![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
38 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Sodium with H2O |
NaOH + H2 |
|
|
|
Mg with Steam |
MgO + H2 ( white powered ) |
|
|
|
Mg with water |
Mg(OH)2 + H2 |
|
|
|
Na with O2 obeservation |
Na2O orange flame with white power |
|
|
|
Mg + O2 O.O |
White solid white flame |
|
|
|
Al +O2 |
Al2O3 white flame white solid power, stong oxide layer |
|
|
|
Si + O2 |
SiO2 glassy solid |
|
|
|
P with oxygen |
P4O10 spontaneously white smoke and powder brillant white flame |
|
|
|
SO2 with oxygen |
blue flame , pungent smell |
|
|
|
Period three oxides with water |

|
|
|
|
Mgo with hcl |
Mgcl2 and h20 |
|
|
|
Al203 with hcl |
Alcl3 and h20 |
|
|
|
Al203 with Na0H solution ( water) |
NaAl(H20)2(OH)4 and water |
|
|
|
SiO2 with NaOH |
Na2sio3 and water |
|
|
|
So2 and NaOh |
NaSO3 and h2o |
|
|
|
Na2so3 + SO2 + H2O |
NaHSO3 |
|
|
|
H3PO4 with NaOH |
NaH2PO4 AND WATER |
|
|
|
(H20)6 colours complex |
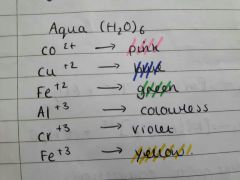
|
|
|
|
NH3 and OH` not excess colours |

|
|
|
|
Metal +2 excess NH3, structures and colours |

|
|
|
|
M+2 excess OH colour and structures |
Same as OH normal , otherwise M+2 doesnt go to completuon |
|
|
|
M+3 NH3 colours and structure |

Only one metal |
|
|
|
M+3 excess OH- colour and structure |

|
|
|
|
What are M+3 further reactes with not in excess NH3 |
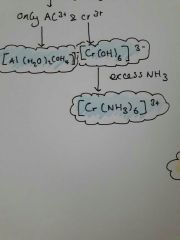
Al is colourless and cr is green solution |
|
|
|
The Cr triangle of colours and reactions |
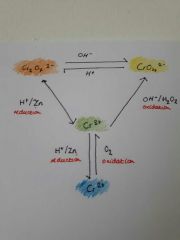
|
|
|
|
Co colours map |
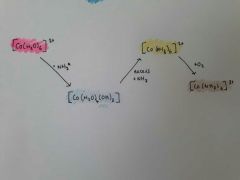
|
|
|
|
Contract process with steps |

|
|
|
|
Haber process with cataylst |
Haber provess with cataylst |

|
|
|
Manufactor of methanl with catatylst |

|
|
|
|
S2O8- with I- reaction with catayst |

|
|
|
|
MnO4- with C2O4 ^2- |

|
|
|
|
Reaction of MnO4 with Fe 2+ |

|
|
|
|
Cr2O7 with Fe |
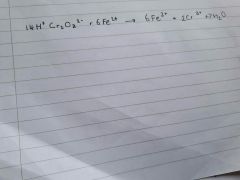
|
|
|
|
Cr2O7 with Zn |

|
|
|
|
Cr2O7 with OH and H+ equilibrium |

|
|
|
|
So2 with NaOH in excess |
Na2so3 + h2o |
|
|
|
So2 with naoh not in excess |
Only NaHso3 |
|
|
|
S03 with Na2So4 |
Na2SO4 |
|

