![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
28 Cards in this Set
- Front
- Back
|
list 4 common features of immune responses against tumors and transplants:
|
1.) reactions to human cells perceived as foreign
2.) antigenic targets can be expressed elsewhere in the body 3.) requires special mechanisms for induction 4.) central is CTL |
|
|
Histopathologic evidence of immune response against tumors
|
lymphocytic infiltrates around tumors and enlargement of draining lymph nodes = better prognosis
therefore: immune response against tumors limit growth to tumor |
|
|
Experimental evidence of immune response against tumors:
|
Transplants of a tumor are rejected by animals previously exposed to that tumor; immunity to tumor transplants can be transferred by lymphocytes from a tumor-bearing animal
Therefore: tumors rejection shows features of adaptive immunity (specificity, memory) and is mediated by lymphocytes |
|
|
clinical and experimental: immunodeficient individuals have increased incidence of tumor types
|
immune system protects against growth of tumors ("immune surveillance")
|
|
|
what needs to happen for tumor antigens to trigger immunity?
|
mutations, over-expression, and aberrant expression (location or timing)
|
|
|
what are types of tumor antigens recognized by T cells? (4 type with examples)
|
1.) mutated self protein: (mutated proteins in melanomas)
2.) product of oncogene or ts gene: (oncogene products mutated RAS, Bcr/Abl fusion proteins) ts gene: mutated p53 protein 3.) overexpressed or aberrantly expressed self protein: (tyrosinase, Mart proteins in melanomas) 4.) oncogenic virus: (human papillomavirus e6, e7 proteins in cervical carcinoma) |
|
|
infection and tumor immunity: 3 immunodeficiencies and 2 inflammation examples resulting in cancer:
|
Immunodeficiencies: HPV (cervical carcinoma), EBV driven leukemia following transplantation; HIV-kaposi sarcoma
inflammation: hepatitis B (hepatocellular carcinoma); helicobacter pylori: gastric carcinoma |
|
|
induction of cd8 T cell responses against tumors: what are the two phases:
|
1.) induction of anti-tumor T cell response (cros priming)
2.) effector phase of antitumor CTL response |
|
|
3 ways to enhance anti-tumor immunity:
|
1.) engage the APC's:
2.) vaccinate against tumor antigens 3.) combination therapies (Chemo + Immuno): damage vasculature, improve access; inflammation; initial tumor cell death feeds the APCs |
|
|
3 ways in which tumors evade immune responses:
|
1.) failure to produce tumor antigen (antigen-loss variant of tumor cell: lack of T cell recognition)
2.) mutations in MHC gene or genes needed for antigen processing: Lack of T-cell recognition 3.) production of immuno-suppressive proteins: inhibition of T-cell activation |
|
|
list three strategies for cancer immunotherapy: (3 types of vaccinations)
|
1.) dendritic cell pulsed with tumor antigen
2.) plasmid expressing cDNA encoding tumor antigen 3.) tumor cell transfected with gene for lymphocyte costimulator (B7/ Il-2): B7: expressing tumor cell stimulates tumor T cell; IL-2 enhances proliferation and differentiation of tumor t cell |
|
|
3 pieces of Evidence for immune responses to transplants:
|
1.) prior exposure to donor MHC molecules accelerated graft rejection
2.) ability to reject a graft can be transferred to a naive individual by lymphocytes 3.) depletion or inactivation of T lymphocytes by drugs: reduced graft rejection |
|
|
transplant vocab: define syngeneic, autologous, allogeneic and xenogeneic:
|
syngeneic: from identical individual
allogeneic: from different individual/ sames pecies autologous: same individual is donor and recipient xenogeneic: different species |
|
|
How many MHC class I and Class II molecules are there and which are polymorphic?
|
6 class I; six class II:
350 HLa-a; 620 HLA-b; HLA c is limited 90 HLA-Dq, 400 HLA-DR; HLA-DP is limited polymorphism |
|
|
what is minor histocompatibility:
|
allelic forms of normal cell proteins
important in blood transfusion |
|
|
What is direct recognition of organ allografts?
|
T cell recognizes unprocessed allogeneic MHC molecules on graft APCs
|
|
|
4 mechanisms of action of antibody immunotherapy in cancer
|
1.) Upper left: direct cytotoxicity, in which mAbs can induce direct cytotoxicity in tumor cells by perturbing oncogenic signaling pathways or in which immunoconjugates can carry cytotoxic agents to targeted cells. Lower left: FcR-mediated immune effector engagement, in which the Fc portion of mAbs can engage immune effector functions, including soluble CMC (through the membrane attack complex MAC) as well as NK cells, macrophages, and dendritic cells, through FcRs, allowing for ADCC, ADCP, and IC uptake. Upper right: Nonrestricted activation of cytotoxic T cells, in which tumor-infiltrating CTLs can be activated against tumor cells—independent of T cell receptor (TCR) specificity—by engaging coreceptors on the T cells and tumor antigens. Lower right: blockade of inhibitory signaling, in which cytotoxic lymphocytes, including NK cells and CTLs, express inhibitory receptors for various ligands that may be expressed by tumor cells. Antagonistic antibodies that target these inhibitory receptors can block ligand-receptor interactions so that targeted cytotoxicity can ensue. These four strategies enhance tumor cell death, which can promote phagocytosis of tumor cell antigens, and induction of adaptive immune responses (bottom right) in two ways: MHC class I cross-presentation and priming of cytotoxic T cells and MHC class II presentation and priming of helper T cells. These adaptive immune responses can lead to enhanced—and possibly persistent—antitumor immunity.
|
|
|
What is indirect recognition of organ allografts?
|
T-cell recognizes processed peptide of allogeneic MHC molecule bound to self MHC molecule on host APC
|
|
|
Three classes of graft rejection:
|
hyperacute (occurs in minutes; thrombosis of graft vessels; necrosis of graft)
acute: occurs in days or weeks; cell damage and inflammation chronic: occurs over months and years; fibrosis, arteriosclerosis, gradual loss of function of grafted tissue |
|
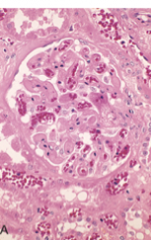
What is this a pic of?
|
hyperacute graft rejection: endothelial damage and thrombi in glomerolus
|
|
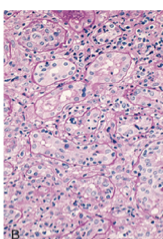
what is this a pic of?
|
acute cellular rejection: inflammatory cells in interstitium and between epithelial cells
|
|
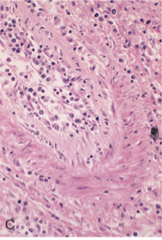
What is this a pic of?
|
acute humoral vasculitis: inflammatory cells and proliferating smooth muscle
|
|
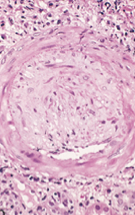
what is this a pic of?
|
chronic: arteriosclerois: accumulation of smooth muscle cells and connective tissue
|
|
|
Prevention, treatment and future of graft rejection ( two each):
|
prevention: MHC match (major and minor)
treatment: T cells: immunosuppression future: tolerance induction and specificity for graft |
|
|
Transplantation of Blood and marrow: transfusion reaction involves ____ but not ____
|
pre-formed antibodies but not T cells
|
|
|
For hematopoeitic stem cell transplantation, there are 3 components to prepare patient and 3 to prepare transplant. List:
|
1.) matching, 2.) make space 3.) immunosuprresive
1.) stem cell enrich 2.) t cell deplete 3. purge of malignant cells |
|
|
What is graft versus host disease? What is acute versus chronic and time frame for each
|
-T cells present in donor cell attack recipient
-acute: occurs within 60 days and CTLs attack skin, liver, and gut chronic :occur after 100 days and increased fibrosis |
|
|
What is the bispecific antibody strategy? Name components against t cell and against tumor cell
|
anti-cd3 and anti EGFR (EGFRBi): bind to t cell and lung tumor cell
|

