![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
236 Cards in this Set
- Front
- Back
|
Matter |
Anything that occupies space |
|
|
Energy |
The ability to do work |
|
|
The two types of energy |
Kinetic energy Potential energy |
|
|
Kinetic energy |
Getting up to the top of the roller coaster Pulling back the rubber band |
|
|
Potential energy |
Going down the roller coaster Letting the rubber and go |
|
|
Four types of potential energy |
Chemical Electrical Mechanical Radiant |
|
|
Chemical energy |
The sun |
|
|
Electrical energy |
Wall outlet |
|
|
Mechanical energy |
Walking |
|
|
Radiant energy |
The sun |
|
|
Elements |
Fundamental units of matter Everything the world is made up of (Example: oxygen) |
|
|
What percentage of our body is made up of four elements |
96% |
|
|
What are the four elements that make up the human body |
Carbon (C) Oxygen (O) Hydrogen (H) Nitrogen (N) |
|
|
What is an atom |
The building block of an element |
|
|
What atoms are contained in a nucleus |
Protons (p+) Neutrons (n0) |
|
|
Is a proton positive, negative or neutral |
Positive |
|
|
Is a neutron positive, negative or neutral |
Neutral |
|
|
Where are electrons located |
Outside of the nucleus |
|
|
Are electrons positive, negative or neutral |
Negative (e-) |
|
|
What makes up a neutral atom |
An equal number of protons and electrons |
|
|
Atomic number |
Equal to the number of protons that the atoms contain |
|
|
True or false All atoms of the same element have the same atomic number |
True |
|
|
True or false All atoms of the same element have different atomic numbers |
False |
|
|
Atomic mass number |
Sum of the protons and neutrons |
|
|
Isotopes have the same number of... |
Protons |
|
|
Isotopes vary in number of ... |
Neutrons |
|
|
Give an example of an isotope for C12 |
C14 |
|
|
What device measures atomic weight |
A geiger counter |
|
|
Atomic weight |
Close to mass number of most abundant isotope |
|
|
What is the atomic weight of C12, C12, C12, C14, C14, C13 and why |
C12, because even though there are six isotopes, C12, is the one that occurs the most
|
|
|
Radioactivity |
The process of spontaneous atomic decay |
|
|
Choose one of the underlined words Radioisotopes tend to be stable or unstable |
Unstable |
|
|
True or false Radioisotopes are heavy isotopes |
True |
|
|
What do radio isotopes do |
Decompose to more stable isoptope |
|
|
Molecule |
Two or more atoms combined chemically (Example: H + H= H2) |
|
|
Compound |
Two or more different atoms combined chemically (Example: H + H + O= H2O) |
|
|
True or false Compounds form a substance that is different from the original atoms |
True |
|
|
Chemical reactions occur when |
Atoms combine or dissociate from other atoms |
|
|
Chemical bonds involve an interaction between the |
Electrons of two or more atoms |
|
|
How are atoms united
|
By chemical bonds |
|
|
Electrons occupy energy levels called |
Electron shells |
|
|
Which electrons are most strongly attracted and why |
The electrons closest to the nucleus because the are lowest in energy |
|
|
Distinct properties of a shell |
Number of electrons has an upper limit Shells closest to the nucleus fill first and can hold two electrons Every other shell can hold eight electrons |
|
|
How many electrons can the shell closest to the nucleus hold |
Two |
|
|
What is the outer shell called |
The valance shell |
|
|
Bonding involves interactions between... |
Electrons in the valence shell |
|
|
If a valance shell is full, does it form a bond |
No, because it is full, it has no space to bond |
|
|
When is the valence shell stable |
When it has a complete compliment of electrons |
|
|
Why do chemically based atoms form bonds |
Because they are trying to complete the valence shell. When the valence shell is complete (full) it is stable. |
|
|
Are inert elements stable |
Yes |
|
|
Will inert elements form a chemical reaction |
No, because it is stable |
|
|
Do inert elements have complete or incomplete valence shells |
Complete |
|
|
Give an example of inert elements |
The noble gases |
|
|
Rule of 8's |
No more than eight electrons in a shell |
|
|
When do ionic bonds form |
When electrons are completely transferred from one atom to another |
|
|
Ionic bonds occur because of the |
Force of attraction between two oppositely charged ions |
|
|
What are ions |
Charged particles |
|
|
Fill in the "blanks" Anions are "blank"; has "blank" electrons |
Anions are "negative"; has "gained" electrons (Example: When your bank account is overdrawn, there is nothing left to take. All you can do is make a deposit. Anions will always GAIN electrons.) |
|
|
Fill in the "blank" Cations are "blank"; has "blank" electrons |
Cations are "positive"; has lost electrons (Example: Bank account is in the positive. A cation can lose |
|
|
What does inert mean |
It is stable, there will not be a chemical reaction |
|
|
True or false Reactive ions have full valence shells |
False |
|
|
Do reactive elements tend to lose, gain or share electrons |
They lose, share and gain |
|
|
In reactive elements, what does losing, gaining and sharing electrons allow for |
Allows for bond formation, which produces stable valence |
|
|
What does the prefix Intra mean |
Inside, or from within |
|
|
Intramolecular bonds |
Bonds within a molecule |
|
|
Purpose of intramolecular bonds |
Holds atoms together to form a molecule |
|
|
Two types of Intramolecular bonds |
Ionic bond Covalent bond |
|
|
What does the prefix Inter mean |
In between |
|
|
Ionic |
Exchange (Example: Two separate valence shells both have seven electrons, they are both missing one electron. So one says, how about if I jump ship and you jump ship, (meaning switching spots) by switching spots they form a bond to help complete that last outside spot, they both EXCHANGE to form a bond) |
|
|
Covalent |
Sharing (Example: Two separate valence shells both have seven electrons, they are both missing one electron they are too lazy to jump ship so they decide to just share the last spot.) |
|
|
Which bond is the strongest and why |
The covalent bond because of SHARING (Example: shaking hands, they are sharing so it is very strong) |
|
|
Dual covalent bond share |
Two pair of electrons |
|
|
Intermolecular bond |
Bonds between molecules; holds molecules together |
|
|
What type of bond is a hydrogen bond |
Intermoleular bond (Example: shaking hands with a piece of paper in between. Easily broken apart) |
|
|
What are the four main types of bonds |
Intermolecular Intramolecular Hydrogen Ionic |
|
|
What type of bond is an ionic bond |
Intramolecular bond |
|
|
What type of bond is a covalent bond |
Intramolecular bond
|
|
|
What bond is the weakest |
Hydrogen bond |
|
|
Name the two types of ions |
Anion Cation |
|
|
Single covalent bonds share |
One pair of electrons |
|
|
Polarity |
Covalent bonded molecules |
|
|
The two types of molecules in covalent bonds |
Polar Non-polar |
|
|
Non-polar molecules have an |
Equal sharing of electrons |
|
|
Fill in the "blank" Non-polar molecules are "blank" as a molecule |
Non-polar molecules are "electrically neutral" as a molecule |
|
|
True or false Polar molecules share electrons equally |
False |
|
|
Fill in the "blank" Polar molecules have a "blank" and a "blank" side |
Polar molecules have a "positive" and a "negative" side |
|
|
What is reverse polarity |
Taking a battery and turning it around. It reverses the polarity from what was to what is |
|
|
Why are non-polar molecules neutral |
Because there is equal sharing going on |
|
|
There is always an odd man out and unequal sharing in which polarity; polar or non-polar |
Polar |
|
|
Chemical reactions |
Making OR breaking bonds between atoms |
|
|
True or false In a chemical reaction, the total number of atoms changes |
False (The total number of atoms remains the same but appear in different combinations) |
|
|
True or false In chemical reactions, the total number of atoms remains the same but appear in different combinations |
True |
|
|
Synthesis |
Building something (which will require energy) |
|
|
Synthesis reaction |
Atoms or molecules combine and energy is absorbed for bond formation (Example: A+B= AB) (Example: Walking to the vending machine, uses energy) |
|
|
In synthesis reaction, why is energy absorbed |
For bond formation |
|
|
Decomposition |
Breaking apart (which will release energy) |
|
|
Decomposition reaction |
Molecule is broken down and chemical energy is released (Example: AB=A+B) (Example: Pile driving the vending machine, releases energy)) |
|
|
What is glycogen |
Stored sugar in animals |
|
|
What are stored sugar in plants |
Starch Fiber |
|
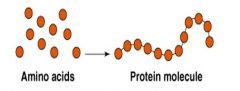
Is an example of... |
Synthesis reaction |
|
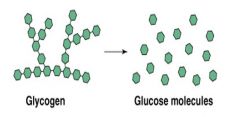
Is an example of... |
Decomposition reaction |
|
|
Exchange reaction |
Involves BOTH synthesis and decomposition reactions (Example: AB+C=AC+B) |
|
|
What happens in exchange reaction |
Switch is made between molecule parts and different molecules are made |
|
|
During exchange reaction, are using AND releasing energy performed |
Yes |
|
|
A stored version of sugar is essentially |
Fat |
|
|
Taking blood glucose and to make ATP is an example of |
Exchange reaction |
|
|
Which compound lacks Carbon, organic or inorganic |
Inorganic |
|
|
Which compound contains Carbon, organic or inorganic |
Organic |
|
|
Which compounds tends to be simpler , organic or inorganic |
Inorganic |
|
|
Which compounds are mostly covalent bonded, organic or inorganic |
Organic |
|
|
C6H12O6 (glucose) is an example of which compound, organic or inorganic |
Organic |
|
|
H2O (water) is an example of which compound, organic or inorganic |
Inorganic |
|
|
What is the most abundant inorganic compound in the body |
Water |
|
|
Vital properties of water (Meaning, what does it do for us) |
High heat capacity Polarity/solvent properties Chemical reactivity - hydrolysis Cushioning (insulation) |
|
|
What does solvent mean |
Breaking down (Usually a liquid base) |
|
|
Hydrolysis |
Cutting water |
|
|
What is a Solution |
A homogenous mixture of two or more components; evenly distributed (Example: bottle of Dr. Pepper) |
|
|
Homogenous |
Uniform |
|
|
What is a Solute |
What is being broken down |
|
|
What is a Suspension |
A mixture that will separate unless continually shaken (Example: Oil and water, dirt and water) |
|
|
What does colloid mean |
Gel |
|
|
Cholloidal suspension |
Solvent is a colloid (gel) and solute is so small that it remains suspended but not equally distributed (Example: An oreo Mcflurry or a Reeses pieces frenzy) |
|
|
What type of compound is salt |
Inorganic (it is not carbon based) |
|
|
Why is salt so important |
It is vital to many body functions |
|
|
Fill in the "blank" Salt includes "blank" which conduct electrical currents |
Salts include "electrolytes" which conduct electrical currents |
|
|
Fill in the "blanks" Salts easily "blank' into ions in the "blank" of "blank" |
Salts easily "dissociate" into ions in the "presence" of "water" |
|
|
What is dehydration |
An imbalance in your body, robbing the body of it's water content and electrolytes |
|
|
With dehydration, which "system" suffers the most |
The nervous system |
|
|
What do acids do |
Release detectable hydrogen ions (H+); proton donors |
|
|
What do bases do |
Release hydroxide ion (OH-); proton acceptors |
|
|
What happens in neutralization reaction |
Acids and bases react to form water and a salt |
|
|
Where does neutralization fall on the pH scale |
7 (7 is neutral) |
|
|
What are you measuring if something is acidic |
You are measuring the H+ or the hydrogen content of that item |
|
|
What are you measuring if the item is a base (or basic) |
You are measuring the OH- ion distribution known as hydroxide |
|
|
What is a salt |
A compound element (Na) |
|
|
What is an electrolyte |
Minerals in your blood and other body fluids that carry an electric charge
|
|
|
What does pH measure |
The relative concentration of hydrogen ions |
|
|
Choose the correct underlined word Everything above or below 7 is considered acidic on the pH scale |
Below (as in number) |
|
|
Choose the correct underlined word
Everything above or below 7 is considered basic on the pH scale |
Above (as in number) |
|
|
The three important compounds |
Salts Acids Bases |
|
|
How are carbohydrates classified |
By their size |
|
|
What does the prefix mono mean |
One |
|
|
What does the prefix di mean |
Two |
|
|
What are monosaccharides |
Simple sugars |
|
|
Name the five simple sugars |
Glucose Fructose Galactose Ribose Deoxyribose |
|
|
Name the three disaccharides |
Sucrose Maltose Lactose |
|
|
What does disaccharides mean |
Two simple sugars joined by dehydration synthesis |
|
|
Why are some people lactose intolerant |
They lack an enzyme (meaning they lack the ability to break down the sugar in milk) |
|
|
What is lactose |
Sugar in milk |
|
|
What does the prefix poly mean |
More than two |
|
|
What does polysaccharide mean |
Long branching chains of linked simple sugars |
|
|
Polysaccharides are commonly referred to as |
Complex carbohydrates |
|
|
What is the polysaccharide in animals known as |
Glycogen |
|
|
What are the polysaccharides in plants known as |
Starch Fiber |
|
|
What are all carbohydrates used by the body for |
Energy |
|
|
Finish the sentence Carbohydrates are used by the body to |
Process energy |
|
|
Are carbohydrates an organic or inorganic compound |
Organic |
|
|
What are lipids |
Fat |
|
|
Are lipids soluble or insoluble in water |
Insoluble |
|
|
What do lipids contain |
Carbon Hydrogen Oxygen |
|
|
Where are triglycerides found |
In fat deposits from the food we eat
|
|
|
Does cholesterol get used by the body |
NO Not ever |
|
|
What are triglycerides also known as |
Neutral fats |
|
|
What are neutral fats (triglycerides) composed of |
Composed of three fatty acids and glycerol |
|
|
Neutral fats (triglycerides) are a source of |
Stored energy |
|
|
What are the two types of cholesterol |
LDL = Low density lipoproteins HDL= High density lipoproteins |
|
|
Which of the two types of cholesterol is the bad one |
LDL |
|
|
Which of the two types of cholesterol is the good one |
HDL |
|
|
Between saturated fats and unsaturated fats, which one is unhealthy to the body |
Saturated fats |
|
|
Why are saturated fats unhealthy to the body |
Saturated fats are full of LDL and our bodies can't process it |
|
|
Why can't our bodies process LDL |
Because our body doesn't make any LDL so it doesn't know how to process it and break it down |
|
|
The human body is capable of making which type of cholesterol, LDL or HDL |
HDL |
|
|
What percentage of our daily needs is the body able to make of HDL |
85% |
|
|
What percentage of our daily needs of HDL has to come from our diet |
15% |
|
|
What organ is responsible for making HDL |
The liver |
|
|
What does LDL cause in the human body |
Plaquing of the arteries (Example: not picking u the garbage on a street for a month even though everyone continues to put the garbage by the road) (Blood vessels end up the same way from LDL) |
|
|
What is the best way to remove LDL from the body |
Exercise, cardio |
|
|
Where does LDL collect in the body |
Blood vessels |
|
|
What are the common lipids found in the human body |
Phospholipids Steroids Neutral fats (triglycerides) |
|
|
What do phospholipids do |
Maintain your membranes at the cellular level |
|
|
What are phospholipids composed of |
Composed of two fatty acids, glyserol, and a phosphate group |
|
|
What do phospholipids do |
Form cell membranes |
|
|
Is the body capable of producing its own steroids |
Yes |
|
|
Is eliminating lipids from your body healthy |
NO |
|
|
What are included in steroids |
Cholesterol Bile salts Vitamin D Some hormones |
|
|
Which lipids does our body need to be healthy |
Phospholipids |
|
|
Main purpose of cholesterol |
The basis for all steroids made in the body |
|
|
In determining which triglyceride is good or bad for you, what does the L in LDL and the H in HDL stand for |
L in LDL stands for Lousy (bad) H in HDL stands for Happy (good) |
|
|
What are proteins made of |
Amino acids held together by peptide bonds (basically long chains of amino acids) |
|
|
How much LDL comes from your diet |
100% |
|
|
ESSENTIAL amino acids |
Has to be acquired from the diet |
|
|
NONESSENTIAL amino acids |
The body can make them |
|
|
If you deny your body of all lipids what are you actually doing to your body |
Decreasing the integrity of your cell membranes because you are depriving yourself of phospholipids |
|
|
What is the phospholipid bylayer and what forms it |
The integrity of your cells and it is formed by phospholipids (hence the name) |
|
|
You can not have a protein if you have no... |
Amino acids |
|
|
Complete proteins |
Contain all the essential amino acids; animal sources |
|
|
Incomplete proteins |
Lacking one or more essential amino acids; vegetable sources must be eaten in combination |
|
|
Can a vegetarian style diet be healthy |
Yes, IF it is done correctly |
|
|
How is a vegetarian style diet done correctly |
You must supplement your diet to get all of the essential amino acids |
|
|
What happens if you are lacking in essential amino acids |
The body is unable to make complete proteins, which causes malnourishment |
|
|
Fill in the "blank" Proteins account for over half of the body's "blank" "blank" |
Proteins account for over half of the body's "organic" "matter" |
|
|
What are the two types of proteins |
Structural Functional |
|
|
Which type of protein is fibrous |
Structural |
|
|
Which type of protein is globular |
Functional |
|
|
Structural (fibrous) proteins |
Provides for construction materials for body tissues |
|
|
Functional (globular) proteins |
Play a vital role in cell function |
|
|
Which protein plays a vital role in cell function |
Functional proteins |
|
|
Which protein provides for construction materials for body tissues |
Structural proteins |
|
|
Functional proteins act as.. |
Enzymes, hormones and antibodies |
|
|
Enzyme |
Act as biological catalyst |
|
|
What do enzymes do |
Increase the rate of chemical reaction |
|
|
Catalyst |
A substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change.
(example: salt in water on the stove, the salt is a catalyst making the water boil faster.) |
|
|
Name the two nucleic acids |
DNA RNA |
|
|
Nucleotide bases for DNA |
Adenine Thymine Cytosine Guanine |
|
|
Nucleotide bases for RNA |
Adenine Uracil Cytosine Guanine |
|
|
What is RNA |
RNA is a copy of the DNA |
|
|
Using the nucleotides that pair up with each other what would AAA CCG turn into for DNA |
TTT GGC |
|
|
In DNA what can Adenine pair up with |
Thymine |
|
|
In DNA what can Thymine pair up with |
Adenine |
|
|
In DNA what can cytosine pair up with |
Guanine |
|
|
In DNA what can guanine pair up with |
Cytosine |
|
|
Using the nucleotides that pair up with each other what would AAA CCG turn into for RNA
|
UUU GGC |
|
|
In RNA what can Adenine pair up with
|
Uracil |
|
|
In RNA what can Uracil pair up with
|
Adenine |
|
|
In RNA what can cytosine pair up with
|
Guanine |
|
|
In RNA what can guanine pair up with |
Cytosine |
|
|
What is the purpose of RNA |
In order to heal ourselves, we need to take our DNA and replicate & copy it to build a new skin cell for the broken area (Example: broken skin) |
|
|
What do the letters DNA stand for |
Deoxyribonucleic acid |
|
|
How are Deoxyribonucleic acids organized |
By complimentary bases to form a double helix
|
|
|
When speaking in construction terms, what is the DNA |
The blueprints |
|
|
When speaking in construction terms, what is RNA |
The instructions |
|
|
Does EVERY cell, or only some cells have a copy of the DNA |
Every cell |
|
|
When must the DNA be replicated |
Before cell division |
|
|
What do the letters ATP stand for |
Adenosine triphosphate |
|
|
What is ATP |
Chemical energy used by all cells |
|
|
How is ATP replenished |
By oxidation of food fuels (Carbohydrates and lipids/fats) |

