl-CoA synthetase 4) Acetyl-S-CoA |
|
|
--- Ethanol Metabolism ---
Maximum Theoretical ATP? 1) Alcohol DH 2) ALdehyde DH 3) Catalase |
1) 13 (5)
2) 8 (-2.5, 2.5) 3) 10.5 (2.5) (-2, ???, ???, 10) |
|
|
--- Ethanol Metabolism ---
Enzyme most important at this concentration... 1) High 2) Low 3) How many pathways are there? |
1) Cytochrome P-450 OR MEOS
2) Alcohol DH 3) 3 |
|
|
--- Cytochrome P-450 ---
1) Falls w/in class called... 2) Consisting of *#* families and *#* subfamilies |
1) mixed-function oxidases OR monooxygenases
2) 18--- 43 4) |
|
|
--- Alcohol Enzymes ---
Ethanol + (???) ⇌ Acetaldehyde + (???) 1) Alcohol DH 2) Cytochrome P-450 3) Catalase |
1) NAD+ ⇌ NADH/H+
2) NADPH/H+/O2 ⇌ NADP+/H2O 3) H2O2 ⇌ (2) H2O |
|
|
--- Aldehyde dehydrogenase (ALDH) ---
Substrates ⇌ Products |
H2O, NAD+, Acetaldehyde
⇌ NADH, H+, Acetic Acid |
|
|
MEOS?
1) Is a mixed function *** associated 2) with the *** *** of liver cells. 3) requires *** and *** to function |
Microsomal Ethanol Oxidizing System
1) oxidase 2) endoplasmic reticulum 3) NADPH and O2 |
|
|
printed
|
printed
|

