![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
33 Cards in this Set
- Front
- Back
|
Essay structure |
Introduction Diagnosis, Measuring poison levels, Antidote, Management Paracetamol poisoning Metabolism, Overdose mechanism,Symptoms, Prevention, Treatment Salicylate (aspirin) Poisoning Symptoms, Poisoning effects,Treatment |
|
|
What is Toxicology? |
interactionbetween a foreign chemical and a biological system that results in damage to a livingorganism |
|
|
Who gets Toxicology
|
· Intrauterine, neonates – poor metabolic andexcretory capacity, infants – accidental, childhood – substance abuse, adults –suicide – CO, aspirin. |
|
|
Diagnosis |
· Clinical features depend on: route of exposure,duration, time since exposure, age and medical background Always test: renal function (serum urea/electrolytes),hepatic function (liver tests), blood glucose (to exclude hypoglycaemia) andacid-base status (blood gases |
|
|
Measuring poison levels |
· Not useful for most poisons as levels will notalter treatment · Useful for paracetamol, Li, CO, ethanol,salicyclate. · Use: prognosis, diagnosis of coma, confirmationof brain death |
|
|
Antidote |
· Minority. Used for paracetamol, snake bites. · Active elimination: haemodialysis used forsalicyclates, ethylene glycol, methanol and also to support renal function whencompromised |
|
|
Management |
· Acute: establish airways, cardiopulmonaryresuscitation and ventilator support · Maintain circulation and ECG – dysrhythmias arecommon · diazepam given if there are repeated orprolonged convulsions · Repeated doses of activated charcoal · maintain core body temperature as hypothermiacan complicate poisoning with sedatives or antidepressants · monitor renal function as very important forrenal excretion of poison |
|
|
Paracetamol poisoning - therapeutic limit, risk factors |
· upper therapeutic limit is 8 g/day in divideddoses · large individual to individual variation inmetabolism 20g fatal, 50g not fatal · Children – relatively resistant to hepatotoxicityand doses low · Risk factors – chronic alcohol intake, fastingand anorexia |
|
|
Metabolism |
· rapidly absorbed from upper GI tract · glucoronide and sulphated conjugates excreted · Cytochrome P450 enzyme converts paracetamol toNAPQI · most NAPQI conjugated to glutathione andexcreted · remainder can be directly hepatotoxic andnephrotoxic by binding to sulfhydryl groups |
|
|
Overdose mechanism |
· sulfate and glucuronide pathways becomesaturated, more shunted to cytochrome P450 enzyme (CYP 2E1) - convertsparacetamol to N-acetyl-p-benzoquinoneimine (NAPQI)· mostNAPQI conjugated to glutathione and excreted, depleting the liver’s naturalantioxidant glutathione · conversion pathways are saturable - higher dose,greater damage · induction of P450 enzymes (chronic alcoholics,patients of phenytoin, phenobarbital) can cause lower paracetamol doses to havetoxic effects |
|
|
Diagram for paracetamol poisoning |
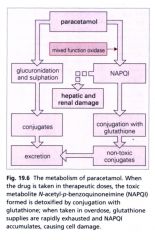
|
|
|
Symptoms |
· may initially be absent · by 24-36 hrs there may be pain in right hypochondriumand a rise in unconjugated bilirubin by 20 hrs – conjugation of bilirubin inliver – index of liver damage. Also, increases ALT and AST levels. · prothrombin time is extended by 1 sec for everyhour since ingestion from 24 hrs· rising prothrombin time after 48 hrs indicatessever hepatotoxicity · signs of hepatic encephalopathy after 48 hrs,death after 5-6 days · plasma levels useful for prognosis is assessedafter 4 hours · levels also used as a guide for treatment |
|
|
Prevention |
· Selling parametamol pre-combined with an emetic(causes vomiting) or antidote. |
|
|
Treatment- overall |
removing drug and replacing glutathione |
|
|
What is more important - · antidote emptying stomach or providing activated charcoal |
Antidote |
|
|
Explain antidote
|
N-acetylcysteinegiven by IV in dextrose solution - rehydrates and treats hypoglycaemia due toliver damage – acts as a precursor for glutathione and can neutralise NAPQI · increases hepatic glutathione production, mayalso aid repair of oxidative damage |
|
|
Explain Activatedcharcoal |
· decreases absorption of paracetamol. Most benefitbetween 30 mins and 2hrs of absorption. However, 40% less antidote is absorbed. |
|
|
how is recovery monitored |
· Check serum creatinine and prothrombin time tomonitor recovery
|
|
|
Serious cases |
· liver transplantation in serious cases. Acidemiais best indicator of probable mortality and need for transplantation.
|
|
|
What is Salicylate metabolised to? |
salicylicacid (active) |
|
|
Compare to paracetamol - side effects |
Moretoxic side effects |
|
|
Symptoms |
· Nausea and vomiting, increased rate and depth ofrespiration (hyperventilation – respiratory alkalosis then metabolic acidosis),sweating, and tinnitus. · Normally volume depleted. · Consciousness initially maintained butconfusion, disorientation, and loss of consciousness may occur in severpoisoning with poor prognosis · Metabolic disturbances - combined compensatedmetabolic acidosis and respiratory alkalosis |
|
|
Poisoning effects- hyperventilation |
Hyperventilation resulting from direct respiratorycenter stimulation, leading to respiratory alkalosis. |
|
|
How is hyperventilation compensated for? |
· Compensated for and eventually overcome by themetabolic acidosis due to acid ingestion and intermediary metabolism effects: - increased lipolysis – increases ketones- inhibition of TCA cycle – increases lactic acid andpyruvate - increased proteolysis increases amino acids - All lead to non-respiratory acidosis |
|
|
Another effect |
· Oxidative phosphorylation uncoupling.
|
|
|
Explain · Oxidative phosphorylation uncoupling. |
- Increased oxygen consumption and CO2production – stimulates respiratory system - Increased ATP demand – increases glycolysis and gluconeogenesis - Hyperprexia (high fever) – sweating – dehydration |
|
|
Explain Vomiting |
· dehydration and electrolyte imbalance |
|
|
Treatment
|
Gastric lavage IVdextrose · No antidote HCO3- for significant ODs Activated charcoal Haemodialysis |
|
|
Explain Gastric lavage |
· (stomach pumping). Followed by 50 g oralactivated charcoal then 25 g every 4 hrs. No longer routinely used soconsidered if patient ingested lethal amounts |
|
|
Explain IVdextrose |
· Volume depleted so rehydrate and correctelectrolyte imbalances - IV dextrose (corrects hypoglycaemia) and normalsaline. Increase urinary output. |
|
|
Explain HCO3- |
significantODs. Salicylates pass into filtrate. Reabsorbed if salicylic acid is notionised. Given to ensure basic urine, pH 7.5-8. |
|
|
Explain Activatedcharcoal |
· absorbs the aspirin in GI tract. |
|
|
Explain Haemodialysis |
enhancesremoval from blood for severe OD. Also restores electrolyte and acid-baseabnormalities. |

