![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
100 Cards in this Set
- Front
- Back
|
Differences between Prokaryotic and Eukaryotic Cells |
Prokaryotic have no nucleus (DNA is unbound in the nucleiod) and no membrane bound organelles |
|
|
Common features of Eukaryotic and Prokaryotic cells |
both have cytoplasm, chromosomes made of DNA, ribosomes, and plasma membranes |
|
|
What does a plant cell have that animal cells do not? |
vacuole, chloroplasts, plasmodesmata, and cell walls |
|
|
The nucleus: Describe and name its functions |
"Info Central" of the cell - regulates all cell activity - contains DNA (chromatin) - enclosed in nuclear envelope (double membrane) - has pores, releasing proteins and regulating what enters and exits |
|
|
Ribosomes: Describe and name their functions |
"Protein Factories" - made of rRNA and proteins - carry out protein synthesis in 2 locations: - in the cytosol (free ribosomes) - outside of ER/nuclear envelope (bound) |
|
|
What organelles does the Endomembrane System consist of? |
the nuclear envelope, golgi, ER, vacuoles, lysosomes, and plasma membrane |
|
|
Endoplasmic Reticulum: Describe and name its functions |
"Membrane Factory" - accounts for more than half the total membrane (continuous with nuclear envelope) Smooth ER: - synthesizes lipids Rough ER: - studded with ribosomes which: - synthesize proteins that enter ER |
|
|
Golgi Apparatus: Describe and name its functions |
"Shipping and Receiving Center" - sorts and packages materials into transport vesicles - modifies ER products - manufactures macromolecules
|
|
|
Lysosomes: Describe and name their functions |
"Digestive Compartments" - sac of hydrolytic enzymes that digest macromolecules - can recycle the cell's own organelles/macromolecules |
|
|
Phagocytosis |
when lysosomes fuse with food vacuoles to digest molecules |
|
|
Autophagy |
an intermolecular degradation system where lysosomes degrade and recycle the cell's own organelles and macromolecules. |
|
|
Vacuole: Describe and name its functions |
"Diverse Maintenance Compartments" - large vesicles derived from the ER and golgi - selective in transporting solutes ex. pumping out excess water |
|
|
Peroxisomes: Describe and name their functions |
"Oxidation" - specialized metabolic compartments - produce hydrogen peroxide and convert it to water - breaks down molecules |
|
|
Mitochondrion: Describe and name its functions |
- the site of cellular respiration - uses O2, sugars, and fats to make ATP - 2 compartments: • intermembrane space • mitochondria matrix |
|
|
Chlorplasts: Describe and name their functions |
- the site of photosynthesis - contain cholorphyll and enzymes • reason for green color in leaves - creates sugars by light energy - contains thylakoids, granum, and stroma |
|
|
Similarities between Chloroplasts and Mitochondria |
Both: - enveloped by double membranes - contain free ribosomes - grow and divide fairly independently |
|
|
What is the Endosymbointic Theory? |
- explains the origin of eukaryotic cells from prokaryotes stating that several key organelles of eukaryotes originated as symbiosis between separate single-celled organisms. - the similarities between chloroplasts and mitochondria lead to this theory |
|
|
Cytoskeleton: Describe and name its functions |
"Support and Motility" - supports cell's shape - interacts with motor proteins for motility
|
|
|
What are the 3 components of the cytoskeleton in thickest to thinnest order? |
1. Microtubules (25nm) 2. Intermediate Filaments (8-12nm) 3. Microfilaments (7nm) |
|
|
Microtubules: Describe and name their functions |
- thickest, 25nm - hollow tubes of α and β tubulin - have polarity in animal cells ~ dynein moves cargo to minus end ~ kinesin moves cargo to plus end - provides motility: • cell: flagella, cilla • movement of chromosomes and organelles |
|
|
Intermediate Filaments: Describe and name their functions |
- 8-12nm - supercoiled keratin proteins - maintains the cells shape - anchorage for organelles |
|
|
Microfilaments: Describe and name their functions |
- 7nm - two intertwined strands of actin proteins - maintains cell's shape - bears tension, muscle contractions - responsible for cell cleavage |
|
|
What is Avagadro's #? |
6.02 x 10^23 |
|
|
what is an acid? |
- an H+ donor - < pH 7
[H3O+][-OH] = 10^-14 pH = -log[H+] [H+] = 10^-pH |
|
|
what is a base? |
- and H+ acceptor or an OH- donor - reduces H+ of a solution - > pH 7
[H3O+][-OH] = 10^-14 pH = -log[H+] [H+] = 10^-pH |
|
|
what does hydrophobic mean? |
- it is non-polar - doesn't react with water |
|
|
what does hydrophilic mean? |
- adheres to water - polar |
|
|
What are the 3 isomers? |
1. Structural Isomers: diff. covalent arrangements
2. Cis-trans isomers: same covalent bonds, diff. in spatial arrangements
3. Entantiomers: mirror images of each other |
|
|
What are the 7 functional groups? |
- hydroxyl - carbonyl - carboxyl - amino - sulfhydryl - phosphate - methyl |
|
|
What functional groups are charged? |
- Carboxyl - Amino - Phosphate |
|
|
What functional groups are hydrophilic? |
- Hydroxyl - Carbonyl - Carboxyl - Amino - Phosphate |
|
|
What functional groups are hydrophobic? |
- Methyl - Sulfhydryl |
|
|
Hyrdoxyl Group |
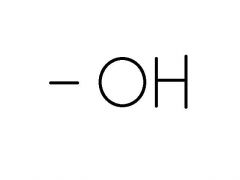
- alcohols - forms hydrogen bonds w/ water - helps dissolve organic compounds like sugar |
|
|
Carbonyl Group |
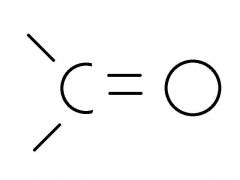
- Ketones (only when with a carbon skeleton) - Aldehydes (only if at the end of carbon skeleton) |
|
|
Carboxyl Group |
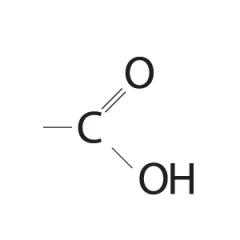
- Carboxyl acids (organic acids) - acts as an acid, donating H+ - found in ionized form in cells as carboxylate ion (-1) |
|
|
Amino Group |
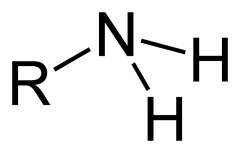
- Amines - acts as a base, accepting H+'s - found in cells in ionized form (+1) |
|
|
Phosphate Group |
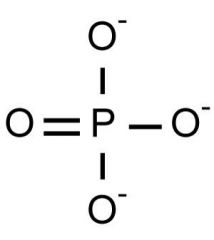
- organic phosphates - contribute negative charge to part of molecule it's on - have potential to react w/ water, releasing energy |
|
|
Methyl Group |
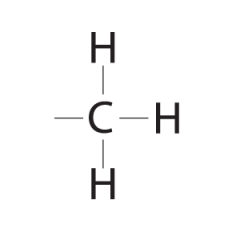
- Methylated compounds - affects expression of genes - arrangement in male/female sex hormones affects their shape/funct. |
|
|
Sulfhydryl Group |

- Thiols - can react with each other to for covalent bonds (cross-linking) -helps stabilize proteins |
|
|
What happens when a dehydration reaction occurs? |
- a polymer is synthesized - 2 monomers bond together through the loss of a water molecule |
|
|
What happens when a hydrolosis occurs? |
- a polymer is broken down - a water molecule is aded, breaking a bond |
|
|
What is a monosaccharide? |
- a single sugar - usually multiples of CH2O - classified by the location of the carbonyl group |
|
|
What are the 2 monosaccharides? |
- Aldose (Aldehyde)
- Ketose (ketone) |
|
|
What are polysaccharides and their roles? |
- polymers of sugars - have storage and structural roles - STORAGE: ~ starch in plants ~ glycogen in liver/muscle cells - STRUCTURAL: ~ cellulose, major component of cell walls (polymer of glucose) |
|
|
What are disaccharides? |
- formed when dehydration joins 2 monosaccharides ( glucose + galactose = lactose ) ( glucose + fructose = sucrose ) |
|
|
What is the role of an Ester Linkage? |
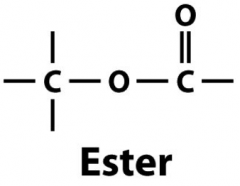
- links 3 fatty acids and glycerol to make trygliceride |
|
|
What is an example of a saturated fatty acid and its characteristics? |
- has the max # of H possible - has no double bonds - is solid at room temp. ex. butter |
|
|
What is an example of an unsaturated fatty acid and its characteristics? |
- has one or moor double bonds - liquidy ex. olive oil |
|
|
what is a phospholipid and its characteristics? |
- two fatty acids and a phosphate group attached to glycerol - has a hydrophilic head and hydrophobic tail - creates a bilayer when in water (cell membrane) |
|
|
what are the 4 levels of a protein's structure? |
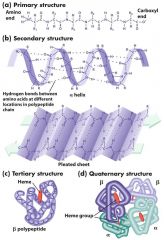
- Primary - Secondary: results from H bonds, backbone interaction - Tertiary: R-group interactions, disulfide bridges support structure - Quatenary: interactions of 2 or more polypeptides |
|
|
Collagen |
- most abundant protein, holding us togethe - 2 polypeptides coiled like rope |
|
|
Hemoglobin |
- 4 polypeptides - 2 alpha, 2 beta chains |
|
|
What nucleic acids do DNA and RNA each have? |
DNA: cytosine, thymine, guanine, and adenine C--->G A--->T RNA: cytosine, uracil, guanine, and adenine C---->G A---->U |
|
|
Name 3 pyrimidines |
cytosine, thymine, and uracil |
|
|
Name 2 purines |
adenine and guanine |
|
|
what does amphipathic mean? |
a molecule has both hydrophilic and hydrophobic regions |
|
|
what are the characteristics of the fluid mosaic model? |
a cell's membrane is a fluid structure with a mosaic of various proteins |
|
|
what is cholesterol's role in a cell's membrane? |
is a membrane fluidity "buffer" it either maintains or prevents fluidity depending on the temp. |
|
|
Peripheral Proteins |
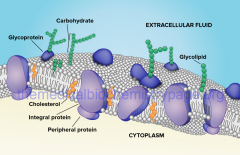
bound to surface or membrane inside cell
horizontal looking like the way you see your peripherals |
|
|
Integral Protein |
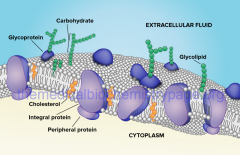
penetrates to the hydrophobic core
integrated into bilayer |
|
|
Membrane Protein Functions |
- transportation - enzymatic activity - signal transduction - cell-cell recognition -intercellular joining -attachment to cytoskeleton |
|
|
two transport proteins |
- channel proteins • move certain molecules through a hydrophilic channel
- carrier proteins • bind to molecules and change shape to shuttle them across the membrane |
|
|
passive transport |
requires no energy to diffuse a substance across a membrane ex. diffusion |
|
|
active transport |
requires energy ex. Sodium Potassium Pump, H+ against concentration gradient |
|
|
concentration gradient |
region along which the density of a chemical substance increases or decreases |
|
|
Osmosis |
diffusion of water across a selectively permeable membrane low ----> high concentration |
|
|
Isotonic, Hypotonic, Hypertonic |
Iso - at an equilibrium Hypo - inside > outside, bloated Hyper - inside < outside, shriveled up |
|
|
Facilitated diffusion |
transport proteins that speed the passive movement |
|
|
Laws of Thermodynamics |
Energy cannot be created nor destroyed.
Every energy transformation increase entropy.hO |
|
|
Redox Reactions |
Oxidation Is Losing
Reduction Is Gaining |
|
|
steps of Cellular Respiration |
-Glycolosis - Citric Acid Cycle - Oxidative Phosphorylation - Electron Transport Chain - Chemiosmosis |
|
|
Glycolosis |
- happens in cytosol of mitochondria - doesn't require O2 - NAD+ is oxidizing agent - breaks down glucose into 2 pyruvates - energy investment phase: 2 ATP used - energy payoff phase: 4 ATPs formed - NET: 2 pyruvate, 2 ATP, 2 NADH |
|
|
Citric Acid Cycle |
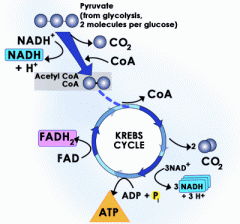
- occurs in the matrix of the cell - completes the break down of glucose - happens in presence of O2 - 2 pyruvate converted to 2 acetyl CoA - NET per turn: 1 ATP, 3 NADH, 1 FADH2 - NET outcome: 2 ATP, 8 NADH, 6 CO2, 2 FADH2 |
|
|
Oxidative Phosphorylation: Electron Transport |
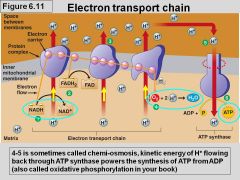
- powers ATP synthesis - doesn't generate ATP directly - won't function w/o O2 - NADH and FADH2 donate electrons to chain - electron transfer causes protein complexes to pump H+ into intermembrane space |
|
|
Oxidative Phosphorylation: Chemiosmosis |
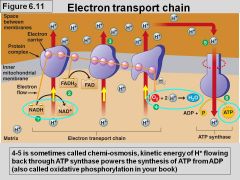
- "Energy Coupling Mechanism" - the use of energy in a H+ gradient to drive cellular work - creates proton gradient - H+ is moved back across the membrane (ATP synthase) - the exergonic flow of H+ is used to drive phosphorylation of ATP |
|
|
two types of Fermentation |
alcohol fermentation: pyruvate converted to ethanol, releasing CO2
lactic acid fermentation: pyruvate reduced to NADH with lactate as end product |
|
|
Fermentation vs Cellular Respiration |
O2 requirements Fermentation: No Respiration: Yes
different electron acceptors Fermentation: pryuvate (2 ATP) Respiration: O2 (32 ATP)
|
|
|
Photosynthesis |
- converts solar energy to chemical energy - outcomes: sugar, O2, and water - chloroplasts split H2O, releasing O2 - has 2 stages: Light reactions: PS II and PS I Non-light reactions: Calvin Cycle |
|
|
Photosystem II and I |
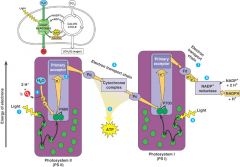
PS II functions first - absorbs wl 680 nm - reaction center: P680 PS I - absorbs wl 700 nm - reaction center: P700 • linear electron flow produces ATP and NADPH • electrons of NADPH available for Calvin Cycle |
|
|
chemiosmosis in cholorplasts vs mitochondria |
- both generate ATP but use different energy sources Mitochondria - pumps protons to intermembrane space driving ATP synthesis - pumped back into matrix Chloroplasts - pumps protons to thylakoid space - drives synthesis - pumped back into stroma |
|
|
Calvin Cycle |
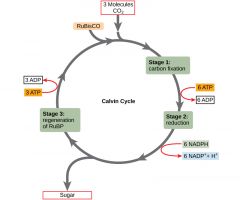
- use chem. energy to reduce CO2 to sugar - C enters as 3CO2, leaves as sugar (G3P) 3 phases: 1. Carbon fixation (catalyzed (rubisco)) 2. Reduction 3. Regeneration of CO2 acceptor (RuBP)
1 G3P = 3 cycle runs
|
|
|
Meiosis |
result in 4 daughter cells each with only half the chromosomes of parent cell |
|
|
Meiosis I |
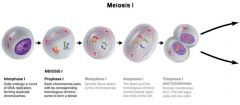
- homologs pair up and split (2 haploid daughter cells) Prophase I - duplicated homologs pair and exchange segments (crossing-over) Metaphase I - chromosomes line up by homolog pairs Anaphase I - each pair of homologs separates Telephase I and ctyokinesis - 2 haploid cells form, both have 2 sister chromatids |
|
|
Meiosis II |
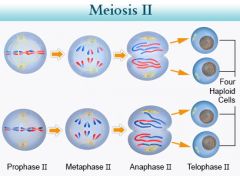
(similar to mitosis) Prophase II - spindle apparatus form Metaphase II - sister chromatids lined at metaphase plate - not identical due to cross-over Anaphase II - chromatids separate - two newly individual chromosomes Telephase II and ctyokinesis - nuclei form, cytoplasm separates - 4 daughter cells |
|
|
Cell Cycle |
Interphase: cell growth and copying in prep. for cell division G1 - (2n) Cells increase in size S - (4n) DNA replication occurs G2 - (4n) Mitososis: (2n and 2n) division into two daughter cells |
|
|
Mitosis |
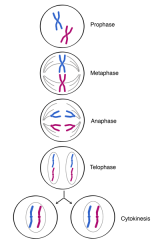
Prophase - spindles form, chromatids dense Prometaphase - spindles attiMach Metaphase - sister chromatids line at metaphase plate Anaphase - chromosomes split Telophase and Cytokinesis - cleavage furrow |
|
|
Mitosis vs Meiosis |
mitosis = identical to parent cells meiosis = differ from parent cells |
|
|
Mendel and genetics |
Garden pea experiment |
|
|
The law of segregation |
anaphase I |
|
|
the law of independent assortment |
metaphase I |
|
|
Griffith |
bacteria and mice |
|
|
Avery, Macleod, and Maccarthy |
Griffith's bacteria |
|
|
Chargaff |
analyzed DNA compostion |
|
|
Hershey and Chase |
viruses infecting bacteria (phages) radioactive S and P |
|
|
Watson and Crick |
DNA replication |
|
|
Meselson and Stahl |
Conservative, dispersive, and semiconservative |
|
|
DNA Replication steps |
unwinding initiation elongation termination |
|
|
Central Dogma |
DNA ----> RNA -----> Protein |
|
|
Start codon |
AUG |
|
|
Stop codons |
UAA, UAG, UGA |

