![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
32 Cards in this Set
- Front
- Back
|
JOHN DALTON MODEL |
- PROPOSED THAT AT THE ROOT OF ALL MATTER WERE VERY SMALL PARTICLES CALLED ATOMS. - DEVELOPED THE PARTICLE MODEL. |
|
|
J.J THOMSON |
- DISCOVERED THE ELECTRON IN 1897 - DEVELOPED THE PLUM PUDDING MODEL |
|
|
NEILS BOHR |
- PROPOSED THAT ELECTRONS GO IN FIXED ORBITS CALLED SHELLS - DEVELOPED THE ELECTRON SHELL MODEL |
|
|
EWIN SCHRODINGER |
-PROPOSED THAT ELECTRONS SHOULD BE REGARDED AS HAVING WAVE-LIKE PROPERTIES.
-QUANTUM MECHANICAL MODEL |
|
|
ERNEST RUTHERFORD |
-FIRED ALPHA PARTICELS AT A SHEET OF GOLD FOIL AND MOST PASSED STRAIGHT THROGH AND A FEW BOUNCED BACK - PROPOSED THAT THE ATOM IS MOSTLY EMPTY SPACE WITH A DENSE NUCLEUS AT THE CENRE. |
|
|
FREDERICK SODDY |
-PROPOSED THAT ATOMS OF THE SAME ELEMENT COULD HAVE DIFFERENT MASSES (ISOTOPES) |
|
|
JAMES CHADWICK |
- DISCOVERED THE NEUTRON. |
|
|
LETTERS THAT REPRESENTS SHELLS |
S,P,D,F |
|
|
GROUND STATE CONFIGURATION |
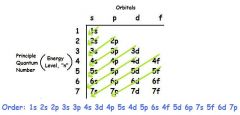
|
|
|
CATION |
-POSITIVE ION - LOSES ELELECTRONS -GROUP 1 ELEMENTS |
|
|
ANION |
-NEGATIVE ION - GAINS ELECTRONS -GROUP 17 ELEMENTS |
|
|
Transuranium elements |
Elements after uranium Artificially synthesised Radioactive |
|
|
Transuranium elements |
Elements after uranium Artificially synthesised Radioactive |
|
|
The last few elements on the periodic table are... |
EXTREMELY UNSTABLE |
|
|
Transuranium elements |
Elements after uranium Artificially synthesised Radioactive |
|
|
The last few elements on the periodic table are... |
EXTREMELY UNSTABLE |
|
|
Elements of the f block |
Lanthanoids and actinoids |
|
|
Transuranium elements |
Elements after uranium Artificially synthesised Radioactive |
|
|
The last few elements on the periodic table are... |
EXTREMELY UNSTABLE |
|
|
Elements of the f block |
Lanthanoids and actinoids |
|
|
Atomic size |
Increases: down a group Decreases: across a period |
|
|
Transuranium elements |
Elements after uranium Artificially synthesised Radioactive |
|
|
The last few elements on the periodic table are... |
EXTREMELY UNSTABLE |
|
|
Elements of the f block |
Lanthanoids and actinoids |
|
|
Atomic size |
Increases: down a group Decreases: across a period |
|
|
Electronegativity |
Increases:across a period Decreases: down a group |
|
|
Metallic character |
Increases: down a group Decreases: across a period |
|
|
Metallic character |
Increases: down a group Decreases: across a period |
|
|
Oxidising strength |
Increases:across a period Decreases:down a group |
|
|
Metallic character |
Increases: down a group Decreases: across a period |
|
|
Oxidising strength |
Increases:across a period Decreases:down a group |
|
|
Reducing strength |
Increases:down a group Decreases:across a period |

