![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
12 Cards in this Set
- Front
- Back
|
what is boyle's law? |
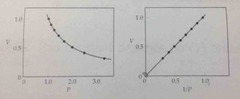
the volume of a fixed quantity of a gas is inversely proportional to the pressure |
|
|
what is Charle's law? |

the volume of a fixed quantity of gas is directly proportional to it's absolute temprature. |
|
|
what's avogadro's hypothesis? |

equal volumes of gases at the same temprature and pressure contain equal number of moles |
|
|
what is the ideal gas equation? |
PV = nRT |
|
|
what are the STP for the ideal gas equation? |
0 C 1atm |
|
|
density of a gas using the ideal gas law? |
ρ=PM/RT |
|
|
state Dalton's law? |
the total pressure of gases equals the sum of the partial pressures of the mixtures components. Pt=ΣPi Pt= nt(RT/V) mole fraction for component i = Xi = ni/nt |
|
|
what are the 4 postulates of the kinetic theory of gases? |
• Gases consist of large number of molecules that are in continuous random motion • the combined volume of all the molecules of the gases is negligible relative to the total volume of the container. • Attractive and repulsive forces between gas molecules are negligible • The average kinetic energy of molecules is proportional to the absolute temprature |
|
|
how do real gases differ from ideal gases? |
at lower pressures (below 10 atm) the deviation from the ideal behavior is small at higher tempratures the behavior of a real gas |
|
|
graph deviation of real from ideal against temp |
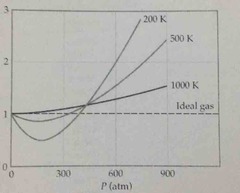
Back (Definition) |
|
|
graph deviation of real from ideal against pressure |
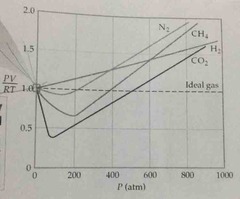
Back (Definition) |
|
|
what is the van der waal equation? |

Back (Definition) |

