![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
21 Cards in this Set
- Front
- Back
|
Unit 6 |
ch 9 |
|
|
How many lone pairs of electrons are on the As atom in AsCl3?
|
1 |
|
|
In the best Lewis structure for NO +, what is the formal charge on the N atom?
|
0 |
|
|
Which of the following processes are endothermic?
|
the reaction associated with the ionization energy of potassium.
|
|
|
Use the bond energies provided to estimate ΔH°rxn for the reaction below. |
-128 kJ |
|
|
Use Lewis theory to determine the chemical formula for the compound formed between Al and O.
|
Al2O3 |
|
|
Which of the following reactions is associated with the lattice energy of CaS (ΔH°latt)?
|
Ca2(g) + S2(g) → CaS(s) |
|
|
Place the following in order of decreasing magnitude of lattice energy. |
NaF > KCl > RbBr |
|
|
Choose the compound below that should have the highest melting point according to the ionic bonding model.
|
AlN |
|
|
Give the complete electronic configuration for S2-.
|
1s22s22p63s23p6
|
|
|
Identify the compound with metallic bonding.
|
Li |
|
|
Identify the shortest bond.
|
triple covalent bond
|
|
|
Place the following elements in order of increasing electronegativity. |
Sr < Na < N |
|
|
Which molecule or compound below contains a pure covalent bond?
|
Cl2 |
|
|
Choose the best Lewis structure for BeF2.
|
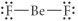
|
|
|
Give the number of valence electrons for ICl5.
|
42 |
|
|
Choose the best Lewis structure for SO42.
|
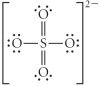
|
|
|
Draw the Lewis structure for CO32- including any valid resonance structures. Which of the following statements is TRUE?
|
The CO32- ion contains two C-O single bonds and one C=O double bond.
|
|
|
Draw the best Lewis structure for the free radical, NO2. What is the formal charge on the N?
|
+1 |
|
|
Which compound has the highest carbon-carbon bond strength?
|
HCCH |
|
|
Place the following in order of increasing bond length. |
NO < NO2 < NO3
|

