![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
45 Cards in this Set
- Front
- Back
|
1. What is the subcellular location of cholesterol?
|
a. Cytosol
b. ER |
|
|
2. What is the substrate for cholesterol synthesis?
|
a. Acetyl-CoA
|
|
|
3. What reduces HMG-CoA to mevalonate?
|
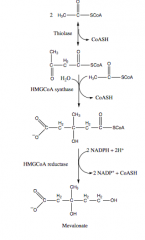
a. HMG-CoA reductase
|
|
|
4. What is the importance of HMG-CoA reductase?
|
a. It is the major control point for cholesterol synthesis
b. Inhibited by statins |
|
|
5. What is the location of HMG-CoA reductase?
|
a. ER membrane
|
|
|
6. How is isopentenyl diphosphate formed from mevalonate?
|
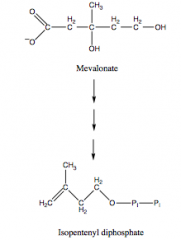
a. Sequential phosphorylation by 3 kinases
b. Decarboxylation |
|
|
7. How is squalene formed from geranyl diphosphate?
|
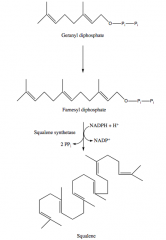
a. 2 isoprenoid units are used to form geranyl diphosphate
b. A third isoprenoid unit is added to form farnesyl diphosphate c. Two farnesyl diphsophates are used to synthesize squalene |
|
|
8. How is lanosterol formed from squalene?
|
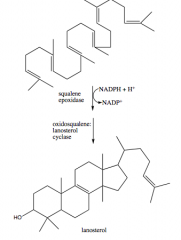
a. Squalene epoxidase requires NADPH and O2
b. Lanosterol represents the first sterol produced in the pathway to cholesterol |
|
|
9. How is cholesterol formed from lanosterol?
|
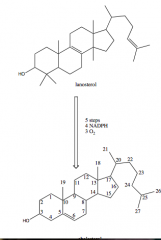
a. Removal of methyl group at C14
b. Removal of two methyl groups at C4 c. Reduction of the C24 double bond d. Moving the delta 8,9 bond to delta 5,6 |
|
|
10. Where does the conversion from lanosterol to cholesterol occur?
|
a. ER
|
|
|
11. How are prenylated proteins formed?
|
a. GTP binding proteins are prenylated with either farnesyl or geranygeranyl residues
b. Residues may anchor protein to the membrane |
|
|
12. How is dolichol formed?
|
a. Formed from farnesyl diphosphate plus up to 16 more isopentenyl residues
|
|
|
13. What is the importance of dolichol?
|
a. Required for synthesis of N-linked glycoproteins
|
|
|
14. How is ubiquinone formed?
|
a. From farnesyl disphosphate plus 3-7 isopentenyl residues
|
|
|
15. What is the importance of ubiquinone?
|
a. Required for ETC
|
|
|
16. What is ACAT? What does it catalyze?
|
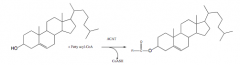
a. Acyl CoA-cholesterol acyltransferase
b. Formation of cholesteryl ester |
|
|
17. What happens to cholesteryl ester once it is formed?
|
a. Packaged into VLDL
b. Stored into liver for future use |
|
|
18. What is the use of cholesteryl esters once they are transferred to other tissues?
|
a. Steroid hormone synthesis
b. Vitamin D synthesis |
|
|
19. Where does cholesterol from the liver go?
|
a. May be secreted into bile
b. May be synthesized to bile acids and their salts |
|
|
20. How is 7α-hydroxycholesterol formed from cholesterol?
|
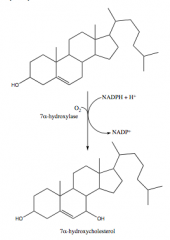
a. 7α-hydroxylase as catalyst
b. Requires NADPH, O2, CP450, vitamin C |
|
|
21. What is the committed step of bile acid synthesis?
|
a. Cholesterol to 7α-hydroxycholesterol
|
|
|
22. How are cholic acid and chenodeoxycholic acid formed from 7α-hydroxycholesterol?
|
a. 12α-hydroxylase as catalyst
b. Propionyl-CoA obtained from carbons removed to form acids c. Require NADPH, O2, and CoA |
|
|
23. Which predominates in humans, cholic acid or chenodeoxycholic acid?
|
a. Cholic acid
|
|
|
24. What happens to cholic acid and chenodeoxycholic acid once they are formed?
|
a. They are conjugated with glycine or taurine
|
|
|
25. What happens once cholic acid and chenodeoxycholic acid are conjugated?
|
a. Four primary bile acids are formed
|
|
|
26. What are the four primary bile acids?
|
a. Taurocholic acid
b. Glycocholic acid c. Taurochenodeoxycholic acid d. Glycochenodeoxycholic acid |
|
|
27. What does the formation of the primary bile acids require?
|
a. Cholyl-CoA
b. Chenodeoxycholyl-CoA |
|
|
28. What happens to the primary bile acids once they are formed?
|
a. They are secreted into the liver with cholesterol
|
|
|
29. How are the primary bile acids metabolized by intestinal bacteria? What results?
|
a. Deconjugation, 7α-dehydroxylation
b. Secondary bile acids |
|
|
30. What are the secondary bile acids?
|
a. Deoxycholic acid
b. Lithocholic acid |
|
|
31. Where are both primary and secondary bile acids reabsorbed? What happens to them thereafter?
|
a. Ileum
b. Returned to liver to become conjugated and secreted→ enterohepatic circulation |
|
|
32. What controls whole-body cholesterol metabolism?
|
a. Liver
|
|
|
33. How does the liver receive excess cholesterol from body tissues?
|
a. LDL receptors
b. Reverse cholesterol transport |
|
|
34. What are the major regulatory targets of the liver to regulated cholesterol metabolism?
|
a. HMG-CoA reductase→ major point
b. LDL receptor c. 7α-hydroxylase |
|
|
35. What stimulates transcriptional control of HMG-CoA reductase?
|
a. Binding of SREBP to SRE of HMG-CoA reductase
|
|
|
36. What do elevated levels of cholesterol in the cell do to the SREBP/SRE interaction?
|
a. Prevents SREBP from binding to SRE
|
|
|
37. How does SREBP overcome high cholesterol levels in the cell?
|
a. SREBP binds to SCAP in the ER membrane
b. SCAP/SREBP complex kept in ER by cholesterol bond to SCAP c. Cholesterol levels drop, cholesterol leaves the SCAP binding site d. SREBP/SCAP moves to the Golgi e. SREBP goes through proteolysis, N-terminal SREBP free to travel to bind the SRE |
|
|
38. What happens to HMG-CoA reductase when bound to cholesterol?
|
a. More susceptible to proteolysis
|
|
|
39. What is the effect of phosphorylation on HMG-CoA reductase?
|
a. Less active
|
|
|
40. What factors promote phosphorylation of HMG-CoA reductase?
|
a. Glucagon
b. Glucocorticoids c. Cholesterol |
|
|
41. What factors promote dephosphorylation of HMG-CoA reductase?
|
a. Insulin
b. Thyroid hormone |
|
|
42. What regulates the number of LDL receptors?
|
a. Intracellular cholesterol content
b. By same mechanism as SREBP |
|
|
43. What will increased bile acids in the liver repress? How?
|
a. 7α-hydroxylase
b. Bile acids bind the farnesyl X receptor c. FXR/bile acid complex suppresses 7α-hydroxylase |
|
|
44. What are the components of cholesterol stones?
|
a. Cholesterol
b. Ca salts c. Bile pigments d. Proteins e. FA |
|
|
45. What are the components of pigment stones?
|
a. Calcium bilirubinate
b. <20% cholesterol |

