![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
158 Cards in this Set
- Front
- Back
|
The early atmosphere
1. What produced the gases that formed the atmosphere? 2. What were the two main gasses in the early atmosphere? 3. How did the oceans form? 4. Where does our evidence about the early atmosphere come from? |
1. Volcanoes
2. Carbon dioxide and water vapour 3. As the Earth cooled down, water vapour in the atmosphere condensed to form liquid water. 4. We can look at modern volcanoes, and at the atmospheres of other planets like Mars and Venus (because they also have volcanoes). |
|
|
Acid rain |
Rain that is more acidic than normal, due to sulfur dioxide and nitrogen oxides dissolved in it. |
|
|
Alkali |
A soluble base |
|
|
Alkane |
A hydrocarbon with only single bonds between two carbon atoms. |
|
|
Alkene |
A hydrocarbon with at least one double bond between carbon atoms. |
|
|
Alloy |
A mixture of more than one metals. OR A mixture containing at least one metal and carbon. |
|
|
Alloy steel |
An alloy of iron with other metals. |
|
|
Antacid |
A compound that is used to neutralise acid in the stomach. |
|
|
Aqueous solution [higher] |
Mixture formed when a substance dissolves in water. |
|
|
Atmosphere |
Layer of gases that surrounds the Earth. |
|
|
Atom |
The smallest part of an element that can take part in chemical reactions. |
|
|
Balanced equation [higher] |
Description of a reaction using the symbols and formulae of the reactants and products. |
|
|
Base |
A substance that will react with an acid to form only salt and water. Common bases: Metal oxides, e.g. Copper oxide Metal hydroxides, e.g. potassium hydroxide Metal carbonates, e.g. calcium carbonate |
|
|
Biodiesel |
Diesel fuel made from plant material. |
|
|
Biofuel |
Any fuel made by humans from animal or plant materials that have recently died.
These are different to fossil fuels, which take millions of years to form. |
|
|
Biogas |
Methane made from animal manure or biodegradable waste from homes and farms. |
|
|
Bitumen |
The fraction of crude oil with the longest molecules. It is used for making roads and roofs. |
|
|
Boiling point |
The temperature at which all of a substance starts to turn into a gas (evaporate).
This can also be described as the condensing point - where the gas will turn back into a liquid. |
|
|
Bonds |
Forces of attraction holding atoms together in a molecule. |
|
|
Bromine test |
A test for unsaturation. Bromine water turns from reddish-brown to colourless when mixed with an unsaturated molecule.
This happens because the double bonds (in an alkene) will open up to bond with the bromine atoms. |
|
|
Bromine water |
A solution of bromine in water that turns colourless when mixed with an unsaturated molecule.
It changes from reddish-brown to colourless because the double bond will open up to bond with bromine atoms. |
|
|
Carats [higher] |
A measure of the purity of gold alloys.
Pure gold is 24 carats (it contains no other metals and is a pure metal rather than an alloy).
50% gold would be 12 carats. |
|
|
Carbon cycle |
A sequence of processes by which carbon moves from the atmosphere, through living and dead organisms, into sediments and into the atmosphere again. |
|
|
Carbon neutral |
A fuel or process that does not add any carbon dioxide to the atmosphere overall. |
|
|
Cement |
Material made by heating limestone with clay. |
|
|
Chalk |
Sedimentary rock, mostly calcium carbonate, made from the remains of marine micro-organisms. |
|
|
Chemical formula |
A combination of symbols and numbers that shows how many atoms of different kinds are in a particular molecule. In compounds that do not form molecules, it shows the ratio of elements in the compound. |
|
|
Chemical potential energy |
The energy stored in matter, which can be released by a chemical reaction (such as combustion). |
|
|
Climate change |
Changes to the Earth's climate, or changes in weather patterns on a global scale. |
|
|
Combustion |
Chemical reaction when substances burn, combining with oxygen to produce heat and waste products such as carbon dioxide. |
|
|
Complete combustion |
Combustion of hydrocarbons with enough oxygen present to convert all the fuel into carbon dioxide and water. |
|
|
Compound |
A substance containing two or more elements chemically joined together. |
|
|
Concentration |
The amount of a substance dissolved in a certain quantity of liquid.
E.g. g/cm3, mg/100 cm3). |
|
|
Concrete |
Material made by mixing cement with sand, stones and water. |
|
|
Control |
An experiment that uses the same apparatus as the main part of the experiment but the variable that is being changed is not applied.
The point is to give you a comparison with the real experiment, to check that the independent variable really is causing a change in the dependent variable. |
|
|
Convection current |
A current caused by parts of a fluid being at a different temperature and so a different density to the rest of the fluid. |
|
|
Convention |
A standard way of doing something. |
|
|
Core |
The middle of the Earth.
This is mainly iron, very hot and solid. Despite the fact that its temperature is ~5700 K, it is solid because the pressure is so high - this keeps it compressed into a solid. |
|
|
Corrosion |
Occurs when a metal is converted to its oxide by the action of moist air. |
|
|
Cracking |
Splitting up long hydrocarbon molecules into shorter ones. |
|
|
Criterion |
Rule or test that can be used to judge how to make a fair decision (plural: criteria). |
|
|
Crude oil |
A mixture of hydrocarbons formed from dead microscopic organisms by heat and pressure over millions of years. |
|
|
Crust |
The outer surface of the Earth. |
|
|
Crystal |
A solid in which the particles are arranged in an ordered pattern. |
|
|
Deforestation |
The destruction of the world's trees and forests.
This can contribute to global warming because destroying trees means there are less to remove carbon dioxide and produce oxygen; this leads to an overall increase in the amount of carbon dioxide in the atmosphere. |
|
|
Denitrifying bacteria [higher] |
Bacteria that break down more complex nitrogen compounds into simpler ones, such as nitrates to nitrites, or nitrites to ammonia. |
|
|
Dense |
Something that has a lot of mass in a given volume. |
|
|
Density |
The mass of a substance per unit volume; the unit is usually g/cm3. |
|
|
Diesel oil |
Fraction of crude oil used as a fuel in lorries and some cars. |
|
|
Double bond |
When two bonds join a pair of the same atoms (usually two carbon atoms). |
|
|
Ductile |
Can be stretched into wires.
E.g. Copper metal is ductile - this property means that it can be pulled (drawn) into thin wires. |
|
|
Earthquake |
A sudden movement within the Earth, releasing a large amount of stored energy. |
|
|
Electrode |
Part of an electrical circuit that makes contact with a sample. |
|
|
Electrolysis |
Passing a direct electrical current through an electrolyte to decompose it.
E.g. Hydrochloric acid (the electrolyte) can be decomposed by electrolysis to produce hydrogen and chlorine gasses. |
|
|
Electrolyte |
A liquid which conducts the electric current and is decomposed by it. |
|
|
Element |
Substance that cannot be split up into simpler substances. |
|
|
Epicentre |
The point on the surface of the Earth directly above the focus of an earthquake. |
|
|
Erosion |
Removal or transportation of broken up rocks. |
|
|
Ethanol |
An alcohol, made by processing sugar beet or sugar cane. |
|
|
Extraction (metal) |
Reaction in which a metal is produced from a compound in an ore.
Unreactive metals are found as pure metals, so do not need extracting. Slightly reactive metals can be extracted by heating with carbon. Ver reactive metals have to be extracted using electrolysis. |
|
|
Fineness [higher] |
A measure of the purity of gold in parts per thousand with pure gold having a fineness of 1000. |
|
|
Focus |
The place where an earthquake begins, usually under the surface. |
|
|
Formula (chemical) |
The composition of a substance showing the symbols of the elements it contains, and the ratios in which their atoms are present. |
|
|
Fossil |
The remains or trace evidence of prehistoric living organisms. |
|
|
Fossil fuel |
Non-renewable fuels such as coal, oil and natural gas that have formed over millions of years from dead plants and animals. |
|
|
Fractional distillation |
The process by which a mixture of two or more liquids is separated, for example crude oil is separated into different fractions. |
|
|
Fractions |
The different mixtures into which crude oil is separated. |
|
|
Fuel cell |
A device which produces electricity from hydrogen and oxygen without burning. |
|
|
Fuel oil |
Crude oil fraction used as fuel for ships and for lubrication. |
|
|
Global warming |
The increase in the Earth's average temperature likely to be caused by increased amounts of carbon dioxide in the atmosphere. |
|
|
Granite |
Igneous rock with relatively large crystals, formed when magma cools and solidifies slowly. |
|
|
Greenhouse effect |
When gases in the atmosphere trap heat energy and keep the Earth warm. |
|
|
Greenhouse gases |
Gases that help to trap heat in the atmosphere. They include carbon dioxide, methane and water vapour. |
|
|
Hazard symbol |
A symbol used on containers that warns people about the dangers of the contents. |
|
|
Hazardous |
Something that could be harmful or dangerous if not used and stored properly. |
|
|
Hyrdocarbon |
A compound containing only hydrogen and carbon atoms. |
|
|
Igneous rock |
Rock formed when magma cools and solidifies. |
|
|
Ignite |
To start burning |
|
|
Incomplete combustion |
Combustion that occurs without enough oxygen to completely oxidise all the fuel. Incomplete combustion of hydrocarbons produces carbon dioxide, carbon monoxide and carbon (soot). |
|
|
Insoluble |
Substance which does not dissolve in a given solvent. |
|
|
Ion |
An atom that has become electrically charged. Atoms become ions when they gain or lose electrons. |
|
|
Iron seeding |
Adding iron compounds to the oceans to encourage organisms to grow and remove carbon from the carbon cycle. |
|
|
Kerosene |
Crude oil fraction used as a fuel for jet engines. |
|
|
Lava |
Molten rock on the Earth's surface. |
|
|
Limestone |
Sedimentary rock, mostly calcium carbonate, made from the remains of marine organisms. |
|
|
Limewater |
A solution of calcium hydroxide that turns milky when carbon dioxide is bubbled through it. |
|
|
LItmus paper |
Blue litmus paper turns red in acid solutions.
Red litmus paper turns blue in alkaline solutions. |
|
|
Magma |
Molten rock inside the Earth's crust. |
|
|
Malleable |
Can be hammered into shape. |
|
|
Mantle |
The part of the Earth between the crust and the core. |
|
|
Marble |
Metamorphic rock, mostly calcium carbonate, formed from limestone or chalk. |
|
|
MIxture |
A substance containing two or more different substances that are not joined together. |
|
|
Model |
An example of something happening, which explains how a scientific idea should be understood. |
|
|
Molecule |
Two or more atoms joined together. |
|
|
Monomer |
Small molecule used to make a polymer. |
|
|
Neutralisation reaction |
Reaction in which a base of alkali reacts with an acid. |
|
|
Nitinol |
An alloy of nickel and titanium, which is a shape memory alloy. |
|
|
Nitrifying bacteria |
Bacteria that make more complex nitrogen compounds from simpler ones, such as nitrates from nitrites, or nitrites from ammonia. |
|
|
Nitrogen cycle |
A sequence of processes by which nitrogen moves from the atmosphere through living and dead organisms, into the soil and back to the atmosphere. |
|
|
Nitrogen-fixing bacteria |
Bacteria that can take nitrogen from the atmosphere and convert it to more complex nitrogen compounds, such as ammonia. |
|
|
Noble gases |
Unreactive gases in Group 0 of the periodic table. |
|
|
Non-renewable |
Any energy resource that will run out one day. |
|
|
Non-renewable resources |
Resources that cannot be replaced once they have been used. Non-renewable resources will eventually run out.
E.g. Fossil fuels - coal, oil and natural gas. |
|
|
Nucleus |
The central part of an atom, containing protons and neutrons. |
|
|
Ore |
Rock from which a metal can be extracted for profit. |
|
|
Oxidation |
Occurs when oxygen is added to an element or compound. |
|
|
Permeable |
A solid that allows a liquid or a gas to pass through it is described as being permeable. |
|
|
Petrol |
Crude oil fraction used as fuel for cars. |
|
|
pH scale |
A scale from 1 to 14 showing acidity or alkalinity. Numbers below 7 are acids. Numbers above 7 are alkalis. pH 7 is neutral. |
|
|
Photosynthesis |
Set of chemical reactions in plants that allow them to produce their own food (glucose) using water and carbon dioxide and releasing oxygen as a waste product. The process is powered by light from the Sun. |
|
|
Plastic |
The common name for many polymers. |
|
|
Pollutant |
A substance that harms living organisms when released into the environment, often waste products of human activity. |
|
|
Poly(chloroethene) |
A polymer made from ethene monomers. |
|
|
Poly(ethene) |
A polymer made from ethene monomers. |
|
|
Poly(propene) |
A polymer made from propene monomers. |
|
|
Poly(tetrafluoroethene) |
The chemical name for PTFE, which is also known as Teflon. |
|
|
Polymer |
A long molecule made by joining many smaller molecules (monomers) together. |
|
|
Polymerisation |
The process of making a polymer. |
|
|
Population growth |
Increase in population size over time. |
|
|
Precipitate |
Insoluble product formed in a precipitation reaction. |
|
|
Precipitation reaction |
Reaction in which an insoluble product is formed from soluble reactants. |
|
|
Primary data |
Data that you collect yourself. |
|
|
Product |
Substance formed by a chemical reaction. |
|
|
PVC |
Another name for poly(chloroethene). |
|
|
Quarry |
Shallow mine on the Earth's surface from where rocks are removed. |
|
|
Rate |
How quickly something happens. |
|
|
Reactant |
Substance used up in a chemical reaction. |
|
|
Reactivity series |
A list of metals arranged in order of reactivity. |
|
|
Recycle |
To extract a material from waste so that it can be reused or processed to make a new object or material. |
|
|
Recycling |
Taking materials from waste and making them into useful products again. |
|
|
Reduction |
Occurs when oxygen is removed from a compound. |
|
|
Renewable |
Any energy resource that will not run out. |
|
|
Renewable energy resources |
Resources that will not run out, such as solar or wind energy. |
|
|
Rusting |
The corrosion of iron. |
|
|
Salt |
A compound formed by neutralisation of an acid by a base. The first part of the name comes from the metal in the metal oxide, hydroxide or carbonate. The second part of the name comes from the acid. |
|
|
Saturated |
A molecule with only single bonds between the carbon atoms. |
|
|
Secondary data |
Data that you use from someone else's report, book, TV programme etc.. |
|
|
Sediment |
Process by which solid material that settles to the bottom of a fluid, such as water. |
|
|
Sedimentary rock |
Rock made by compression of layers of solid material (sediment) that settles to the bottom of a fluid, such as water.
Sedimentary rocks normally form at the bottom of the ocean. |
|
|
Shape memory alloy [higher] |
An alloy that returns to its original shape with a change in conditions, often temperature. |
|
|
Smart material |
A material with a property that changes with a change in conditions. |
|
|
Solidify |
To become solid. |
|
|
Soluble |
Substance which will dissolve in a given solvent. |
|
|
Soot |
Tiny particles of solid carbon produced by incomplete combustion. |
|
|
Stable |
Something that stays the same, without changing. |
|
|
State symbol [higher] |
Letter or letters to show the state of a substance. |
|
|
Tar |
Sticky black substance in tobacco smoke that contains carcinogens. |
|
|
Thermal decomposition |
Reaction in which one substance breaks down when heated to form two or more new substances. |
|
|
Thermal energy |
Energy transferred by heating. |
|
|
Trace |
A tiny amount of something. |
|
|
Transfer |
To move something. |
|
|
Universal indicator |
A mixture of different indicators giving a different colour at different points on the pH scale. |
|
|
Unsaturated |
A molecule with at least one double bond between carbon atoms. |
|
|
Viscosity |
How thick or runny a liquid is. Low viscosity is very runny, high viscosity is thick. |
|
|
Word equation |
Description of a reaction using the names of the reactants and products. |
|
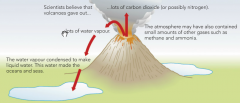
The Earth's first atmosphere was formed by gases produced from volcanoes. We think these gases were mainly carbon dioxide and water vapour. |
The Earth was very hot to start with, and there were no oceans. As the earth cooled down the water vapour in the atmosphere condensed to form liquid water. This liquid water became the oceans. Explain why scientists cannot be certain about the composition of the early atmosphere? There were no humans around to measure the atmosphere, so they have to use clues. There are different sources of information, but not all the evidence leads to the same conclusion. This means that it is difficult to know exactly what the early atmosphere was like and how it has changed. |
|
|
The early atmosphere 1. What produced the gases that formed the atmosphere? 2. What were the two main gasses in the early atmosphere? 3. How did the oceans form? 4. Where does our evidence about the early atmosphere come from |
1. Volcanoes 2. Carbon dioxide and water vapour 3. As the Earth cooled down, water vapour in the atmosphere condensed to form liquid water. 4. We can look at modern volcanoes, and at the atmospheres of other planets like Mars and Venus (because they also have volcanoes). |
|
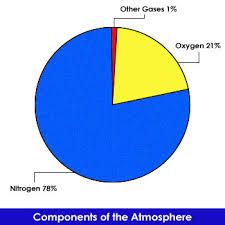
The amounts of different gases in the atmosphere are shown in the pie chart and table. The atmosphere also contains water vapour, but this is not usually included because the amount changes depending on the weather. |
The composition of the atmosphere today is not constant. It can be changed by human activities. Burning fossil fuels releases carbon dioxide into the atmosphere. Sulphur dioxide can also be released from burning coal. |

