![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
64 Cards in this Set
- Front
- Back
|
What is the normal bonding pattern for hydrogen?
|
One valence electron.
One bond. No lone pairs. |
|
|
What is the normal bonding pattern for a halogen?
|
7 valence electrons.
One bond. 6 lone pairs |
|
|
What is the normal bonding pattern for nitrogen?
|
5 valence electrons.
3 bonds 1 lone pair. |
|
|
What is the normal bonding pattern for oxygen?
|
6 valence electrons
2 bonds. 2 lone pairs. |
|
|
What is the normal bonding pattern for hydrogen cation?
|
1 bond
+1 charge no lone pairs |
|
|
What is the normal bonding pattern for hydrogen anion?
|
1 bond
-1 charge 1 lone pair. |
|
|
What is the normal bonding pattern for the carbocation?
|
3 bonds
no lone pairs +1 charge. |
|
|
What is the normal bonding pattern for the carboanion?
|
3 bonds
1 lone pair -1 charge. |
|
|
What is the normal bonding pattern for the C dot ion?
|
3 bonds
1 single electron no net charge. |
|
|
What is the normal bonding pattern for the nitrogen cation?
|
4 bonds
no lone pairs +1 charge. |
|
|
What is the normal bonding pattern for the nitrogen anion?
|
2 bonds
2 lone pairs -1 charge. |
|
|
What is the normal bonding pattern for the oxygen anion?
|
1 bond.
3 lone pairs. -1 charge. |
|
|
What is the normal bonding pattern for the oxygen cation?
|
3 bonds.
1 lone pair. +1 charge. |
|
|
What is the normal bonding pattern for a halogen anion?
|
0 bonds
4 lone pairs. -1 charge |
|
|
Alcohol
|
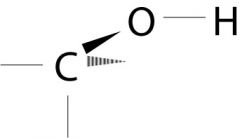
What is the name of this functional group?
|
|
|
Aldehyde.
|
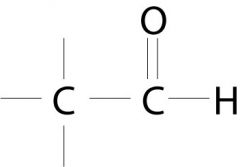
What is the name of this functional group?
|
|
|
Carboxylic acid
|
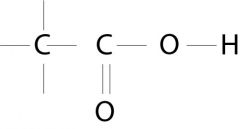
What is the name of this functional group?
|
|
|
Ether
|
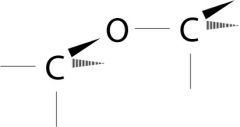
What is the name of this functional group?
|
|
|
Ketone
|
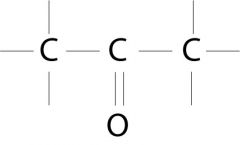
What is the name of this functional group?
|
|
|
What are isomers?
|
Compounds that have the same molecular formula but different arrangements of bonding in their atoms.
|
|
|
What are the two main types of isomers?
|
1. Constitutional isomers.
2. Steroisomers. |
|
|
What are constitutional isomers?
|
Compounds with different connections between the atoms.
|
|
|
What are stereoisomers?
|
Compounds with same connections betwen the atoms but different arragement of atoms in space.
|
|
|
What are the three different relevant types of orbitals for organic chemistry?
|
1. S-orbitals
2. P- orbitals 3. D-orbitals |
|
|
What are s-orbitals?
|
Spherical in shape, they can only have 2 electrons in the orbital. 2s orbitals have 2 nodes, 3s orbitals have 3.
|
|
|
What are p-orbitals?
|
Dumbell shaped, 3 different spatial orientations, can only hold 2 in each lobe (for a total of 6 for all p-orbitals.
|
|
|
What are d-orbitals?
|
4 Clovershaped, 1 dumbbell w/spherical center.
|
|
|
What are the two different types of covalent bonding?
|
1. sigma bond
2. pi bond |
|
|
What is a sigma bond?
|
A covalent bond where the electrons are centered about the axis between the two nuclei.
|
|
|
What is a pi bond?
|
A covalent bond where the shared electrons occupy regions above and below axis between two nuclei.
|
|
|
What are hybrid orbitals?
|
Orbitals where the wave function s of the different oribitals combine to form new orbitals.
|
|
|
What is an sp-orbital?
|
A combination of an s-orbital and a p-orbital.
|
|
|
What is an sp2-orbital?
|
A combination of an s-orbital and 2 p-orbitals.
|
|
|
What is an sp3-orbital?
|
A combination of an s-orbital and 3 p-orbitals.
|
|
|
What are the four types of bonding?
|
1. Ionic
2. Polar covalent 3. Non-polar covalent 4. Hydrogen |
|
|
What is an ionic bond?
|
Electrons are given away ... not shared and held together by +/- attractive force of the ions.
|
|
|
What is a polar covalent bond?
|
Shared electron bond where the electrons are strongly attracted to one element more than another.
|
|
|
What is a non-polar covalent bond?
|
Shared electron bond where the electrons are equally distributed about all elements.
|
|
|
What is hydrogen bonding?
|
An attractive interaction between a hydrogen atom bonded to an oxygen, nitrogen, or fluorine atom and an unshared electron pair on another atom.
|
|
|
What is the trend in the Periodic Table for valence electrons?
|
The group number is equal to the number of valence electrons for the element.
|
|
|
What trends are associated with an elements position on the Periodic Table with respect to electronegativity?
|
1. Electronegativity increases to the right in periods.
2. Electronegativity increases as you travel up the group. |
|
|
What are the rules for drawing Lewis structures?
|
1. Based on molecular formula, count the number of valence electrons.
2. Given the connectivity, connect the bonded atoms by a shared electron pair or a dash. 3. Count the number of electrons in the bonds and get the remainder. 4. Add remainder to the atoms to get 8 in valence shell. 5. Calculate the formal charge. |
|
|
What is the formula to calculate formal charge?
|
FC = Group # - (# unshared electrons - 1/2 # shared electrons)
|
|
|
What are line structures?
|
Structures with all C's and H's omitted.
|
|
|
What are condensed structures?
|
Structures which show all the structural units (e.g. CH3)
|
|
|
What is VSEPR?
|
Valence Shell Electron Pair Repuslion ... a method for determining molecular geometry.
|
|
|
What are the important principles of VSEPR?
|
1. Electron pairs in carbon always form at tetrahedral angles.
2. Molecular shape dependent upon the bonded atoms and NOT their unshared pairs. |
|
|
What are the most important VSEPR shapes in organic chemistry?
|
1. Tetrahedral
2. Pyramidal 3. Trigonal Planar 4. Bent |
|
|
What is the trend in association between bond angles and unshared electron pair repulsion.
|
2 bonded pairs < 1 bonded pair + 1 unshared pair < 2 unshared pairs
|
|
|
What are the two different types of stereoisomers?
|
1. diastereoisomers
2. enantiomers |
|
|
What are diastereoisomers?
|
Non-mirror image stereoisomers.
|
|
|
What are enantiomers?
|
Mirror image stereoisomers.
|
|
|
What is a Bronsted-Lowry acid?
|
A substance that donates an H+.
|
|
|
What is a Bronsted-Lowry base?
|
A substance that accepts an H+.
|
|
|
What is a Lewis acid?
|
An electron pair acceptor.
|
|
|
What is a Lewis base?
|
An electron pair donator.
|
|
|
What is the acid-dissociation constant?
|
Ka = [H30+][A-]/[HA]
The equilibrium constant for the dissociation of an acid in water. |
|
|
What is the pKa?
|
- log Ka
|
|
|
What is the relationship between Ka and acid strength?
|
The larger the Ka the stronger the acid.
|
|
|
What is the relationship between pKa and acid strength?
|
The smaller the pKa the stronger the acid.
|
|
|
What are the indicators of acid strength?
|
1. H-A bond strength.
2. Electronegativity. 3. Electron delocalization in conjugate base. |
|
|
What is the trend associated with the H-A bond strength?
|
The weaker the H-A bond the stronger the acid.
|
|
|
What is the trend associated with electronegativity?
|
The more electronegative an H+ substrate the stronger an acid it is.
|
|
|
Why does electron delocalization increase acidity?
|
Affects the equilibrium of the reaction by stabilizing the conjugate base pulling equilibrium towards it.
|

