![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
25 Cards in this Set
- Front
- Back
|
What does Imidazole look like?
|
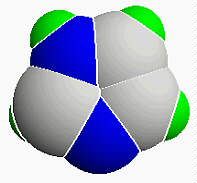
Like this
|
|
|
Metabolite
|
A chemical intermediate in the enzyme-catalyzed reactions of metabolism
|
|
|
Nucleus
|
In eukaryotes, a membrane-bounded organelle that contains chromosomes
|
|
|
Genome
|
All the genetic information encoded in a cell or virus
|
|
|
Eukaryote
|
A unicellular or multicellular organism with cells having a membrane-bounded nucleus, multiple chromosomes, and internal organelles
|
|
|
Prokaryote
|
A bacterium; a unicellular organism with a single chromosome, no nuclear envelope, and no membrane-bounded organelles
|
|
|
Archaebacteria
|
One of five kingdoms of living organisms; includes many species that thrive in extreme enviroments of high ionic strength, high temperature, or low pH
|
|
|
Eubacteria
|
One of five kingdoms of living organisms; They have a plasma membrane but no internal organelles or nucleus
|
|
|
Cytoskeleton
|
The filamentous network providing structure and organization to the cytoplasm; includes actin filaments, microtubules, and intermediate filaments
|
|
|
Stereoisomers
|
Compounds that have the same composition and the same order of atomic connections but different molecular arrangements
|
|
|
Configuration
|
The spatial arrangement of an organic molecule that is conferred by the presence do either (1) double bonds, about which there is no freedom of rotation, or (2) chiral centers, around which substituent groups are arranged in a specific sequence. Configurational isomers cannot be interconverted without breaking one or more covalent bonds
|
|
|
Chiral Center
|
An atom with substituents arranged so that the molecule is not superimposible on its mirror image
|
|
|
Conformation
|
The spatial arrangement of substituent groups that are free to assume different positions in space, without breaking any bonds, because of the freedom of bond rotation
|
|
|
Entropy (S)
|
The extent of randomness or disorder in a system
|
|
|
Enthalpy (H)
|
The heat content of a system
|
|
|
Free-Energy Change (^G)
|
The amount of free-energy released (negative ^G) or absorbed (positive ^G) in a reaction at constant temp and pressure
|
|
|
Endergonic Reaction
|
A chemical reaction that consumes energy (that is, for which ^G is positive)
|
|
|
Exergonic Reaction
|
A chemical reaction that proceeds with the release of free energy (that is, for which ^G is negative)
|
|
|
Equilibrium
|
The state of a system in which no further net change is occuring; the free energy is at a minimum
|
|
|
Standard Free-Energy Change (^G')
|
The free-energy change for a reaction occuring under a set standard of conditions: temp, 298K; Pressur, 1atm or 101.3kPa; and all solutes at 1M concs. ^G' denotes the standard free-energy change at pH 7.0 in 55.5M water
|
|
|
Activation Energy (^G*)
|
The amount of energy (in joules) required to convert all the molecules in 1 mole of a reacting substance from the ground state to the transition state
|
|
|
Catabolism
|
The phase of intermediary metabolism concerned with the energy-yielding degradation of nutrient molecules
|
|
|
Anabolism
|
The phase of intermediary metabolism concerned with the energy-requiring biosynthesisof cell components from smaller precursors
|
|
|
Metabolism
|
The entire set of enzyme-catalyzed transformations of organic molecules in living cells; the sum of anabolism and catabolism
|
|
|
Mutation
|
An inheritable change in the nucleotide sequence of a chromosome
|

