![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
321 Cards in this Set
- Front
- Back
|
What are the eight specific characteristics of life? |
-Made up of Cells -Reproduce -Universal genetic code -Growth and Development -Obtain and use materials for energy -Respond to Stimuli -Homeostasis -Evolve |
|
|
What is homeostasis? |
the regulation and maintenance of the internal environment of the body |
|
|
What are negative feedback loops? |
they stop the change |
|
|
What are positive feedback loops? |
they increase change |
|
|
What's an example of a negative feedback loop? |

breathing
|
|
|
What's an example of a positive feedback loop? |
torn blood vessel stimulates release of clotting factors |
|
|
What are the steps of the scientific method? |
1. Find a Problem 2. Research the problem/Gather information 3. Form a hypothesis 4. Perform Experiment 5. Gather data/information 6. Analyze date 7. Conclusion |
|
|
What is a hypothesis? |
an educated guess (written in the if, then format) |
|
|
What is an experiment? |
the test of a hypothesis |
|
|
What is data? |
information collected during an experiment |
|
|
What is the control group? |
a group in which you make no changes to. The control group is the standard to compare all the others to |
|
|
What's the independent variable? |
the variable that you as the experimenter change. You have control over this variable. |
|
|
What's the dependent variable? |
the variable that the experimenter has no control or power over (results) |
|
|
What is a theory? |
the best explanation possible for a known phenomenon, based on many experiments |
|
|
What is a law? |
scientific fact |
|
|
What's an example of a theory? |
Evolution, Big Bang, etc. |
|
|
What's an example of a law? |
Law of Gravity, Boyles's Gas Laws, etc. |
|
|
What are the three aspects of a controlled experiment? |
1. Control Group 2. 1 or more experimental groups 3. Only change one variable at a time |
|
|
What are atoms? |
atoms are the smallest particles of an element |
|
|
What is the center of an atom? |
Nucleus |
|
|
What is the positively charged particle in the nucleus? |
Proton |
|
|
What is the negatively charged particle outside the nucleus? |
Electron |
|
|
What's the particle with no change located in the nucleus? |
neutron |
|
|
What's an element? |
a substance which cannot be broken down into simpler substances |
|
|
How many elements occur naturally in nature? |
90 |
|
|
What are energy levels? |
Regions where electrons travel around the nucleus |
|
|
How many electrons does the first energy level hold? |
2 |
|
|
How many electrons does the second energy level hold? |
8 |
|
|
How many electrons does the third energy level hold? |
8 |
|
|
What must happen for an element to be stable? |
All energy levels being used must be full |
|

What is this number?
|
Atomic Number
|
|
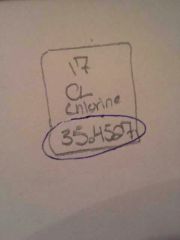
What is this number?
|
Atomic Mass |
|

Draw the energy levels. Is this element stable or unstable?
|
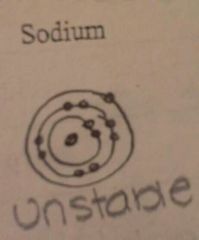
|
|

Draw the energy levels. Is the element stable or unstable?
|
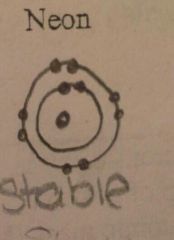
|
|
|
What is an isotope? |
an alternate form of an element that differs in it's number of neutrons |
|
|
What is a compound? |
A substance made of two or more different elements bound together |
|
|
What is a molecule? |
A group of atoms held together by a covalent bond |
|
|
What's a covalent bond? |
Where two atoms share electrons |
|
|
How many electrons need to be shared in a covalent bond? |
"to get one you gotta give one" you have to give as many electrons as you get |
|
|
What are polar covalent bonds? |
they occur when the electrons of a bond are not shared equally as a result some will become slightly negative, some will become slightly positive |
|
|
How can you tell if you have a polar covalent bond? |
ON if you ever see O or N with H you have a Polar Covalent bond |
|
|
What is a nonpolar bond? |
it occurs when the elements of a bond are shared equally |
|
|
What is an ionic bond? |
when two elements of opposite charge combine |
|
|
What is an ion? |
an atom that has acquired a full positive or negative charge by losing or gaining an electron |
|
|
When do chemical reactions occur? |
-when bonds are formed -when bonds are broken -substances change/ are altered into different substances |
|
|
What is metabolism? |
refers to all the chemical reactions that take place within an organism |
|
|
What are products? |
substances formed from a reaction |
|
|
Why is water important? |
it provides a place for chemical reactions to occur and it can dissolve most polar and ionic substances |
|
|
What are hydrogen bonds? |
water molecules attracted to each other -weak bond -important, these bonds help hold things like DNA together |
|
|
What is cohesion? |
when the same type of molecules "stick" together |
|
|
what is adhesion? |
type of attraction which occurs between unlike molecules |
|
|
What is a solution? |
uniform mixture of two or more substances |
|
|
What is a solvent? |
the substance dissolving another substance |
|
|
What is a solute? |
the substance being dissolved |
|
|
What is surface tension? |
allows liquid to resist an external force due to cohesion |
|
|
What is specific heat? |
amount of heat required to raise the temperature by 1 degree celsius |
|
|
What is density? |
-the amount of matter in a given volume -in most substances solid is more dense than liquid state |
|
|
What is pH? |
refers to the Hydrogen ion(H+) versus hydroxide ions(OH-) concentration in a solution |
|
|
What is the range of the pH scale? |
0-14 |
|
|
What is an acid? |
a substance that has more H+ ions and has a pH below 7 |
|
|
What is a base? |
a substance that has more OH- ions and has a pH above 7.1 |
|
|
What does a subscript in a chemical reaction represent? |
the number of atoms of each element in a molecule |
|
|
What does a coefficient in a chemical reaction represent? |
the number of molecules of that substance |
|
|
How do you determine the number of molecules? Atoms of an individual element? |
Molecule: Add coefficients
Individual element atoms: multiply subscript by coefficient |
|
|
Diaphragm |
regulates the amount of light passing up toward the eyepiece |
|
|
Course Adjustment |
moves the body tube to focus the image |
|
|
Fine Adjustment |
moves the body tube slightly to sharpen the image |
|
|
Stage |
supports the slide being so observed |
|
|
Eyepiece |
contains a magnifying lens |
|
|
What are the magnifications of the three objectives? |
4x, 10x, 40x |
|
|
How do you determine total magnification? |
multiply the eyepiece lens by the objective |
|
|
How does the image change when viewed through a microscope? |
It's upside down and to the left |
|
|
How do you convert cm to mm? How do you convert mm to microns? |
1 cm = 10mm 1mm= 1,000 microns |
|
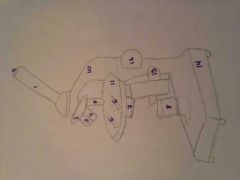
|
1. Body Tube |
|
|
Why are carbon atoms so common in living things? |
-very abundant -can form single, double, triple, or quadrouple bond with itself |
|
|
What is a polymer? |
a long chain of linked monomers |
|
|
What is an organic molecule? |
carbon-based molecule |
|
|
What is an inorganic molecule? |
non-carbon-based molecules |
|
|
what does it mean if something is hydrophilic? |
water loving |
|
|
What is a monomer? |
small molecular units |
|
|
What is a functional group? |
a group of atoms within a molecule that interacts in predictable ways |
|
|
What is a dehydration reaction? |
when a monomer is added to a chain and a water molecule is released |
|
|
What is a hydrolysis reaction/condensation synthesis? |
water is added to bonds between monomers to break them |
|
|
What four categories are large molecules classified as? |
Carbohydrates, lipids, proteins, and nucleic acids |
|
|
What are the of elements that make up Carbohydrates? |
CHO Carbon, Hydrogen, Oxygen 1:2:1 |
|
|
What are the building blocks of carbohydrates? What's an example? |
monosaccharides ex: glucose, fructose |
|
|
What are the two subgroups of Carbohydrates and an example of each? |
Disaccharides ex: sucrose
Polysaccharides ex:starch, cellulose, glycogen |
|
|
What is the function of carbohydrates? |
quick immediate energy |
|
|
What are some examples of carbohydrates? |
Sugars, starches, grains, fibers, and roughage |
|
|
What are the elements of proteins? |
CHONS Carbon, Hydrogen, Oxygen, Nitrogen, Sulfur |
|
|
What are the building blocks of proteins? |
Amino Acids |
|
|
How many types of Amino acids are there? |
20 |
|
|
What bond links amino acids together? |
Peptide bonds |
|
|
What is the structure of a primary amino acid? |
linear sequence of amino acids |
|
|
what is the structure of a secondary amino acid? |
helical or pleated sequence |
|
|
what is the structure of a tertiary amino acid? |
globular sequence of amino acids |
|
|
what is the structure of a quaternary amino acid? |
protein made up of more than one amino acid chain |
|
|
What are the functions of proteins? |
-makes up fibers of muscles -make up antibodies -make up enzymes -used as messengers (transport oxygen in bloodstream) -hair |
|
|
What is denaturation? |
breakdown of a proteins structure |
|
|
What elements make up lipids? |
CHO Carbon, Hydrogen, Oxygen No ratio |
|
|
What are the building blocks of lipids? |
Fats and glycerol |
|
|
What are the subgroups of lipids? |
Saturated fats and unsaturated fats |
|
|
what state are saturated fats in at room temperature? |
Solid |
|
|
What kind of bonds do saturated fats have? |
single bonds |
|
|
What state are unsaturated fats in at room temperature? |
liquid |
|
|
What kind of bonds do unsaturated fats have? |
double bonds |
|
|
What is cholesterol? |
-fat -important for making up hormones -soft waxy membrane found in bloodstream with lipids -used to form cell membranes, some hormones, and is needed for other cell functions |
|
|
How much cholesterol does the body make? |
It makes all it needs |
|
|
What do lipoproteins do? |
they transport cholesterol to and from cells |
|
|
What is LDL (Low Density Lipoprotein)? |
-bad cholesterol -carries cholesterol in blood, too much can form plaque in arteries |
|
|
What is HDL (High Density Lipoprotein)? |
-good cholesterol -takes cholesterol out of the blood and back to the liver |
|
|
What are Trans Fat? |
-hydrogenated oils -made by hydrogenating oils to make them solid -when consumed it increases LDL concentration |
|
|
What are the functions of lipids? |
-Major part of cell membranes -long term energy storage -insulation |
|
|
What are some examples of lipids? Healthy Fats? Saturated Fats? Unsaturated Fats? |
Healthy Fats: fish, meat, vegetables Saturated Fats: butter and lard Unsaturated Fats: oils (vegetable, olive, corn, etc.) |
|
|
What are the elements in Nucleic Acids? |
CHONP Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorus |
|
|
What are the building blocks of nucleic acids? |
nucleotides |
|
|
What are the functions of Nucleic Acids? |
Stores genetic or hereditary information |
|
|
What are some examples of Nucleic Acids? |
DNA - master copy of an organisms information RNA- codes for a copy of DNA, used in protein synthesis |
|
|
What element is added to make trans fats? |
hydrogen |
|
|
What is an enzyme? |
catalysts that help reduce the amount of activation energy needed for a chemical reaction |
|
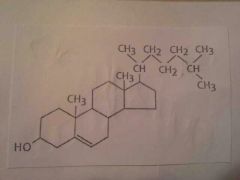
|
Steriod |
|
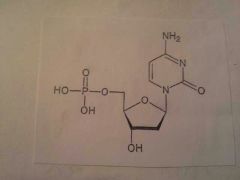
|
Nucleotide
|
|
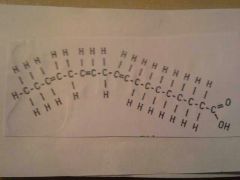
|
Unsaturated Fat
ex: oil |
|
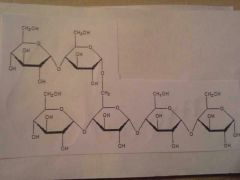
|
Polysaccharides
ex: starch, cellulose, glycogen |
|
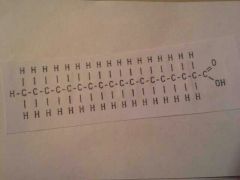
|
Saturated Fat
ex: butter, lard |
|
|
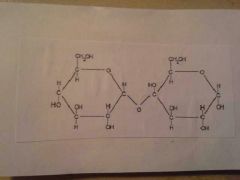
Disaccharide |
|
|
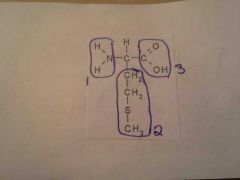
Amino Acid
1. Amino Group 2. R Group 3. Carboxyl Group |
|
|
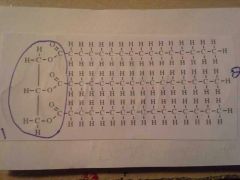
Triglyceride (Lipid)
1. Glycerol Head 2 Fatty Acids **Unsatured |
|
|
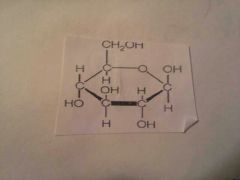
Monosaccharide |
|
|
What is an Exergonic Reaction? |
-reaction releases energy -products have less energy than initial reactant -some energy lost as heat |
|
|
What is an Endergonic Reaction? |
-products have more energy than that of initial reactants -reaction requires energy to initiate reactant |
|
|
What is Competitive Inhibition? |
when a molecule other than a substrate bonds to the active site -usually involves inhibitors, or poisons, that do not belong in a cell ex: cyanide |
|
|
What polysaccharide is used to store exess glucose in humans? |
glycogen |
|
|
Which polysaccharide is in plant walls? |
Cellulose |
|
|
What are the end products of hydrolysis of a triglyceride? |
Glycerol and Fatty Acids |
|
|
What functional groups are essential to chemistry life? |
-hydroxyl -carbonyl -carboxyl -amino |
|
|
Up to how many bonds can carbon make? |
4 |
|
|
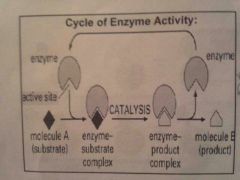
What is a substrate? |
Also known as a reactant, the substance that bonds to an enzyme |
|
|
What is activation energy? |
the energy required to perform a chemical reaction *enzymes lower this amount |
|
|
Which polysaccharide is used to store excess glucose in plants? |
Starch |
|
|
What are factors that that affect enzymes? |
-temperature -pH -substrate |
|
|
What is the Enzyme-Substrate Complex? |
when enzyme and substrate are bonded together |
|
|
What's an allosteric inhibition? |
when an inhibitor bonds to an enzyme somewhere other than the active site *it could be a poison **could also be natural |
|
|
.
|
|
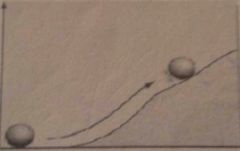
Which reaction is this, exergonic or endergonic?
|
Endergonic
|
|
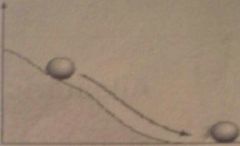
which reaction is this exergonic or endergonic?
|
exergonic
|
|
|
What is feedback inhibition? |
It occurs when a product of the metabolic pathway inhibits the pathway of it's own creation -used to regulate reactions |
|
|
What is a cell? |
The basic unit of life |
|
|
Who was Robert Hooke? What did he do? |
He was a the first to use a compound microscope. He viewed cork underneath of it and saw many "little rooms" he called cells |
|
|
What three parts of the cell theory did Schleiden and Schwann come up with? |
1. All living things are made of cells 2. Cells are the basic units of structure and function 3.All cells are produced by existing cells. |
|
|
What's a prokaryote? |
a cell without a nucleus, DNA floats freely ex: bacteria |
|
|
What's a eukaryote? |
-larger more complex -have nucleus that contains DNA -organelles -may live as single cells or multicellular organisms |
|
|
What's an organelle? |
membrane-bound structures that act like individual organs, each carries out a specific function |
|
|
Cytoplasm |
area between the nucleus and cell membrane -composed of water, protein, carbs, and salt -acts as insulation |
|
|
Nucleus |
contains all the DNA within a cell |
|
|
Nucleolus |
center region of the nucleus where DNA is located *also where ribosomes are made |
|
|
Nuclear Envelope/Nuclear Membrane |
membrane that protects the nucleus and DNA inside |
|
|
Ribosomes |
small grain-like particles of RNA inside the cytoplasm, located throughout the cell, make protein from information in DNA, can be in cytoplasm or attached to rough E.R. |
|
|
Rough Endoplasmic Reticulum |
covered in ribosomes which produce protein and transports it thought the cell *directly attached to nucleus |
|
|
Smooth Endoplasmic Reticulum |
produces cell membrane lipids and breaks down drugs *can be attached to rough e.r. or floating |
|
|
Golgi Body |
puts finishing touches on proteins |
|
|
Vacuoles |
stores food, water, and waste *huge in plants |
|
|
Lysosomes |
little organelles filled with digestive juices(enzymes and acids) *plants don't have them |
|
|
Chloroplasts |
make their own food via phtosynthesis |
|
|
Plastid |
makes it's own food |
|
|
Mitochondria what is the outer membrane's purpose? The inner? |
produces energy outer membrane = protection inner membrane = produce energy |
|
|
Cytoskeleton |
structure that maintains cell shape, helps to protect the cell and useful in cell movement and cell division |
|
|
Cell Wall |
thick layer of cellulose that provides additional structure and support for a cell *found in plans, bacteria, and fungi |
|
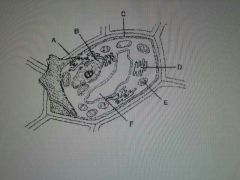
|
A. Cell Wall |
|
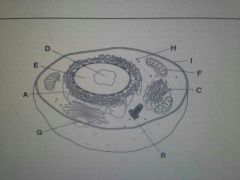
|
A. Rough E.R. |
|
|
Plant vs. Animal |
Plant: Cell wall, chloroplasts Animal: lysosomes |
|
|
What's the path of protein production? |
RNA, Ribosomes (in cytoplasm or rough e.r.), glogi bodies, cell membrane |
|
|
What is Passive Diffusion? |
Moving from high to low concentration, no ATP spent |
|
|
What is Simple Diffusion? |
when small particles with nonpolar bonds move directly through lipid bilayer ex: oxygen, CO2 |
|
|
What is Facilitated Diffusion? |
particles pass through transport proteins due to size or charge ex: H+ ions, glucose |
|
|
What is Active Diffusion? |
moving from low to high concentration, costs ATP |
|
|
What is endocytosis? |
large materials engulfed by cell |
|
|
What is phagocytosis? |
endocytosis of solids |
|
|
What is pinocytosis? |
endocytosis or liquids |
|
|
What is Receptor mediated? |
Endocytosis of multiple solids at once |
|
|
Active vs. Passive |
Active: ATP, low to high Passive: No ATP, high to low |
|
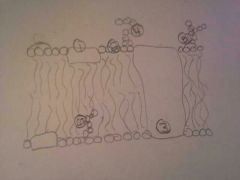
|
1. Peripheral Protein
2. Phospholipids 3. Integral Protein 4. Glycolipid 5. Glycoprotein 6. Cholesterol |
|
|
What is Exocytosis? |
when large molecules are released from the membrane |
|
|
What is osmosis? |
the diffusion of water across a selectively permeable cell membrane |
|
|
What happens when a cell is isotonic? |
solute and water are at equal concentration |
|
|
What happens when a cell is hypertonic? |
There's more solute than water, and the cell will lose weight and shrink |
|
|
What happens when a cell is hypotonic? |
there's more water than solute and the cell will gain weight and get bigger |
|
|
What is cytolysis? |
when cells rupture or explode because they have been placed in a hypotonic solution and gotten too big |
|
|
What is plasmolysis? |
when cells shrivel and die because they have been placed in a hypertonic solution |
|
|
Why don't plants have to worry about cytolysis and plasmolysis? |
they have a cell wall |
|
|
What is an autotroph? |
an organism that makes it's own food |
|
|
What is a heterotroph? |
an organism that consumes to get energy |
|
|
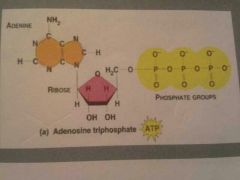
What is ATP? |
Adenine Tri-Phosphate an energy carrier when food is released this is what it turns into |
|
|
Equation for Photosynthesis |
6CO(2) + 6H(2)O + light ---> C(6)H(12)O(6) + 6O(2) |
|
|
Upper/Lower Epidermis |
Provides support and protection for the inside of the leaf |
|
|
Palisade Mesophyll |
Top layer of cells inside the leaf. This is where most light absorption takes place and the most photosynthesis occurs |
|
|
Spongy Mesophyll |
Some photosynthesis occurs here but this is mostly where gas exchange occurs; CO(2) is released and O is absorbed |
|
|
Cuticle |
Waxy layer on top of leaf that prevents water loss |
|
|
Stomate (singular is Stoma) |
the opening between guard cells, where gas is exchanged between leaf and atmosphere |
|
|
Guard Cell |
Open or close the stomate by pumping water in or sucking it out. |
|
|
Phloem |
bottom portion of vein that brings food down from leaves |
|
|
Xyleom |
Top portion of the vein that carries water up from the roots |
|
|
Transpiration |
loss of water through the leaf |
|
|
Parts of leaf involved in transpiration |
Stomate, guard cells, and cuticle |
|
|
Chloroplast |
organelle responsible for Photosynthesis |
|
|
Thylakoid |
individual disks which contain pigments that absorb light |
|
|
Grana (singular form is Granum) |
Stack(s) of thylakoids |
|
|
Stroma |
liquid filled center of the chloroplasts where Calvin Cycle occurs |
|
|
Light Reactions |
Produce ATP and NADPH |
|
|
Calvin Cycle |
Uses ATP and NCDPH to make glucose |
|
|
Pigments |
What absorbs energy from the sun |
|
|
What color will a pigment be? |
the color of the light it DOES NOT absorb |
|
|
Thomas Engelmann (1883) |
He confirmed that conversion of light energy takes place within chloroplast. |
|
|
Thomas Engelmann's Experiment |
He placed a strand of algae on a slide and put a white prism on top, allowing all colors of light to pass through. His theory was that air loving bacteria would group in areas where the most photosynthesis occurs because of the release of oxygen during this process. |
|
|
Main plant cell pigment |
Chlorophyll |
|
|
What are the two types of chlorophyll that make up most of the pigments in the cell? |
Chlorophyll a and Chlorophyll b |
|
|
Chlorophyll A |
absorbs blue-violet and red light |
|
|
Chlorophyll B |
absorbs blue and red light |
|
|
Accessory Pigments |
make up the rest of the pigments in a chloroplast. They absorb green and reflect red, orange and yellow |
|
|
Why do leaves change color? |
The days getting shorter cues the tree that winter is coming and that it should lose it's leaves and go to sleep because soon it will be too cold for it to get the amount of water it needs. Chlorophyll a and b begin to die, which allows the accessory pigments to be visible and is why we see colors. |
|
|
What did chloroplasts evolve from? |
Bacteria |
|
|
ATP is a nucleotide consisting of? |
Three phosphate groups, the sugar ribose, and the nitrogen base adenine |
|
|
electromagnetic spectrum |
all possible emittes radiation |
|
|
can plants survive without oxygen? |
no, it needs oxygen to break down it's food |
|
|
What is the range of visible wavelengths? |
400nm -700nm |
|
|
what is glucose made of? |
two G3P molecules |
|
|
where does the energy for Photosynthesis come from? |
the sun |
|
|
Grana are connected to each other by what? |
lamellae |
|
|
Deciduous |
latin meaning"to fall or cut" |
|
|
needle like leaves vs. large broad leaves |
Advantages of needle-like: they can conserve water |
|
|
zone of abscisson |
where stem of leaf connects to branch amd a scab forms to cut off leaf |
|
|
advantage of zone of abscisson not fully forming? |
the leaves don't fall off and animals will eat the leaves before eating the tree itself allowing it to survive the winter with less damage done to itself. |
|
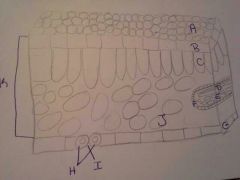
|
A. Cuticle |
|
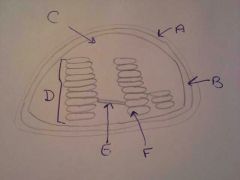
|
A. Outer Membrane |
|
|
What bond is broken in ATP to release energy? |
The bond between the second and third phosphate groups |
|
|
Is ATP rechargable? How? |
yes, it's rechargable through ATP synthase |
|
|
What are the 6 colors of white light? |
red, orange, yellow, green, blue, violet |
|
|
Why do you see certain colors? ie: Why are plants green? |
Pigments absorb all but a certain few lights and those lights are reflected creating the color you see |
|
|
Phosphorylation |
when plants use light energy to add P to ADP |
|
|
Where do the light reactions take place? |
Thylakoid Membrane |
|
|
What are the three things needed for the light reactions? |
H2O, light, and pigments |
|
|
What is Photolysis? |
Splitting of water |
|
|
What is produced? |
ATP, NADPH, O2 |
|
|
What is NADPH and what is it used for? |
It's an electron carrier that carries electrons to be used in the Calvin Cycle |
|
|
Explain what happens in the light reactions. |
PSII loses two electrons as it absorbs light. The electrons become excited and move to a higher energy level, water replaces the electrons that are lost. Electrons jump to electron carriers which form the EC and replace the electrons PI lost, PI's lost electrons turn NADP+ into NADPH As the electrons moved through the ETC they pulled in H+ ions which move across ATP Synthase converting ADP into ATP |
|
|
Where does the Calvin Cycle take place? |
Stroma |
|
|
What are the 3 things needed for it to occur? |
ATP, NADPH, CO2 |
|
|
How many ATP and NADPH are needed? |
9 ATP and 6 NADPH |
|
|
NADPH and ATP are converted into what two rechargable molecules? |
NADP+ and ADP |
|
|
Trace the path of the Calvin Cycle. |
3CO2 join 3RuBP to form 3, 6-C sugars (formed through enzyme rubisco) The 6-C sugars split into 3-C sugars called 3-PGA ATP and NADPH are used to convert 3-PGA into 6G3P One G3P is released, and ATP is used to convert the rest back into 3 RuBP's. The one released G3P molecule is used to make glucose |
|
|
How many turns of the Calvin Cycle does it take to create one 6-C Glucose molecule? |
Two |
|
|
Factors that affect Photosynthesis |
-shortage of water -Temperature -Light Intensity -CO2 concentration |
|
|
What is photorespiration? |
pathway in plants where oxygen is used instead of CO2 |
|
|
When does photorespiration occur? |
when stomates stay closed for a long period of time (dry weather, full sun, high temperatures) |
|
|
What is Cellular Respiration? |
process that releases energy by breaking down glucose, or other organic molecules, in the presence of oxygen |
|
|
What's the overall equation of Cellular Respiration? |
6O2 + C6H12O6 yeilds 6CO2 + 6H2O + Energy (ATP) |
|
|
Structure |
|
|
What is energy? |
The ability to do work |
|
|
What is Glycolysis? |
the process where one molecule of glucose is broken in half, making two molecules of Pyruvate |
|
|
Describe Glycolysis. |
Glucose turns into Frutose 1, 6 Bisphosphate Fructose 1,6 Bisphosphate is turned into two 3 carbon sugar phosphates these molecules are then rearranged into two molecules of pyruvate |
|
|
What is fermentation? |
the breakdown of glucose in the absence of oxygen |
|
|
Alcoholic fermentation |
Pyruvic Acid + NADH yeilds Alcohol + CO2 + NAD+ |
|
|
Alcoholic Fermentation causes bread dough to rise, why? |
The CO2 that's created causes bubbles that make the bread rise |
|
|
Lactic Acid Fermentation |
Pyruvic Acid + NADH yeilds Lactic Acid + NAD+ |
|
|
What are prokaryotes that undergo lactic acid used for? |
To produce some beverages, cheese, yogurt, buttermilk, and sour cream |
|
|
What's an Obligate Aerobe? |
Requires O2 to produce ATP |
|
|
What's an Obligate Anaerobe? |
Do not require oxygen, produce all energy from anaerobic respiration |
|
|
What are Facultativve Aerobes? |
can survive a long time without O2, switch back and forth between Aerobic and Anaerobic Fermentation |
|

|
1. Matrix |
|
|
Describe the Prep Step. |
2, 3-C Pyruvate molecules enter mitochondria. Each is rearranged into Acetyle-CoA One CO2 released per Pyruvate One NADH produced per Pyruvate Acetyle CoA enters Krebs Cycle
|
|
|
Describe Krebs Cycle. |
Acetyle-CoA enters and Acetate binds with Oxaloacetate(4-C) to produce Citrate(6-C) Citrate goes through a series of arrangements, turning into Ketoglutarate(5-C) and eventually back into Oxaloacetate |
|
|
What does one turn of the Krebs Cycle generate? |
2CO2 3NADH FADH2 ATP |
|
|
Describe the Electron Transport Chain. |
NADH and FADH provide electrons that move through transport proteins, pulling H+ ions from matrix into innermembrane space H+ ions then move from high to low concentration through ATP Synthase, creating ATP |
|
|
H+ gradient in photo vs. cellular |
Photo: water molecules split, H+ ions build up Cellular: As electrons move pull H+ ions in from matrix |
|
|
Totals for Aerobic Respiration: |
38 ATP per glucose |
|
|
Total for Anaerobic Respiration: |
2 ATP |
|
|
Somatic Cells |
Body cells that have 46 chromosomes |
|
|
Gametes |
Sex cells: egg and sperm Have 23 chromosomes |
|
|
What decreases as a cell grows and is the cause for cells dividing? |
Diffusion |
|
|
Can cell size vary? |
Yes |
|
|
Is there a limit to how big cells can get? |
Yes |
|
|
Chromatin |
the form of DNA when a cell is not dividing (looks like spaghetti) |
|
|
Which form of DNA is visible in a cell? |
Chromosomes |
|
|
Chromosomes |
form DNA is in when a cell is dividing |
|
|
Why do cells divide? |
-to replace old, worn out cells -to replace injured/damaged cells -for an organism to grow -for reproduction to occur |
|
|
What cells are involved in the cell cycle? |
Somatic Cells |
|
|
What are the three subphases of Interphase? |
G1, S, and G2 |
|
|
What happens in G1? |
The cell grows and prepares for DNA synthesis |
|
|
What happens in S? |
The DNA and chromosomes are copied |
|
|
What happens in G2? |
The organelles are copied and the DNA is checked for errors |
|
|
What is G0? |
A phase for cells that never get too large and never reach the restriction point and have to divide. They will live infinitely unless destroyed at which point they're gone forever |
|
|
What are the three types of G0 cells in the human body? |
Nerve cells, Brain cells, and Heart cells |
|
|
What is the point in G1 phase that when a cell reaches it has no choice but to divide called? |
Restriction Point |
|
|
What are the four phases of Mitosis? |
Prophase, Metaphase, Anaphase, Telophase |
|
|
What happens in prophase? |
DNA coils into chromosomes and the nuclear envelope begins to disappear |
|
|
What happens in metaphase? |
the chromosomes line up along the equator and spindles connect to the centromeres of the chromosomes |
|
|
What part of the centromere do the spindles attach to? |
kinetochore |
|
|
What is the purpose of the centromere? |
it's the glue that holds the two chromatids together |
|
|
What happens in Anaphase? |
The sister chromosome pairs split and begin moving to each side of the cell |
|
|
What happens in Telophase? |
the nucleus begins to reappear and the chromosomes turn back into chromatin |
|
|
What happens in Cytokinesis? |
the cytoplasm pinches off splitting the cell into two |
|
|
What forms in cytokinesis in plant cells? |
A cell plate |
|
|
What does the cell plate turn into? |
The cell wall |
|
|
In cytokinesis which cells are separate and which are not? |
Animal cells, while possibly close together, are separate, while plant cells are not separate cells |
|
|
How do you find the number of chromosomes in a cell? |
count the number of centromeres |
|
|
Chromatid |
a copy of a chromosome |
|
|
What is a telomere? |
A cap at the ends of chromosomes that gets progressively shorter with each cell division, regulating cell division |
|
|
How are cancer cells and telomeres related? |
Cancer cells can reproduce telomeres allowing them to live forever |
|
|
What is cancer? |
the uncontrolled growth and dividing of cells |
|
|
How does cancer start? |
It begins when mutations build up in DNA during the S phase of mitosis |
|
|
What do the mutations cause the cell to do? |
Begin dividing in an uncontrolled way |
|
|
How does a tumor begin to form? |
As cells begin to divide at an uncontrolled rate |
|
|
What is a benign tumor? |
a tumor that doesn't spread |
|
|
What is a malignant tumor? |
a tumor that can spread to other parts of the body |
|
|
What is angiogenesis? |
Cancer sends out signals to trick the body into feeding the tumor through blood vessels |
|
|
What is metastasis? |
the spreading of cancer through the bloodstream |
|
|
What is a carcinogen? |
Anything that causes cancer |
|
|
What are the three main ways cancer is treated? |
Surgery, Radiation, Chemotherapy |
|
|
What is surgery? |
the removal of cancerous cells from the body |
|
|
What is radiation? |
Exposing cancer to radiation -can also kill normal cells |
|
|
What is chemotherapy? |
the use of drugs to kill cancer -can also kill normal cells |

