![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
49 Cards in this Set
- Front
- Back
|
Evidence Benzene has a delocalised structure |
1) Benzene all lengths are the of the single C-C bond and the double C=C bond is the same. 2) Hydrogenation of benzene is less exothermic than expected when compared to the kekule structure. As benzene is more thermodynamically stable. 3)Benzene is resistant to electrophilic attack. Goes under electrophilic substitution not addition. |
|
|
Nitration of benzene |
*Reagents: Concentrated HNO3 and H2SO4 *Conditions: 55 Celsius *Electrophile generation: HNO3 + H2SO4 ----> NO2+ +HSO4- + H2O *Regeneration of catalyst: H+ +HSO4- ------> H2SO4 |
|
|
Halogenation of benzene |
*Reagents: Cl2 / Br2 / I2 with (e.g) FeCl3/AlCl3
*Conditions: room temp & pressure *Electrophile generation: Cl2 + FeCl3 ----> FeCl4- + Cl+ *Regeneration of catalyst: H+ + FeCl4- ------> FeCl3 + HCl |
|
|
Bromination of Phenol |
- Observation: Orange to Colorless and White precipitate. - 3Br2 is needed and Br is attached to 2,4,6 position on the ring. + 3HBr |
|
|
Uses of Phenol |
1) Disinfectants 2) Production of plastic 3) Antiseptics |
|
|
Comparing Bromination of Cyclohexene, Benzene and Phenol |
[Benzene] Electrons are delocalised, this gives Benzene a lower electron density so it cannot polarise Br2 unless there is a halogen carrier. [Cyclohexene] have ㅈ electrons that are localised, giving a higher electron density to polarise Br2 [Phenol] A lone pair of electrons from the oxygen is partially delocalised into the ring, it has a higher electron density so it can polarise Br2 |
|
|
In Carbonyls why C=O bond does not react with electrophiles. |
The C=O is polarised as O is more electronegative than C. The ㅈ bond is unavailable to this polarisation. So electrophiles are repelled but nuclephiles are attracted to delta + Carbon. |
|
|
Reduction of Carbonyl Compounds |
* Type of reaction : Nucleophilic addition * Reagent: NaBH4 * Conditions: Aqueous * Nucleophile: H- 2[H] needed for every reducing |
|
|
Dectecting Carbonyl Compounds |
* Use 2,4 -DNPH * Observation: Orange precipitate 1) Filter and recrystallize the ppt 2) Measure melting point of ppt 3) Measure against a known vaule.
|
|
|
Distinguishing between Aldehydes and Ketones. |
* Use: Tollen's Reagent [Ag(NH3)2]+ * Aldehydes = Silver mirror is formed Ag+ (aq) + e- -------> Ag (s) RCHO (l) + [O] -------> RCOOH (l) * Ketones = No reaction |
|
|
Solubility of C.ACIDS & Esters. |
Acids are soluble in organic solvents, as they can form hydrogen bonding. As mass increases, the solubility decreases. |
|
|
Uses of esters. |
1) Flavours 2) Perfume 3) Solvents |
|
|
Alcohol + Carboxylic Acid |
=Ester Concentrated H2SO4. Heat under reflux, |
|
|
Acid anhyrides & alcohols / phenol |
= Ester Conditions: Gently Warm |
|
|
Hydrolysis of Esters |
1) Hot dilute aqueous HCl
2) Hot aqueous NaOH |
|
|
Glycerol / Propane-1,2,3-triol |
draw |
|
|
Fatty acids - Saturated / Unsaturated |
* Saturated straight chain. Can pack closely together, many points of contact + inter molecular forces = solid at rtp. BAD. * Mono-nsaturated: one C=C bond, liquids at rtp. * Trans fatty acids may increase cholesterol leading to increased risk of corony heart disease and strokes. |
|
|
Making Biodiesel |
Triglyceride + 3Alcohol -------> 3Biodiesel + glycerol. * Conditions : NaOH catalyst + heat * Alcohol is added in excess to shift the equilibrium to the right * Methanol or ethanol is suitable alcohol. |
|
|
Formation of amine |
*Reagents: Excess Ethanolic Ammonia
(In excess to prevet further subsitution) & (Ethanol is a weaker nucleophile than water, excellent solvent) *Conditions: Heat under reflux *Nucleophile: NH3 CH3CH2Cl + 2NH3 ----> CH3CH2NH2 +NH4CL *Type of reaction: Nucleophilic substitution |
|
|
C6H5NO2 + 6[H] -------> C6H5NH2 + 2H20
Reduction of Nitrobenzene |
* Reagents: Tin & Concentrated HCL. * Conditions: Heat Under Reflux. * Type of reaction: Reduction 6[H] for every NO2 & 2 H2O |
|
|
Azo dye sythesis. (Formation of diazonium salt) |
* Reagents: HNO2 + HCL * Conditions: below 10 Celsius |
|
|
Coupling with phenols. |
* Reagents:Phenol + NaOH
* Conditions: Alkaline, below 10 Celsius Use: dyes |
|
|
What modern day technology has disproved kekule's theory. |
X-ray diffraction, it showed all the benzene lengths are the same |
|
|
Alkylation |
Benzene + RCl -----> C6H5R + HCl |
|
|
a-amino acid definition |
The -NH2 and the -COOH group is attached to the same carbon atom |
|
|
Amino acid general formula |
RCH(NH2)COOH |
|
|
Zwitterion definition |
An ion that has both a positive and a negative charge |
|
|
Isoelectric point definition |
The pH at which the zwitterion exsits |
|
|
Isoelectric point which group exists |
1) More alkaline (pH increased) O- 2) More acidic (pH decreased) NH3+ |
|
|
Z |
CIS (on same side) |
|
|
E
|
Trans (on different sides) |
|
|
ACID Hydrolysis of peptides |
*Conditions : Hot Aqueous HCL, under reflux NH3+ |
|
|
ALKALINE Hydrolysis of peptides |
*Conditions : Hot Aqueous NaOH, under reflux
O-Na+ |
|
|
Optical isomers |
Non-superimposable mirror images with 4 different groups attached to the central chiral carbon atom. Each chiral carbon gives 2 non-superimposable mirror images. |
|
|
Condensation polymerisation |
Two different monomers that join together and a small molecule is given off as a side product such as H20 or HCL |
|
|
Addition polymerisation |
2 or more monomers join together to form a polymer, no other product is formed |
|
|
Hydrolysis of polymers |
1) C=O bond in condensation polymers make it photodegradable
2) Mirco-organisms can break it down (biodegradable) 3) ester or amide links can be hydrolysed |
|
|
Absorbption (Solid stationary phase) |
Solids are held on the stationary phase depending on the differences in polarity |
|
|
Relative solubility (Liquid stationary phase) |
Solutes more between stationary phase and mobile phase, based on how much they dissolve. |
|
|
Rf |
distance travelled by solute (y) --------------------------------------------------(divide) distance travelled by solvent (x) |
|
|
Nylon-6,6 |
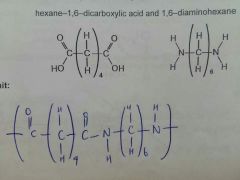
|
|
|
Kevlar |
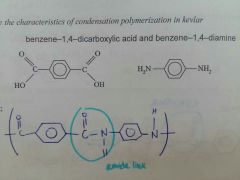
|
|
|
Nylon-6,6 (using acyl chlorides) |
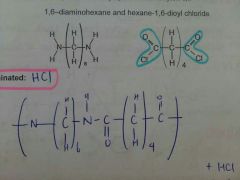
|
|
|
Retention time |
Time taken from injection to dectection. • Retention time depends on: 1) boiling point (high = long) 2) solubility in the liquid phase (greater = long) |
|
|
HNMR region on the electromagnetic spectrum |
Low energy radiowaves |
|
|
Ad/dis of sythesis single optical isomer |
1) side effects are eliminated 2) improves pharmlogical activity. •Done by using enzymes or bacteria to promote steroselectivity. •Natural chiral molecules. |
|
|
Chromatography |

|
|
|
TLC |

|
|
|
TLC |
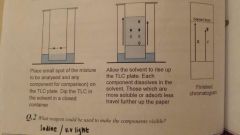
|

