![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
39 Cards in this Set
- Front
- Back
|
nd(corr) |
nd(obs) + .00045 (T-20) |
|
|
bp(Corr) |
bp(obs) +bp(obs)/33 |
|
|
as elevation gets higher, boiling point gets |
Lower |
|
|
Density formula |
Mass/volume |
|
|
Lucas Test |
ROH + HCl(aq) equilibrium with ZnCl2 ---------> RCL H2O |
|
|
A bilayer will form if R is |
Big, >2 Carbons |
|
|
If bilayer forms quickly, |
Then carbon chain is long |
|
|
If 2 boiling points have >50° difference, use |
Simple distillation |
|
|
If boiling point has <50° boiling point difference, use |
Fractional distillation |
|
|
What distillation lowers pressure and lowers boiling point? |
Vacuum filtration |
|
|
What distillation method is good for temperature sensitive molecules? |
Vacuum distillation |
|
|
What distillation method is good for volatile compounds? (They evaporate readily) |
Steam |
|
|
Which distillation includes a beaker on a hot plate? |
Practical |
|
|
Which distillation can you NOT put volatile compounds in? |
Practical |
|
|
What distillation is used for small quantities? |
Micro |
|
|
You would use micro distillation if you have ____ml or less |
5 |
|
|
What does the silver nitrate test do |
To test for halogens |
|
|
What is a potassium permanganate test for |
Test for acidity |
|
|
What four functional groups do not oxidize? |
Alkanes, ketones, alkyl halides, and tertiary alcohols |
|
|
Potassium permanganate reaction |
KMnO4 + FG -----> MnO4 + FG |
|
|
Silver nitrate reaction |
RX + AgNO3 -------> R-ONO2 + AgX |
|
|
Strong acid |
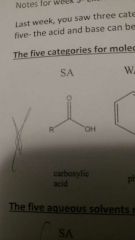
|
|
|
Weak Acid |
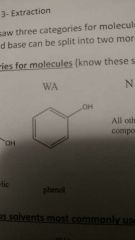
|
|
|
Weak Base |
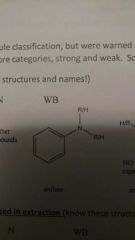
|
|
|
Strong base |
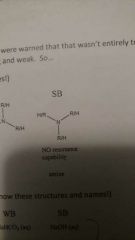
|
|
|
Aqueous solvents: strong acid |
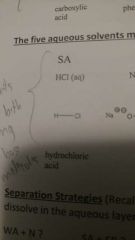
|
|
|
Aqueous solvents: weak acid |

|
|
|
Aqueous solvents: neutral |
Water |
|
|
Aqueous solvents: weak base |
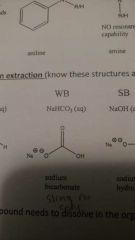
|
|
|
Aqueous solvents: strong base |

|
|
|
Strong acids react with |
Strong and weak bases |
|
|
Weak acids react with |
Strong bases only |
|
|
Weak bases react with |
Strong acids only |
|
|
Strong bases react with |
Strong and weak acids |
|
|
How do you use pH paper? |
Always dip a glass today into the solution and then touch pH paper |
|
|
What does red on pH paper mean |
Acid |
|
|
What does blue on pH paper mean |
Basic |
|
|
The silica gel is polar or nonpolar? |
Polar |
|
|
Will a polar spot move farther than a nonpolar spot during tlc? |
No |

