![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
51 Cards in this Set
- Front
- Back
|
How are biological molecules evidence for evolution |
Both plants and animal cells have similarities in structure All organisms share some biochemistry (contain the same carbon based compounds that interact in similar ways) eg both use DNA and RNA as genetic material |
|
|
What are monomers and polymers |
Monomers are small basic molecular units that can join to other monomers via condensation to make up a polymer |
|
|
Making / breaking a polymer |
Condensation reaction between monomers. Water is released as a chemical bond is formed Hydrolysis reaction breaks chemical bonds between monomers using (adding in) h20 |
|
|
Sugars are |
A general term for monosaccharides and disaccharides |
|
|
Name 3 monosaccharides |
1)glucose (a/b) 2)fructose 3) galactose |
|
|
Draw two isomers of hexose sugar glucose |
Back (Definition)s |
|
|
Which elements to carbohydrates contain |
C ,H ,O |
|
|
Disaccharides are |
Two monosaccharides joined together by a glycosidic bond. Formed in a condensation reaction (h20 released) |
|
|
Name three disaccharides and the monomers that form them |
Lactose (glucose + galactose) Sucrose(glucose+ fructose) Maltose (glucose +glucose) |
|
|
What is a reducing sugar |
Any sugar (mono/disaccharides) able to act as a reducing agent (because it has a free aldehyde/ ketone group) |
|
|
Which sugars are reducing |
All monosaccharides and some disaccharides eg.maltose and lactose |
|
|
What can we use to test for reducing sugars |
Heat sample in a boiling tube with Benedicts reagent in a water bath that’s boiling . Positive sample will turn from blue to green yellow and brick red finally. Higher the concentration of the reducing sugar the further the colour change |
|
|
Testing for non-reducing sugars |
You have to break the non reducing sugar down into monosaccharides (all reducing) . You add dilute HCL heat in boiling water bath and then neutralise using sodium hydrogen carbonate. Then carry out the reducing sugars test (heat with benadicts reagent) |
|
|
Polysaccharides are |
Carbohydrates made from large numbers of their monomers (two or more) join together via condensation reactions . Can also be broken down by hydrolysis. |
|
|
Starch use |
Plants store excess glucose as starch for hydrolysis to glucose when it needs |
|
|
Starch is made up of 2 carbohydrates |
1) amylose - a coiled unbranched (compact for storage) chain of alpha glucose . 2) amylopectin- a highly branched chain of a-glucose that allows a bigger surface area for enzymes to quickly hydrolyse it |
|
|
How is starch adapted to its functikn |
1) insoluble so doesn’t move out of the cell 2) insoluble so doesn’t effect water potential 3)coiled so it’s compact and takes less space up 4) highly branched (big surface area) so enzymes can work quickly to release glucose when it’s needed |
|
|
Glycogen adaption for function |
Used as animal store for energy (excess glucose) 1) It’s compact for storage to take up less space 2) it’s even more highly branched to increase surface area for enzymes to act on |
|
|
Cellulose adaption to function |
Used for structural support in plants eg. Cell wall Made of long unbranched chains of beta glucose Different chains are linked to one another using hydrogen bonds to form strong fibres (microfibril) |
|
|
How do you test for starch |
Add iodine (in potassium iodide) to a sample . It turns from brown/orange to blue/black of starch is present |
|
|
How are lipids different to proteins and carbohydrates |
They are not polymers made from long chains of monomers . They are made from many different components joined together |
|
|
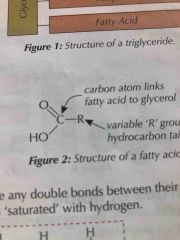
Triglycerides made of |
1)one molecule of glycerol 2)Three fatty acids with long hydrocarbon tails (insoluble) 3) joined together by Ester bonds O-C=O |
|
|
Structure of Fatty acids |
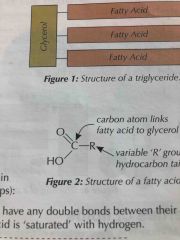
Same basic structure with different hydrocarbon tails (r) |
|
|
Two types of fatty acids |
Saturated fatty acids don’t have any double bonds between their carbon atoms (saturated with hydrogen) Unsaturated do have double bonds between carbons in their chain causing a change in the zig zag shape |
|
|
Triglyceride formation how does it happen |
3 condensation reactions occur , 3 molecules of h20 are released as 3 fatty acids join to glycerol |
|
|
Phospholipids are |
(Same structure are triglycerides except one fatty acid is swapped for a phosphate group) -glycerol -2 fatty acids -phosphate group |
|
|
Triglycerides adaption for function |
Used as an energy store 1)more carbon-hydrogen bonds then c-c these release more energy 2) low mass to energy ration (good for storage) 3) insoluble in water so don’t move out of area and don’t affect water potential 4)high ratio of hydrogen to oxygen atoms so water is released |
|
|
Phospholipids adaption to function |
Make up the bilayer of cell membrane control what enters and leaves a cell Naturally form bilayer The tails face in and heads out so water soluble substances can’t pass through easily Long lipid tails release lots of energy if bonds are broken |
|
|
How to conduct the emulsion test for lipids |
Shake sample with ethanol for 1 minute Pour into water , milky emulsion is indication of lipids More milky means more lipids |
|
|
What are Proteins made up of |
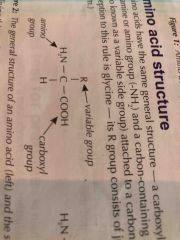
Monomers of proteins are amino acids they join together via peptide bonds in a condensation reaction |
|
|
Di and polypeptides are |
Two amino acids joined together by a peptide bond in a condensation reaction More that two amino acids joined together by a peptide bond in a condensation reaction |
|
|
To form a peptide bond what is lost |
OH from the carboxyl group of one amino acid and the H from the amino group on another amino acid |
|
|
Basic amino acid structur |
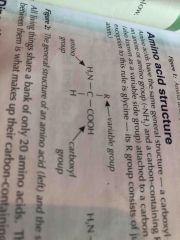
|
|
|
What is the primary structure of a protein |
The sequence of the amino acids in a polypeptide chain |
|
|
Two ways to measure Enzyme activity |
1) how fast the product is made 2) how quickly substrate is broken fown |
|
|
Temperature effect on enzyme activity |
1)As temperatures increases more kinetic energy is given to molecules that move faster increasing likelihood of collisions 2)energy is also more likely to reach activation energy. 3)Until you get above the optimum molecules in the enzyme vibrate so much bonds in the structure break 4) the shape (active site) changes so it can no longer fit to substrate and can’t catalyse it (denatured) |
|
|
ph effect on enzymes activity |
Above and bellow the optimum ph for an enzyme excess h+ and oh- interfere with the hydrogen and ionic bonds holding an enzymes tertiary structure together. The enzyme becomes denatured and the active site changes . |
|
|
Substrate concentration effect on enzyme activity |
More substrates mean a higher rate of reaction as collision frequency is higher. Up to saturation point when all enzymes active sites are occupied so adding more makes no difference. |
|
|
Enzyme concentration |
Increasing it increases rate of reaction until there are more than enough enzymes to deal with the amount of limited substrate then increasing enzyme concentration has no further effect |
|
|
What are Enzyme inhibitors |
Molecules that prevent enzyme activity by binding to the enzymes that they inhibit |
|
|
Competitive inhibitors |
They have a similar shape to the substrate so bind to the active site (no reaction as it doesn’t cause the change) blocking it . Increasing the substrate concentration can counteract this as it’s more likely to bind to enzyme than the inhibitor |
|
|
Non competitive inhibitors |
These bind to an enzyme away from its active site it causes the active site to change shape so the substrate molecules can no longer bind to it Non competitive inhibitor molecules don’t compete with the substrate so increasing substrate concentration has no effect on rate of reaction |
|
|
Secondary structure |
This is the way the gain folds , hydrogen bonds form between amino acids making it into an a- coil or a b- pleated sheet |
|
|
Tertiary structure |
The way in which the polypeptide is further coiled and folded by more hydrogen bonds , ionic bonds and disulphide bridges forming between different parts of the polypeptide. |
|
|
Quaternary structure |
The way different polypeptide chains are held together in a protein made form more than one chain |
|
|
Biurette test for protein |
The test needs to be alkaline so add sample to sodium hydroxide the. Add copper sulphate. It will turn from blue o purple if there is a protein |
|
|
Enzymes are ? |
Biological catalysts They catalyse metabolic reactions at a cellular level (eg.respiration) or for the whole organism eg. Digestipn |
|
|
What is an active site |
The part of an enzyme which has a specific shape complementary to substrates . Enzymes are highly specific (catalyse one substrate reaction) due to tertiary structure. |
|
|
How enzymes speed up reactions |
In a chemical reaction a certain energy is needed before reactions can happen . Enzymes lower this activation energy by forming enzyme substrate complex this can 1)hold to substrates together reducing repulsion and joining them 2)weaken bonds in a substrate so it can break down more easily (less energy required) |
|
|
Lock and key model |
Early model suggests enzymes active sites are perfectly complementary to their substrate. And the enzyme remain unchanged after reaction |
|
|
The induced fit model |
shape of Active Sites are not exactly Complementary, but change shape in the presence of a specific substrate to become Complementary. When a substrate molecule collides with an enzyme, if its composition is specifically correct, the shape of the enzyme's Active Site will change so that the substrate fits into it and an Enzyme-Substrate Complex can form. The reaction is then catalysed and an Enzyme-Product Complex forms. |

