![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
131 Cards in this Set
- Front
- Back
|
What percent of the GI is the small intestine? What percent of tumors occur here?
|
- SI is 75% of length
- But only 3-6% of tumors are here |
|
|
What are the most common tumors in the small intestine? Others?
|
- Most common: Adenoma (near ampulla)
- Others: carcinoid and adenocarcinoma have equal incidence - Rare: mesenchymal tumors, lipoma, GIST, lymphoma |
|
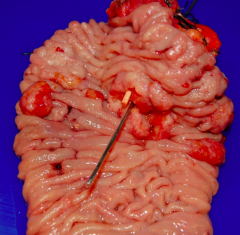
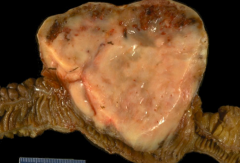
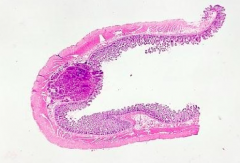
What does this specimen show?
|
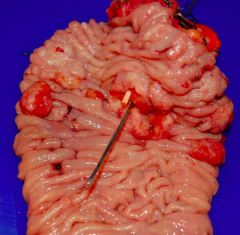
Tumors in small intestine by the Ampulla of Vader (cause back up of bile and pancreatic juices)
|
|
|
What are the risk factors for SI Adencarcinoma?
|
- Crohn’s disease
- Adenomas - Celiac disease - Familial polyposis syndrome |
|
|
What is the most common non-epithelial (soft tissue) tumor in the GI tract?
|
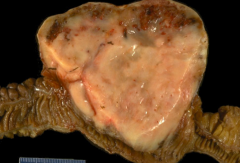
Gastrointestinal Stromal Tumor (GIST)
|
|
|
What is the origin of the tissues in Gastrointestinal Stromal Tumor (GIST)?
|
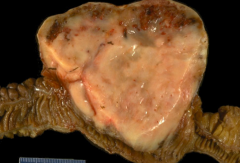
Mesenchymal origin, derived from interstitial cells of Cajal (pacemaker cells)
|
|
|
What is Gastrointestinal Stromal Tumor (GIST) seen in?
|
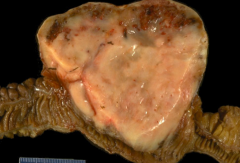
- Carney triad
- Neurofibromatosis (no KIT or PDGFRA mutation) - Carney-Stratakis syndrome (succinate dehydrogenase) |
|
|
What drug is clinically useful in Gastrointestinal Stromal Tumor (GIST)?
|
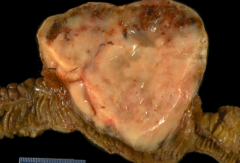
Gleevac (Imatinib)
|
|
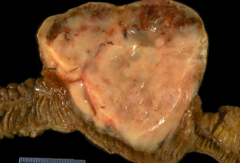
What kind of tumor is this?
|
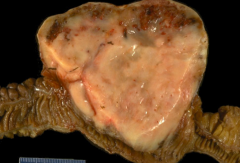
Gastrointestinal Stromal Tumor (GIST)
|
|
|
What are the most common locations of Gastrointestinal Stromal Tumors (GIST)?
|
- Stomach (60%)
- Small intestine (30%) - Colon (4%) |
|
|
What mutations are common in Gastrointestinal Stromal Tumor (GIST)?
|
- C-Kit mutations (80%)
- PDGFRA mutations (5-10%) |
|
|
What are the immunohistochemical identifications of Gastrointestinal Stromal Tumor (GIST)?
|
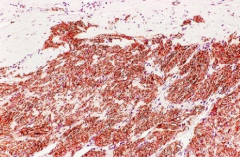
- CD117 (C-Kit) - positive in 90-100% of cases
- DOG1 - specific marker - CD34 - positive in 85-95% of cases - Muscle markers - actin (25%), desmin (10%), and S-100 (5%) |
|
|
What are the microscopic identifications of Gastrointestinal Stromal Tumor (GIST)?
|
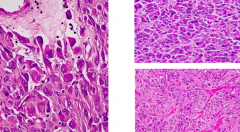
- Cells are EPITHELIOID
- More pleomorphic and with deeply acidophilic cytoplasm - Features of smooth muscle differentiation (at immunohistochemical level - see markers of actin, desmin, and S-100) |
|
|
What are the immunohistochemical signs that are diagnostic for Gastrointestinal Stromal Tumor (GIST)?
|
- Few cells show c-KIT immunoreactivity, rendering a
diagnosis of gastrointestinal stromal tumor not that secure. - Extensive positive staining for DOG1 supports the diagnosis |
|
|
What kind of genes are c-kit (CD117) and PDGFRA?
|
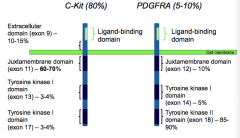
Cell surface receptors - Tyrosine Kinase types
|
|
|
What kind of tumor is a Carcinoid Tumor? What does that mean?
|
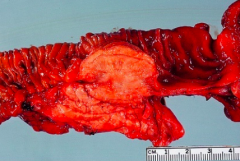
Neuroendocrine tumor:
- Secretes bioactive compounds (biogenic amines) - Elevated serotonin (5-HT) |
|
|
What are the signs of Carcinoid Syndrome?
|
- Vasomotor disturbances
- Intestinal hypermotility - Wheezing - Hepatomegaly - Cardiac involvement |
|
|
How does a Carcinoid Tumor present?
|
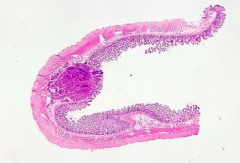
Submucosal nodule
|
|
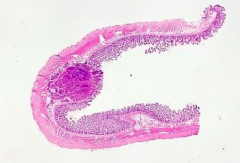
What does this slide show?
|
Submucosal Nodule - this is how Carcinoid Tumors present
|
|
|
What do you see in MALT Lymphoma in the small intestine?
|
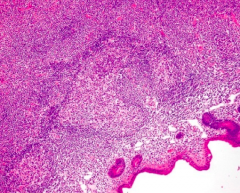
Colonization of the germinal centers by the tumor cells
|
|
|
What is the term for an epithelium-derived tumor mass which protrudes into the gut lumen?
|
Polyp
|
|
|
What is a polyp? What are the types?
|
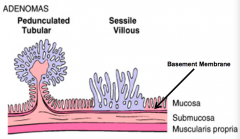
- Epithelium-derived tumor mass which protrudes into the gut lumen
- Pedunculated polyps - Sessile polyps |
|
|
What is a non-neoplastic polyp the result of? Prognosis?
|
Abnormal mucosal maturation, inflammation, distorted architecture - no malignant potential
|
|
|
What is a neoplastic polyp the result of? Prognosis?
|
Arises as a result of proliferation and dysplasia (adenomas) - precursors of carcinoma
|
|
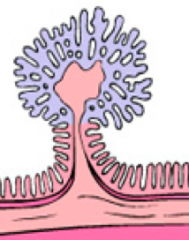
What is this?
|
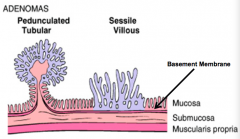
Pedunculated, Tubular Polyp
|
|
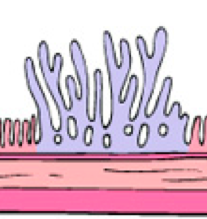
What is this?
|
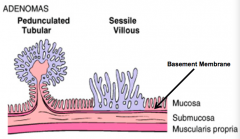
Sessile, Villous Polyp
|
|
|
What kind of polyps are the result of abnormal mucosal maturation, inflammation, and distorted architecture? Types? Malignant potential?
|
Non-neoplastic polyps:
- Hamartomatous - Inflammatory - Lymphoid No malignant potential |
|
|
Hamartoma:
- Benign or malignant - Structure - Characteristics - Cause - Location |
- Benign tumors
- Focal, mass of tissue resembling a tumor, pedunculated, 1-3 cm - Composed of mature, histologically normal elements from a site that grow in a disorganized manner - Due to developmental errors - Occur in many sites (80% in rectum) |
|
|
What kind of polyp is composed of mature, histologically normal elements that grow in a disorganized manner?
|
Hamartomas
|
|
|
What are the types of / syndromes with Hamartomatous Polyps?
|
- Juvenile Polyposis Syndrome
- Peutz-Jeghers Syndrome - Cowden Syndrome - Cronkhite-Canada |
|
|
What is the cause of Hamartomas? Who gets them?
|
- Due to developmental error
- Juvenile: kids <5 years |
|
|
What is the most common location of Hamartomatous polyps? Appearance / size?
|
- 80% occur in rectum
- Pedunculated, 1-3cm (this is large by polyp standards) |
|
|
What happens to the components of the mucosa in Hamartomatous Polyps?
|
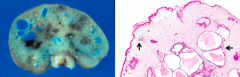
- Expanded lamina propria with variable inflammation
- Lamina propria constitutes bulk of polyp - Abundant, cystically dilated, tortuous glands |
|
|
What are the criteria of Juvenile Polyposis Syndrome? How is it inherited?
|
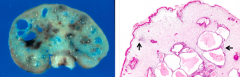
- Multiple juvenile hamartomatous polyps (>5, 50-100)
- Found in stomach, small intestine, colon, and/or rectum - Increased risk of adenomas, 10-50% lifetime incidence of colon cancer, also gastric, small intestine, and pancreas - Autosomal dominant |
|
|
How is Juvenile Polyposis Syndrome inherited? What genes are abnormal? What are these patients at increased risk for?
|
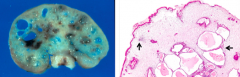
Autosomal dominant:
- SMAD4 (20%) - BMPR1A (20%) - PTEN (0%) Increased risk of adenomas - 10-50% lifetime incidence of colon cancer - Also gastric, small intestine, and pancreas |
|
|
What are the criteria of Peutz-Jeghers Syndrome? How is it inherited?
|
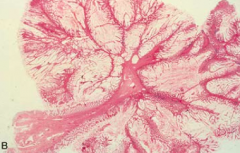
- Multiple Hamartomatous GI Peutz-Jeghers polyps (non-malignant)
- Hyperpigmentation: mucosal (mouth) and cutaneous (fingers) - Large and pedunculated polyps containing CT and smooth muscle as well as abundant glands rich in Goblet cells - Autosomal dominant: STK11 |
|
|
How is Peutz-Jeghers Syndrome inherited? What genes are abnormal? What are these patients at increased risk for?
|
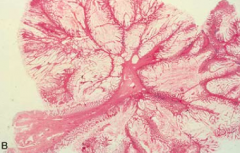
Autosomal dominant:
- STK11 Increased risk of: - Intussusception (inversion of one portion of the intestine within another) - Cancer of pancreas, breast, lung, ovary, and uterus (50% lifetime risk of all types of cancer) |
|
|
What are the characteristics of Cowden Syndrome? How is it inherited?
|
- Hamartomatous GI polyps (non-malignant)
- Facial trichilemmomas, oral papillomas, acral keratoses - Autosomal dominant |
|
|
How is Cowden Syndrome inherited? What genes are abnormal? What are these patients at increased risk for?
|
Autsomal dominant
Increased risk of: - Thyroid and breast cancer |
|
|
What are the characteristics of Cronkhite-Canada? How is it inherited?
|
- GI hamartomatous polyps
- Ectodermal abnormalities (nail atrophy, alopecia) - Non-hereditary |
|
|
What are the other non-neoplastic polyps?
|
- Inflammatory polyps (pseudopolyps)
- Lymphoid polyps |
|
|
What are the characteristics of inflammatory polyps?
|
- Non-neoplastic
- Pseudopolyps - regenerating mucosa adjacent to ulceration (severe IBD) |
|
|
What are the characteristics of lymphoid polyps?
|
- Non-neoplastic
- Mucosal bumps - Caused by intramucosal lymphoid follicles - normal |
|
|
What are the types of Serrated Polyps?
|
- Hyperplastic Polyps
- Sessile Serrated Polyps |
|
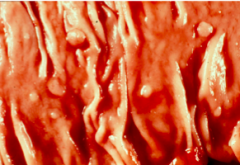
What are the smooth protrusions of mucosa usually at the tops of mucosal folds?
|
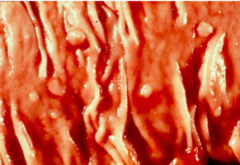
Serrated Polyps
|
|
|
What is the most common location of Serrated Polyps?
|
Over half are found in the rectosigmoid colon
|
|
|
What is the histological appearance of Serrated Polyps?
|
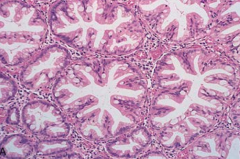
Serrated lumina with increased numbers of goblet cells
|
|
|
What is the size of Serrated Polyps?
|
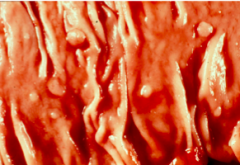
Very small - less than 5 mm in diameter
|
|
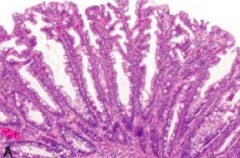
What kind of polyp is this? Location? Malignant potential?
|
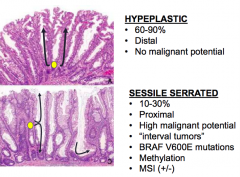
Hyperplastic Serrated Polyp
- 60-90% of serrated polyps - Distal colon - No malignant potential |
|
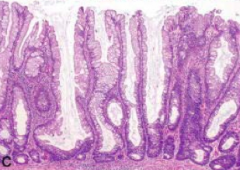
What kind of polyp is this? Location? Malignant potential?
|
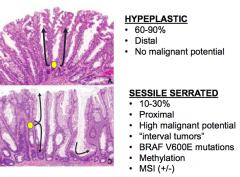
Sessile Serrated Polyp
- 10-30% of serrated polyps - Proximal colon - High malignant potential |
|
|
What kind of mutations are seen in Serrated Polyps?
|
- Hyperplastic: none discussed
- Sessile Serrated: BRAF V600E mutations |
|
|
What is the progression of polyps to cancer?
|
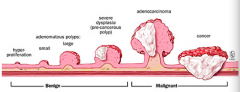
Benign
1. Starts as hyper-proliferation 2. Grows into small and large adenomatous polyps 3. Leads to severe dysplasia (pre-cancerous polyp) Malignant 4. Continues to grow into adenocarcinoma 5. Cancer progresses deep into tissues |
|
|
What do Adenomatous Polyps (Adenomas) arise from?
|
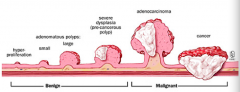
As a result of epithelial proliferative dysplasia
|
|
|
What can Adenomatous Polyps progress to?
|
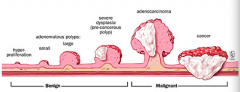
Precursor lesion for adenocarcinoma (4x greater risk if you have adenomatous polyps)
|
|
|
How common are Adenomatous Polyps? Who is at greatest risk?
|
- Common: 40-50% after age 60 (20-30% before age 40)
- 4x greater risk if you have 1st degree relative with adenomas - 4x greater risk of carcinoma if you have adenoma - M = F |
|
|
What are the types of Adenomatous Polyps?
|
1. Tubular Adenoma
2. Villous Adenoma 3. Tubulovillous Adenoma |
|
|
What is the morphological appearance of Tubular Adenomas? Location?
|
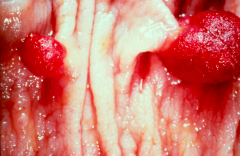
- Tubular glands
- Often small, pedunculated - Can be single or multiple - Rarely >2.5 cm - Dysplastic epithelium - elongated, pseudostratified, hyperchromatic nuclei, w/ loss of mucin production - 90% found in colon |
|
|
What is the morphological appearance of Villous Adenomas? Location?
|
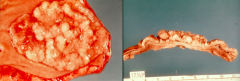
- Villous projections
- Often large and sessile - Up to 10 cm in diameter - Most common in older people in rectosigmoid colon |
|
|
When invasive carcinoma is present, what is the appearance of the Villous Adenoma? Where does invasion occur?
|
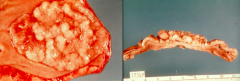
- When invasive carcinoma is present, there is no stalk to act as a buffer zone
- Cancer invasion occurs directly into the colon wall |
|
|
When is the cancer risk rare in Adenomas?
|
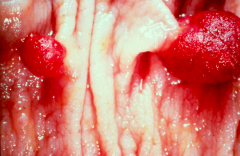
Tubular Adenomas < 1 cm
|
|
|
When is the cancer risk increased in Adenomas?
|
- 40% in patients with sessile, villous adenomas > 4cm
- High grade dysplasia is often found in the villous area - High grade dysplasia or carcinoma can be found in any polyp |
|
|
What are the symptoms of Adenomatous Polyps?
|
- May be asymptomatic
- May present with rectal bleeding or anemia |
|
|
Where does intramucousal carcinoma invade? Metastatic potential?
|
Invades lamina propria - no metastatic potential
|
|
|
Where does invasive carcinoma in a pedunculated adenoma invade? Metastatic potential?
|
- Invades through muscularis mucosa
- Metastatic potential |
|
|
How should you treat an invasive carcinoma in a pedunculated adenoma?
|
Endoscopic removal adequate if:
- Resection margins negative - No vascular or lymphatic invasion - Carcinoma is not poorly differentiated |
|
|
How should you treat an invasive carcinoma in a sessile polyp?
|
Polypectomy inadequate, requires partial colectomy
|
|
|
How should you treat Adenomatous Polyps?
|
- Regardless of whether carcinoma is present, the ONLY ADEQUATE TREATMENT is COMPLETE RESECTION
- If adenomatous epithelium remains int he patient, there is potential for carcinoma |
|
|
What is the most common type of Colorectal Cancer?
|
98% Adenocarcinoma (99% occur singly)
|
|
|
What is the prognosis of Colorectal Cancer? When is the peak incidence? Who is at increased risk?
|
- 9% of all cancer deaths in US
- 90% diagnosed at age 50+ - 1-3% occur in familial syndromes or IBD |
|
|
What are the potential locations of Colorectal Cancer? How common in each location?
|
- Rectosigmoid colon (55%)
- Cecum and ascending colon (22%) - Transverse colon (11%) - Descending colon (6%) |
|
|
Where are the highest death rates from Colorectal Cancer?
|
- US
- Australia - New Zealand - Eastern Europe |
|
|
What are the lifestyle risk factors for Colorectal Carcinoma?
|
Dietary practices:
- Excess dietary caloric intake - Low fiber - High content of refined carbohydrates - Red meat - Decreased intake of micronutrients Obesity and physical inactivity |
|
What are the clinical implications of right-sided Colon Cancer?
|
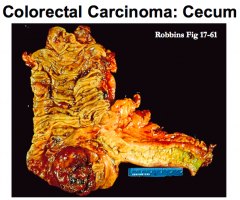
- Non-obstructive
- Fatigue, weakness - Iron deficiency anemia - Polypoid, exophytic lesions |
|
What are the clinical implications of left-sided Colon Cancer?
|
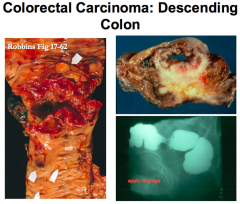
- May be obstructive
- Occult bleeding - Changes in bowel habits - Abdominal discomfort - Annular "napkin ring" constrictions - Tend to be more infiltrative |
|
|
Colorectal carcinoma on what side is more severe, may be obstructive, and tends to be more infiltrative?
|
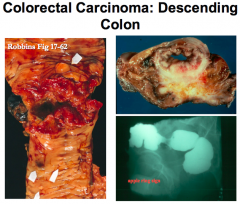
Left-sided lesions
|
|
|
What is the most common clinical presentation of colorectal cancer?
|
- Asymptomatic for years w/ insidious onset
- Iron deficiency anemia in an older male signifies colon cancer unless proven otherwise |
|
|
If you have an older male that is presenting with iron deficiency anemia, what do you suspect?
|
Colon cancer until proven otherwise
|
|
|
How do adenomas relate to colorectal cancer?
|
- Populations with a high prevalence of adenomas have a high prevalence of colorectal cancer, and vice versa
- Similar distribution of polyps and cancer in the colorectum - Peak age for adenomas precedes peak age for cancer - When cancer is identified at an early age, surrounding adenomatous tissue is often present - Cancer risk directly related to number of adenomas - Removal of polyps decreases cancer incidence |
|
|
How can you decrease your risk of colorectal cancer?
|
Remove any adenomatous polyps
|
|
|
What can pretty much guarantee your development of colorectal cancer?
|
FAP Syndrome: Familial Adenomatous Polyposis
|
|
|
What is the molecular understanding of the adenoma-carcinoma relationship?
|
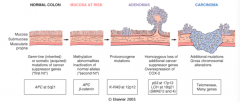
- Multi-hit hypothesis
- Accumulation of mutations is more important that the specific order of mutations |
|
|
What molecular findings are associated with the adenoma-carcinoma pathway?
|
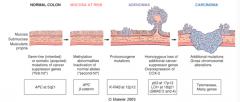
- >80% of colon cancers have inactivated APC
- 50% of cancers w/ APC mutations have β-catenin mutations (10% overall) - Loss of p53 occurs late in colon carcinogenesis |
|
|
What are the implications of inactivated APC gene?
|
Dysfunction of APC leads to:
- Increased WNT signaling - Decreased cell adhesion - Increased cellular proliferation >80% of colon cancers have inactivated APC |
|
|
What genetic change occurs late in colon carcinogenesis?
|
Loss of p53
|
|
|
What pattern of Colorectal Cancer is associated with Ulcerative Colitis?
|
Infiltrative Colorectal Cancer:
- Insidiously infiltrative - Difficult to identify grossly - Exceedingly aggressive - Spreads at early stage in evolution |
|
|
How can you grade the depth of invasion of Colorectal Cancer?
|
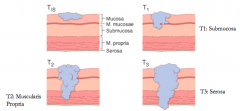
- T(in situ / IS) = just in mucosa
- T1 = invaded into submuocsa - T2 = invaded into muscularis propria - T3 = invaded through serosa - T4 = invades other organs or structures |
|
|
How can you grade the lymph node involvement of Colorectal Cancer?
|
- N0 = no LN metastasis
- N1 = metastasis in 1-3 LNs - N2 = metastasis in 4+ LNs |
|
|
How can you grade the metastasis of Colorectal Cancer?
|
- M0 = no distant metastasis
- M1 = distant metastasis |
|
|
What does a Stage 0 colon carcinoma indicate?
|

In situ carcinoma (tumor has not spread past the mucosa)
|
|
|
What does a Stage 1 colon carcinoma indicate?
|

- Tumor may have spread through submucosa (T1) or through muscularis propria (T2)
- No spread to lymph nodes or metastatic sites |
|
|
What does a Stage 2 colon carcinoma indicate?
|

- Tumor may have spread through serosa (T3) or into adjacent structures/organs (T4)
- No spread to lymph nodes or metastatic sites |
|
|
What does a Stage 3 colon carcinoma indicate?
|

- Tumor may have spread through any layer of the colon wall
- SPREAD TO LYMPH NODES - No distant metastasis |
|
|
What does a Stage 4 colon carcinoma indicate?
|

- Tumor may have spread through any layer of the colon wall
- Tumor may have spread to any number of lymph nodes - DISTANT METASTASES |
|
|
What is the most important prognostic indicator of colorectal carcinoma?
|
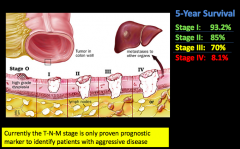
Extent of the tumor (stage) at the time of diagnosis
|
|
|
What is the prognosis for the different stages of Colorectal Cancer?
|
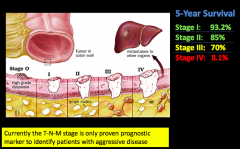
5-year survival:
- Stage 1: 93% - Stage 2: 85% - Stage 3: 70% - spread to lymph nodes - Stage 4: 8% - distant metastasis |
|
|
Where can Colon Cancer metastasize to?
|
- Direct extension into adjacent structures
- Spread to lymph nodes and vessels - Goes to regional lymph nodes, liver, lungs, and bones |
|
|
How common is metastatic spread of colon carcinoma upon presentation? Implications?
|
25-30% have metastasis (Stage 4) which indicates an 8% 5-year survival
|
|
|
What are the most common sites of metastasis of colon cancer?
|
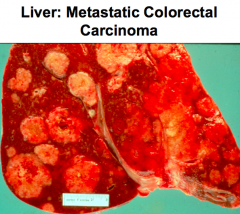
- Liver
- Lungs - Bones |
|
|
What can we use to figure out the best therapy for a tumor and the prognosis of the cancer?
|
Prognostic and Predictive Biomarker Assessment (look at molecular features of tumor)
|
|
|
What is the utility of analyzing biomarkers on tumors?
|
Helps determine prognosis and best treatment (surgery, surgery + chemo, or best supportive care)
|
|
|
What are the traditional agents for treating Colorectal Cancer? Characteristics?
|
- 5FU / LV
- Capecitabine - Irinotecan - Oxaliplatin - These cause all cells to die not just tumor cells; lots of side effects |
|
|
What are the targeted agents for treating Colorectal Cancer? Characteristics?
|
- Bevacizumab
- Cetuximab - Panitumumab - These only harm tumor cells, thus have a lot less side effects |
|
|
What are the kinds of biomarkers used to guide adjuvant therapy for colorectal cancer? Functions?
|
- Prognostic biomarkers - provide information about the patient's overall outcome, regardless of therapy (will it be more or less severe?)
- Predictive biomarkers - provide information about the effects of a particular therapeutic intervention (helps us figure out what drug to use) |
|
|
What gene is important in Colorectal cancer?
|
EGFR (ErbB1 / Her1)
|
|
|
Activation of EGFR (ErbB1 / Her1) gene leads to what?
|
Stimulates key processes involved in tumor growth and progression:
- Proliferation - Angiogenesis - Invasion - Metastasis |
|
|
What 3 major pathways are activated when EGFR (ErbB1 / Her1) is activated?
|
- RAS-RAF-MAP kinase
- PI3K-AKT - PLCγ |
|
|
What is the structure of EGFR (ErbB1 / Her1)?
|
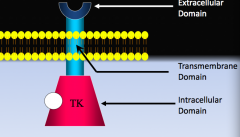
Transmembrane receptor with:
- Ligand binding domain (extracellular) - Transmembrane domain - Tyrosine kinase domain (intracellular) |
|
|
What drugs are being used to target EGFR (ErbB1 / Her1) = anti-EGFR?
|
- Cetuximab
- Panitumumab |
|
|
What percent of Colorectal Cancer cases benefit from EGFR monoclonal Ab therapy (cetuximab and panitumumab)?
|
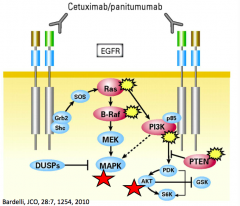
- Only 10-20% of cases benefit
- Patients who do not respond have mutations in signaling pathway further down (thus blocking the EGFR receptor does nothing) |
|
|
What is the major negative predictor of efficacy in EGFR monoclonal Ab therapy? What other mutation correlates with poor prognosis and lack of response?
|
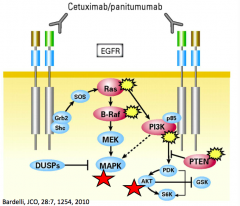
- KRAS mutation = major negative predictor
- BRAF mutation = poor prognosis and lack of response |
|
|
What are the hereditary syndromes involving the GI tract?
|
- Peutz-Jeghers, Cowden disease, and Juvenile polyposis
- Familial adenomatous polyposis (Gardner's syndrome and Turcot's syndrome) - MYH Associated Polyposis (MAP) - Lynch Syndrome / HNPCC (hereditary non-polyposis colorectal carcinoma syndrome) |
|
|
How are the hereditary syndromes involving the GI tract inherited?
|
Usually autosomal dominant
|
|
|
Familial Adenomatous Polyposis:
- Inheritance pattern - Number of lesions - Presence of adenocarcinoma - Treatment - Other |
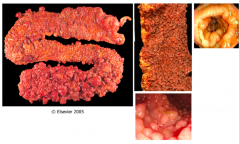
- Autosomal dominant
- Typically 500-2500 colonic adenomas (minimum of 100) - Colon adenocarcinoma occurs in ~100% - Treat with prophylactic colectomy |
|
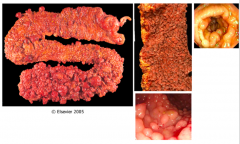
What is this syndrome?
|
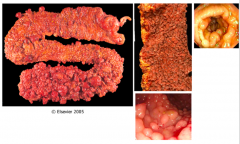
Familial Adenomatous Polyposis:
- Autosomal dominant - Typically 500-2500 colonic adenomas (minimum of 100) - Colon adenocarcinoma occurs in ~100% - Treat with prophylactic colectomy |
|
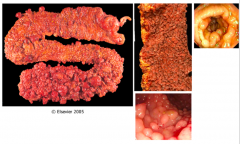
What causes Familial Adenomatous Polyposis?
|
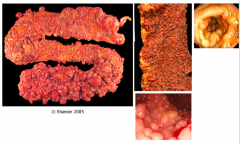
- Inheritance (autosomal dominant) of germline mutation in the APC gene
- ~25% of FAP patients have no family history (new mutations in APC gene) - Tumors result when the second normal allele is mutated in a cell during life ("somatic mutation") |
|
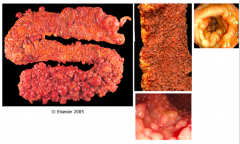
What is the age of onset of polyps in Familial Adenomatous Polyposis? Onset of colorectal cancer?
|
Polyps:
- Median age 16 years - Range 5-38 years - Increasing number are detected with age Colorectal Cancer: - Late 30s, early 40s |
|
|
What are the variants of Familial Adenomatous Polyposis?
|
- Attenuated FAP (<100 polyps)
- Gardner's syndrome - Turcot syndrome |
|
|
How does Attenuated Familial Adenomatous Polyposis differ from Classical FAP?
|
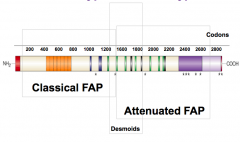
<100 polyps
|
|
|
How does Gardner's Syndrome differ from Classical Familial Adenomatous Polyposis?
|
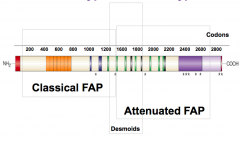
- Adenomatous polyposis with osteomas
- Epidermoid cysts - Desmoid tumors |
|
|
How does Turcot Syndrome differ from Classical Familial Adenomatous Polyposis?
|
Adenomatous polyposis with medulloblastoma
|
|
|
MYH Associated Polyposis (MAP):
- Inheritance pattern - Number of lesions - Other |
- Autosomal recessive
- Typically 20-100 adenomatous polyps (can be >100) - Phenotypic overlap with FAP (mostly attenuated FAP), but present at older age than for FAP |
|
|
What causes MYH Associated Polyposis (MAP)?
|
- Mutations in MYH gene (1p34.3-p32.1) inherited in autosomal recessive pattern
- MYH protein is important for DNA repair (base excision repair and repairs oxidation induced DNA damage by removing A mis-paired with G) |
|
|
What kind of cancer are patients with Lynch Syndrome at increased risk for?
|
- Colorectal Cancer
- Extra-Intestinal Cancer (endometrial cancer in females, also small intestine, ureter, and renal pelvis) |
|
|
What happens in Lynch Syndrome / Hereditary Non-Polyposis Colorectal Cancer?
|
- Adenomas occur considerably earlier than in the normal population, but in low numbers
- Increased risk of colorectal cancer and extra-intestinal cancer (endometrial cancer in females, small intestine, ureter, and renal pelvis cancer too) - Colonic carcinomas are often multiple and not necessarily associated with the adenomas |
|
|
What is the cause of Lynch Syndrome / Hereditary Non-Polyposis Colorectal Cancer?
|
Genetic defect involves DNA mismatch repair genes (microsatellite instability pathway)
|
|
|
How long does it take an adenoma to progress to carcinoma in Lynch Syndrome / Hereditary Non-Polyposis Colorectal Cancer? Normally?
|
- Lynch syndrome: accelerated to 2-3 years
- Sporadic CRC: 8-10 years |
|
|
How common is Lynch Syndrome in Colorectal Carcinoma patients?
|
1 in 35 have Lynch Syndrome
|
|
|
What is the risk for a second primary cancer in Lynch Syndrome?
|
16% in 10 years
|
|
|
What is the risk for new cancer in first/second degree relative of patient with Lynch syndrome?
|
35%-45% by age 70
|
|
|
What is a variant of Lynch Syndrome?
|
Muir-Torre Syndrome - associated with multiple sebaceous adenomas, sebaceous carcinomas, and keratoacanthomas
|
|
|
What is the average age of presentation of the different types of cancer associated with Lynch Syndrome?
|
- Colon ~44 years
- Endometrial ~46 years - Stomach ~56 years - Ovarian ~43 years |

