![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
29 Cards in this Set
- Front
- Back
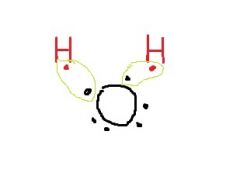
Why do the H atoms in water form a bent molecule instead of in a straight line? |
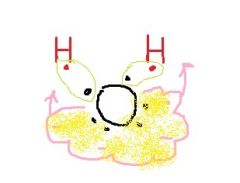
The unpaired electrons tend to push them away. |
|
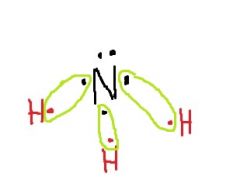
What shape does ammonia have? |
trigonal pyramidal |
|
|
What does the dot diagram for hydrogen look like? |
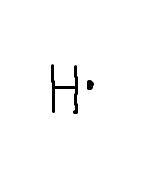
|
|
|
What does the dot diagram for oxygen look like? |

|
|
|
What does the dot diagram for fluorine look like? |

|
|
|
What does the dot diagram for nitrogen look like? |
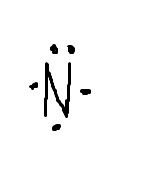
|
|
|
What does the dot diagram for carbon look like? |
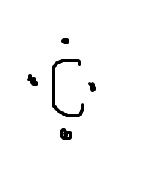
|
|
|
What does the dot diagram for chlorine look like? |

|
|
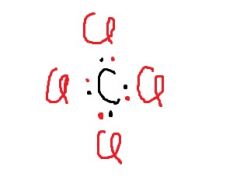
What shape is CCl₄ likely to have? |
tetrahedral |
|
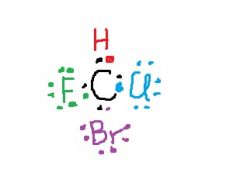
What shape is CHClBrF like to have? |
tetrahedral |
|
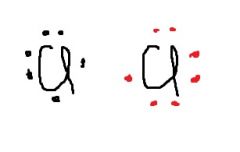
A ___ ____ bond is likely to exist between two chlorine atoms. How many electrons shared? |
single covalent; two shared |
|
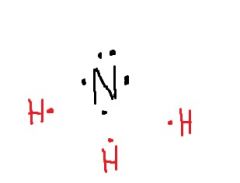
How will nitrogen bond to the 3 H atoms? How many unshared electron pairs will there be? |
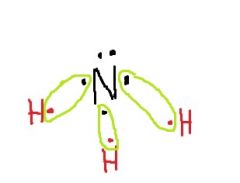
Three single covalent bonds; One unshared electron pair |
|
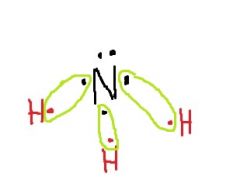
What does the unshared electron pair tend to do? |
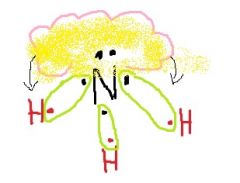
Push away the electron clouds on the hydrogen, making bond angle with each other smaller. |
|
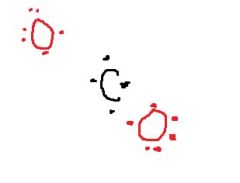
When CO₂ forms, two _____ ____ bonds must happen between the Oxygens and the Carbon. |
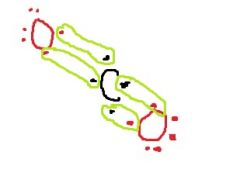
double covalent |
|

When the C bonds to N in this molecule, a ____ ____ bond must form. It consists of ___ electron pairs for a total of ___ electrons |
triple covalent 3 (pairs) 6 (electrons) |
|
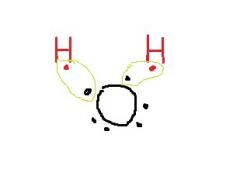
What shape is this? |
bent |
|
|
What does the dot diagram for sulfur look like? |

|
|
|
VSEPR stand for V__ S__ E__ P__ R__. |
Valence Shell Electron Pair Repulsion |
|
|
"Valence Shell" refers to.... |
the outermost s & p electrons |
|
|
"Electron Pair" means.... |
two non-singlet electrons together that are not in a bond with another atom |
|
|
"Repulsion" means.... |
negative electron clouds pushing away from each other |
|

What shape? |
tetrahedral |
|

What shape? How many unpaired electron pairs? |
trigonal pyramidal 1 pair |
|

This is an N₂ molecule. What shape? |
Linear |
|

How many electrons are being shared in this N₂ molecule? Thus it has a ___ ___ bond. |
6 triple covalent |
|

This represents HCl. What shape? How many unshared electron pairs are there? |
linear 3 pairs |
|

This represents H₂. What shape? How many unshared electron pairs? |
Linear No unshared pairs |
|

This represents CO₂. How many electrons does each O share with C? Thus there are two ___ ___ bonds |
4 each double covalent |
|

This represents water. What shape is it? How many unshared electron pairs does it have? |
Bent 2 pairs |

