![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
49 Cards in this Set
- Front
- Back
|
The Lewis structure for Ammonium
|
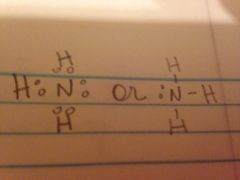
|
|
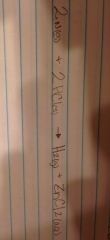
|
Single Displacement
|
|
|
Around the nucleus; have a negative charge
|
Electrons
|
|
|
In the nucleus; positive charge
|
Protons
|
|
|
In the nucleus; neutral charge
|
Neutrons
|
|
|
Phosphorus pentachloride
|
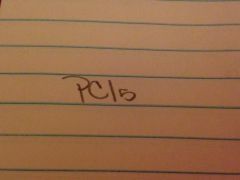
|
|
|
Chemical bonds can be classified as 100% ionic, covalent, or metallic
|
False
|
|
|
The type of formula that is the simplest ratio in compound formula unit
|
Empirical Formula
|
|
|
A Molecule like methane with four bonding pairs of electrons has what kind of geometry
|
Tetrahedral
|
|
|
Intermolecular force responsible for the unexpected high boiling point of water
|
Hydrogen Bond
|
|
|
Very hard substances such as diamond, silicon carbide, quartz
|
Network Covalent
|
|
|
The body temperature of a human averages 98.6 degrees Fahrenheit, what temperature is this in Celsius to the third sig fig
|
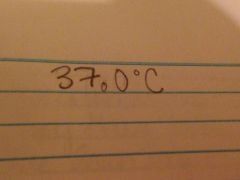
|
|
|
Of these-Sulfur, Calcium, Chlorine, Cesium- which one has the greatest electronegativity
|
Chlorine
|
|
|
Outermost electrons in an atom that are involved in chemical bonding
|
Valence
|
|
|
Magnesium, Calcium, Barium
|
Alkaline Earth Metals
|
|
|
Boron, Silicon, Germanium
|
Metalloids
|
|
|
Fluorine,Bromine , Iodine
|
Halogens
|
|
|
States that light consists as bundles or packets of energy
|
Quantum theory of light
|
|
|
Individual packets of light
|
Photons
|
|
|
The mass of a substance per unit of volume
|
Density
|
|
|
water having two bonding pairs and two non-bonding pairs
|
Angular
|
|
|
A molecule described as a dipole is characterized by
|
Negative and positive ends
|
|
|
Gas that adheres directly to the gas laws
|
Ideal gas
|
|
|
Bonding situation in which a Lewis structure cannot accurately represent the arrangement of valence electrons
|
Resonance
|
|
|
Not true about exothermic
|
Heat energy is absorbed
|
|
|
The volume of three moles of an ideal gas under stp would be
|
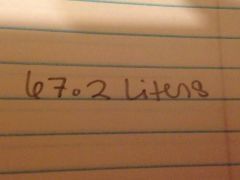
|
|
|
The amount of heat required to raise the temperature of one gram of a substance by one degree Celsius
|
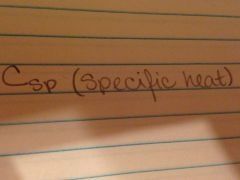
|
|
|
Absolute zero in Celsius
|
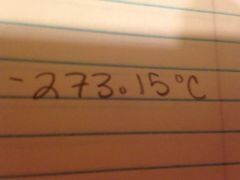
|
|
|
The speed of light in a vacuum
|
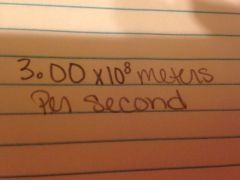
|
|
|
Covalent bond in which the bonding electron pair is pulled closer to the more electronegative element
|
Polar covalent
|
|
|
The second law of thermodynamics
|
The entropy of the universe always increases
|
|
|
Transfer of electrons from one atom to another
|
Ionic
|
|
|
Produces solids that are malleable and ductile
|
Metallic
|
|
|
Atoms of the same element can differ in...
|
The number of neutrons they have
|
|
|
The simplest repeating unit in a crystal
|
The simplest repeating unit in a crystal
|
|
|
Solid whose particles are characterized by regular repeating thee dimensional pattern
|
Crystalline solid
|
|
|
The bright colored lines in hydrogen
|
Electrons dropping from higher to lower
|
|
|
Kinetic energy is associated with an objects..
|
Motion
|
|
|
Two polar bonds that point symmetrically in opposite directions
|
Non-polar
|
|
|
Characterized by the sharing of valence electrons forming a "sea"
|
Metallic
|
|
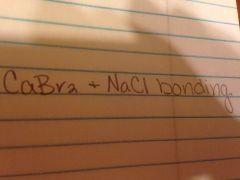
|
Ionic
|
|
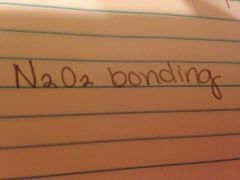
|
Covalent
|
|
|
Bonding characterized by the sharing of one or more electron pair between two atoms
|
Covalent bond
|
|
|
Type of chemical bond between metal and non-metal atoms
|
Ionic bond
|
|
|
States that position and momentum of particles cannot be precisely measured
|
Uncertainty principle
|
|
|
Amount of heat required to melt once gram of a substance at it's melting point
|
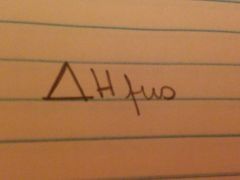
|
|
|
How many mm are in 2.54cm
|
25.4 mm
|
|
|
Absolute zero in Kelvins
|
0 Kelvins
|
|
|
Three ways in which compounds differ from mixtures
|
1)compounds are chemically combined and mixtures are physically combined;2) compounds are pure substances and mixtures are not;3) compounds can only be separated by chemical means and mixtures can be separated by physical processes
|

