![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
51 Cards in this Set
- Front
- Back
|
What is the definition of biochemistry? |
The description of structure, organization, and function of life at the molecular level |
|
|
What is an example of how biochemistry is applied to medicine/disease? |
Sickle cell anemia |
|
|
What is an example of how biochemistry is applied to pharmacology/toxicology? |
Cancer chemotherapy/cis-platin |
|
|
What is an example of how biochemistry is applied to nutrition? |
Vitamins |
|
|
What is an example of how biochemistry is applied to agriculture? |
Herbicides, pesticides, GMOs |
|
|
What is an example of how biochemistry is applied to the production of chemicals? |
Catalysts/enzymes |
|
|
What are the four most abundant elements in biological molecules? |
1. Carbon 2. Hydrogen 3. Nitrogen 4. Oxygen |
|
|
What are some elements that are moderately abundant in biological molecules? |
Sodium Magnesium Potassium Calcium Phosphorus Sulfur Chlorine |
|
|
What are some elements that are only present in trace amounts in biological molecules? |
Boron Flourine Aluminum Silicon Vanadium Chromium |
|
|
What are the building blocks of proteins? |
Amino acids |
|
|
What three components does an amino acid contain? |
1. Amino group 2. Carboxylic acid group 3. Side chain |
|
|
What are monosaccharides and sugars classified as? |
Carbohydrates |
|
|
What is the general chemical formula of a carbohydrate? |
(CH2O)n, where n is greater than or equal to 3 |
|
|
In what two ways can a carbohydrate be drawn? |
1. Fisher projection (linear) 2. Hawthorn projection (cyclical) |
|
|
How are carbohydrates linked? |
Via glycosidic bonds |
|
|
What are the building blocks of nucleic acids? |
Nucleotides |
|
|
What are the three main components of nucleotides? |
1. Phosphate groups 2. Sugar (ribose) 3. Base (changes from five different options) |
|
|
How do nucleotide residues link? |
Via phosphodiester bonds |
|
|
What are fats classified as? |
Lipids |
|
|
What is one of the most important lipids in human health? |
Cholesterol |
|
|
Why do monomers connect? |
To form larger structures |
|
|
When monomers connect to form larger structures, what are these larger structures called? |
Polymers |
|
|
Once part of a polymer, what is a monomer referred to as? |
Residue |
|
|
What is enthalpy and what units is it measured in? |
The heat content of a system Units = J . mol-1 |
|
|
What is entropy and what units is it measured in? |
A measure of the system's disorder or randomness Units = J . K-1mol-1 |
|
|
What does deltaG refer to? |
Gibbs free energy change |
|
|
What does deltaH refer to? |
Enthalpy change |
|
|
What does deltaS refer to? |
Entropy change |
|
|
What is the equation to calculate Gibbs free energy change? |
deltaG = deltaH - T(deltaS) where T = temperature in Kelvin |
|
|
What is the definition of Gibbs free energy change? |
A measure of the free energy of a system based on enthalpy (H) and entropy(S) |
|
|
What are the units for deltaG? |
Units = J . mol-1 |
|
|
What does it mean for a reaction when deltaG is < 0? |
The reaction is spontaneous |
|
|
What does it mean for a reaction when deltaG is > 0? |
The reaction is nonspontaneous |
|
|
What is an exergonic reaction? |
A spontaneous reaction |
|
|
What is an endergonic reaction? |
A nonspontaneous reaction |
|
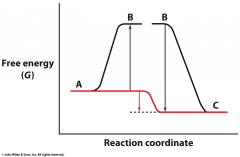
Describe this graph and what occurs because of it? |
deltaG is positive for the A to B transition deltaG is negative for the B to C transition The reactions will be coupled |
|
|
Why do coupled reaction occur? |
Nonspontaneous reactions do not generally occur Spontaneous reactions can be coupled with nonspontaneous reactions so that the net reaction occurs spontaneously |
|
|
T or F: The energy from photosynthesis does not involve a coupled chemical reaction |
False |
|
|
What are the major and minor functions of proteins? |
Major functions: Carry out metabolic reactions and support cellular structures Minor function: Store energy |
|
|
What are the major and minor functions of nucleic acids? |
Major function: Encode information Minor functions: Carry out metabolic reactions and support cellular structures |
|
|
What are the major and minor functions of polysaccharides? |
Major functions: Store energy and support cellular structures Minor function: Encode information |
|
|
What is the hypothesis for the synthesis of biological materials from lightning? |
Experiment to determine if it would be possible for H2, H2O, NH3, and CH4 to give rise to amino acids when struck by lightning |
|
|
What is the hypothesis for the formation of polymers from a mineral surface? |
Positively charged clay is able to promote the polymerization of the nucleotides into RNA indicating that the initial polymerization of nucleotides could have been facilitated by a charged mineral surface |
|
|
What four steps occur for the hypothesis for the self-replication of polymers? |
1. The polyA molecule serves as a template for the synthesis of a polymer containing uracil nucleotides(U), which are complementary to adenine nucleotides 2. The two polymer chains separate 3. The polyU molecule serves as a template for the synthesis of a new complementary polyA chain 4. The chains again separate and the polyU polymer is discarded, leaving the original polyA molecule and its exact copy |
|
|
What are prokaryotes? |
Small, unicellular organisms that lack a discrete nucleus and have no internal membrane system |
|
|
What are bacteria and archaea classified as? |
Prokaryotes |
|
|
What are eukaryotes? |
Large cells, contain a nucleus and other membrane-bound cellular compartments (organelles) |
|
|
What are microscopic organisms as well as plants and animals included as? |
Eukaryotes |
|
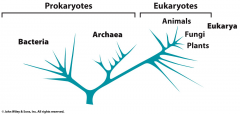
What is this? |
Phylogenetic tree showing bacteria, archaea, and eukaryotes |
|
|
How do you convert Celsius to Kelvin? |
Add 273.15 to the Celsius numeral 0 C = 273.15 K |
|
|
How do you convert Fahrenheit to Kelvin? |
Add 255.372 to the Fahrenheit numeral or 32 F = 273.15 K |

