![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
15 Cards in this Set
- Front
- Back
|
Reactant |
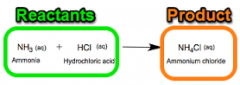
A reactant is a substance that takes part in and undergoes change during a reaction. |
|
|
Product |
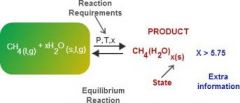
A product is a substance that is formed as the result of a chemical reaction. |
|
|
Catalyst |
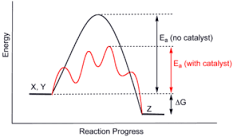
A catalyst is a substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change. |
|
|
Inhibitor |
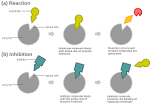
A inhibitor is a substance that slows down or prevents a particular chemical reaction or other process, or that reduces the activity of a particular reactant, catalyst, or enzyme. |
|
|
Rate of Reaction |
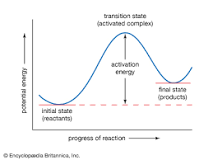
Reaction rate is the speed at which a chemical reaction proceeds. It is often expressed in terms of either the concentration of a product that is formed in a unit of time or the concentration of a reactant that is consumed in a unit of time. |
|
|
Conservation of Mass |
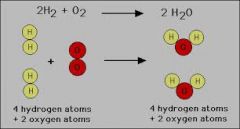
Conservation of Mass is a principle stating that mass cannot be created or destroyed. |
|
|
Decomposition Reaction |
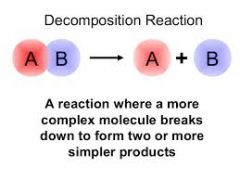
Chemical decomposition is a analysis or breakdown is the separation of a chemical compound into elements or simpler compounds. |
|
|
Synthesis Reaction |
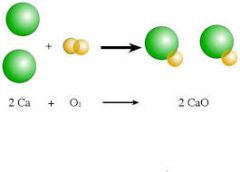
A synthesis reaction or direct combination reaction is a type of chemical reaction in which two or more simple substances combine to form a more complex product. The reactants may be elements or compounds. The product is always a compound. |
|
|
Precipitation Reaction |

A precipitation reaction is an insoluble solid that emerges from a liquid solution. The emergence of the insoluble solid from solution is called precipitation. |
|
|
Single-displacement Reaction |
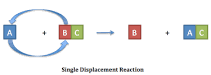
A single replacement reaction is also known as a substitution reaction, is a type of chemical reaction where one element replaces another element in a compound. |
|
|
Double-displacement Reaction |
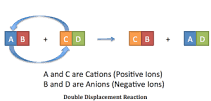
A double displacement reaction, also known as a double replacement reaction or metathesis, is a type of chemical reaction where two compounds react, and the positive ions and the negative ions of the two reactants switch places, forming two new compounds or products. |
|
|
Chemical Reaction |
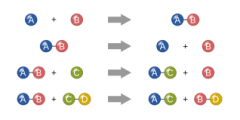
Chemical Reaction is a process that involves rearrangement of the molecular or ionic structure of a substance, as opposed to a change in physical form or a nuclear reaction. |
|
|
Chemical Equation |
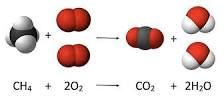
A chemical equation is the symbolic representation of a chemical reaction in the form of symbols and formulae, wherein the reactant entities are given on the left-hand side and the product entities on the right-hand side. |
|
|
Endothermic Reaction |
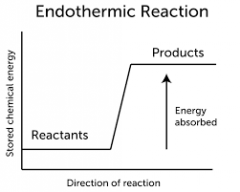
A endothermic process describes a process or reaction in which the system absorbs energy from its surroundings; usually, but not always, in the form of heat. ... The opposite of an endothermic process is an exothermic process, one that releases, "gives out" energy in the form of heat. |
|
|
Exothermic Reaction |
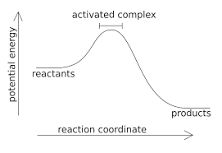
An exothermic reaction is a chemical reaction that releases energy by light or heat. It is the opposite of an endothermic reaction. Expressed in a chemical equation: reactants → products + energy. |

