![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
156 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
define epidemiology
|
the study of patterns of health and disease in populations and the factors that influence these patterns.
|
|
|
|
epidemiology is concerned with what?
|
who in the pop is getting the dz
who is not why are they getting the dz why are some sick and some not sick *concerned with the forest and the trees* |
|
|
|
epidemiology involves a logical approach to _______ regarding _____, ______, & ______
|
A logical approach to solving disease problems regarding cause, predisposing factors & intervention
|
|
|
|
epidemiology involves describing patterns of dz in relation to ______, _______, & _____.
|
individuals
place time |
|
|
|
uses of epidemiology (6)
|
Investigation and control of a disease of unknown etiology
Identification of the etiology & risk factors of a disease Determination of the origin of a disease of known etiology To study the natural history and prognosis of a disease Planning, monitoring & assessment of d’se control programs Assessment of the magnitude and impact of a disease |
|
|
|
epidemiology involves a systematic approach for ______
|
A systematic approach for the critical evaluation of scientific literature.
|
|
|
|
4 types of epidemiology
|
descriptive epidemiology
- Describes patterns & formulates hypotheses - Questions answered: What, Who, Where, When? analytical epidemiology - Involves analysis of data to test hypotheses - Questions answered: Why, How? experimental epidemiology - Involves pop. experiments to test hypotheses - Example: Vaccine and drug trials theoretical epidemology - Uses mathematical models to study disease |
|
|
|
7 epidemiological sub-disciplines
|
CLINICAL EPIDEMIOLOGY
- Uses epidemiological principles in the care of indivs - Is an important component of evidence-based medicine COMPUTATIONAL EPIDEMIOLOGY - Uses computer science in the study of epidemiology - Is used to formulate disease control strategies GENETIC EPIDEMIOLOGY - Is the study of epidemiology in related individuals FIELD (SHOE-LEATHER) EPIDEMIOLOGY - Used to investigate disease outbreaks PARTICIPATORY EPIDEMIOLOGY - Uses local knowledge for improving anl. health MOLECULAR EPIDEMIOLOGY - Uses molecular techniques in epi investigations SPATIAL (GEOGRAPHICAL) EPIDEMIOLOGY - Concerned with spatial patterns of disease & health |
|
|
|
what is the unit of concern in clinical medicine?
pathology? epidemiology? |
Clinical Medicine
- Sick Animal Pathology - Dead Animal Epidemiology - Healthy, sick and dead |
|
|
|
What is the diagnostic procedure for clinical medicine?
pathology? epidemiology? |
Clinical Medicine
- Identify d’se based on signs & symptoms Pathology: - Identify d’se based on host responses Epidemiology: - Determine patterns of d’se distribution |
|
|
|
What is the primary objective of clinical medicine?
pathology? epidemiology? |
Clin. Medicine
- Treatment of sick animals Pathology - Treatment of future animals Epidemiology - Control d’se & prevent future occurrence |
|
|
|
What questions are asked in clinical medicine?
pathology? epidemiology? |
Clinical Medicine:
- What is it? - How do I treat it? Pathology: - What is it? - What is its mechanism? - What caused it? Epidemiology: - What is it? - Which animals have it? - Where/ When is it occurring? - What caused it? - Why did it occur? |
|
|
|
What is a determinant of dz/ health?
|
any factor that when altered produces a change in the frequency or characteristics of disease
Physical factors Biological factors Behavioral factors |
|
|
|
primary vs secondary determinants
|
Primary determinants
- Have a major effect in inducing disease. - Example: rabies virus Secondary Determinants - Are predisposing or enabling factors to disease. - Example: poor hygiene |
|
|
|
intrinsic vs extrinsic determinants
|
Intrinsic (Endogenous) Determinants
- Are Internal to the host - Example: breed, species, sex, etc Extrinsic (Exogenous) Determinants - Are external to the host - Examples: poisons, trauma, radiation, etc |
|
|
|
epidemiological triad
|
agent
host environment |
|
|
|
epidemiological triad
|
agent
host environment |
|
|
|
The major component of epidemiology that differentiates it from other medical specialties is its focus on
a. Population b. Animals c. Diseased animals d. Healthy animals e. The environment |
a. Populations
|
|
|
|
The epidemiologic triad consists of
a. Individual, place, time b. Epidemiology, clinical medicine, pathology c. Agent, host, environment d. Intrinsic, extrinsic, & primary cause of disease e. None of the above |
c. Agent, host, environment
|
|
|
|
if we change a determinant, it will _______
|
change the nature or frequency of the dz
|
|
|
|
host range
|
the spectrum of hosts the agent can survive in
The broader the host range, the better the survival The narrower the host range, the easier to control |
|
|
|
Agent factors (determinants)
|
host range
infectivity infectious dose viabililty pathogenicity virulence immunogenicity antigenic stability |
|
|
|
infectivity
|
Ability to enter, multiply & produce change (may or may not be disease) in a host
Infectivity is variable from agent to agent Infectivity may vary within one spp. of agent. |
|
|
|
infection vs contamination vs pollution
|
Infection (not always disease)
- The entry and multiplication of an infectious agent in the body - Infection may not necessarily lead to d’se (inapparent infections) Contamination - Infectious agents on exterior surface of the body/object - Can be internal (e.g. Pasteurella multocida – in air sacs) Pollution - Presence of offensive matter in the environment - The offensive matter does not have to be infectious |
|
|
|
infectious dose
|
Quantity of agent necessary for transmission & infection.
Varies by agent |
|
|
|
viability
|
Ability of an agent to withstand environmental stress.
Anthrax spores can be viable in the soil for over 20 yrs |
|
|
|
viability
|
Ability of an agent to withstand environmental stress.
Anthrax spores can be viable in the soil for over 20 yrs |
|
|
|
pathogenicity
|
Is the power of an agent to produce clinical disease.
Also used for toxic chemicals, etc. Mathematically, it is the ratio of number animals developing d’se to the number exposed to infection |
|
|
|
what is the difference b/w virulence and pathogenicity?
|
Virulence is the degree of pathogenicity (the severity of disease)
Pathogenicity is the ability to produce clinical disease (no association with severity of disease) Both are dependent on host and agent factors |
|
|
|
does virulence depend on host or agent factors
|
both
|
|
|
|
does pathogenicity depend on host or agent factors
|
both
|
|
|
|
virulence
|
Is the degree of pathogenicity of an agent
Indicated by the severity of disease Mathematically, it is the ratio of the number of clinical cases to the number of infected animals. E.g. CFR |
|
|
|
which animals are the most dangerous epidemiologically speaking
a. those with subclinical infections b. those with mild disease c. those with moderate disease d. those with severe disease e. susceptible individuals |
a. subclinical dz
|
|
|
|
immunogenicity
|
Ability of an agent to stimulate an immune response.
The likelihood of repeated infections is reduced if the agent is highly immunogenic. Canine distemper– highly immunogenic Herpes virus– poorly immunogenic |
|
|
|
antigenic stability
|
The probability that the genome governing antigenic structure of an agent will undergo antigenic change.
Canine distemper virus is stable; influenza virus is not. |
|
|
|
host factors (determinants)
(9) |
genotype
age sex species & breed immunological status occupation size & conformation social & ethological coat color |
|
|
|
genotype
|
Genotype is the genetic makeup of an organism.
Genetic d’ses are inherited e.g. hemophilia A&B in dogs Genetic diseases belong to one of the three categories: Chromosomal disorders Mendelian (inherited) disorders Multifactorial disorders – variable genetic component - Interplay b/w environment and genetic factor (need both) |
|
|
|
how does age affect disease
|
Most diseases have an association with age.
Lower immunity in the young & very old Salmonellosis is deadly in chicks but mild in adult chickens Knowledge is useful in disease control Brucellosis vaccine - given to calves |
|
|
|
how does sex affect dz (4)
|
Hormonal:
- Males don’t have the stress of pregnancy, parturition & lactation - Sex hormones may increase risk of diabetes mellitus in bitches Occupational: - Heartworm infection risk is higher in male hunting dogs Behavioral: - Higher risk of bite wound abscesses in male than female cats Genetic differences in susceptibility between sexes - Sex-linked: Canine hemophilia A & B - more common in males - Sex-limited: Orchitis (inflammation of testicles) and metritis (inflammation of the uterus) - Sex-influenced: Lower d’se threshold in one sex than the other |
|
|
|
how do species & breed affect dz
|
Species
- Some d’ses affect almost all spp. e.g. rabies, tetanus - Other d’ses are host-specific e.g. canine distemper - Vultures & Jackals in Africa are resistant to anthrax (spread?) Breed Rottweillers and Dobermann Pinschers - more susceptible to canine parvovirus infection than other breeds. N’dama cattle: - Very susceptible to rinderpest. - Resistant to trypanosomiasis Zebu cattle - more resistant to tick-borne d’ses than European breeds. |
|
|
|
differentiate b/w immunological status and immunogenicity
|
Immunological status is the status of the host (is it resistant, immunocompromised)
Immunogenicity is the ability of the agent to produce clinical disease |
|
|
|
how does immunological status affect dz
|
Immunological status impacts d’se patterns
Immunity could result from: - Dam to offspring - Vaccination - Natural infection |
|
|
|
how does occupation affect dz?
|
Race horses - higher risk of leg injuries
Veterinarians - higher risk of zoonotic infections |
|
|
|
how does size and conformation affect dz?
|
Size: Is a disease determinant (regardless of breed)
- Large dog breeds have higher risk of hip dysplasia Conformation: is also a determinant of disease - Cows with a small pelvic outlet relative to their size - (e.g. Belgian Blue) are predisposed to dystocia |
|
|
|
host factors: social & ethological
|
Tail biting in pigs
Gastric torsion in horses – rolling behavior Lung cancer – higher risk in smokers Lantana camara poisoning – some breeds learn to avoid the plant |
|
|
|
how does coat color affect dz
|
Cutaneous squamous cell carcinoma in white cats
- Due to lack of pigmentation which protects from uv radiation Photosensitization seen in animals with: - Lack of pigmentation in parts exposed to direct uv radiation - Liver problems |
|
|
|
environmental factors (determinants)
(3) |
location (geography)
climate (macro & microclimate) husbandry (housing/ nutrition/ mgmt) |
|
|
|
how does location affect dz
|
Geological formations, vegetation and climate
Non-specific chronic canine pulmonary d’se in old dogs - Associated with urban residence. Why? Noise is associated with: - Decreased milk production in dairy cows - Increased incidence of abortion in rats. Temporal disease patterns affected by geography - Temperate regions – seasonal variations in disease prevalence - Tropics – variations during dry & wet months of the year |
|
|
|
how does climate affect dz
|
Macroclimate: includes the weather conditions, etc
- Hurricanes - Cold stress predisposes animals to disease - Wind may carry infectious agents over long distances(FMD virus/ Arthropod vectors) - Risk of respiratory diseases increases in cool damp weather - Drought may lead to decreased milk yield and weight gain Microclimate: Is climate in a small defined space. Terrestrial microclimate – over the surface of leaves affect the development of arthropods and helminths - Right humidity and temp will help arthropods thrive Biological microclimate – over surface of a host’s body change during the course of a disease & enhance d’se spread - Malaria during certain times (pathogenic state) there is a fever which leads to sweating which attracts more mosquitos to bite and spread disease Larger microclimates – Examples: calf house, a pen, etc - Poor ventilation increases risk of chronic equine resp. d’se |
|
|
|
how does husbandry affect dz
|
Housing
- Ventilation, bedding, and floor surfaces - Housing protects from a harsh macroclimate. Nutrition - Well-nourished animals may show prolonged survival - Feeding regimes are associated with gastric torsion Management: - Stocking densities - Replacement policy - Occupation - ‘Hump-sore’ (‘yoke gall’) |
|
|
|
interaction & association of factors (determinants)
|
Association of Factors
- Association of a factor & d’se does NOT necessarily imply cause-effect relationship. - Carrying matches is associated with lung cancer but doesn’t cause the disease. Interaction of Factors - Determinants do not act in isolation – they interact - Porcine stress syndrome -Interaction between the host (gene) & Environment (stressors). |
|
|
|
What will happen with a not very pathogenic agent in a non-immune population
|
Many will contract the agent and develop immunity w/o causing clinical disease????
|
|
|
|
What will happen with a very pathogenic agent in an immune population
|
It won’t get passed b/c the pop is immune
|
|
|
|
causation web
|
Is a method of conceptualizing how multiple factors combine to cause disease.
The causal web consists of direct and indirect causes of disease These causes are interconnected through a series of interconnected causal chains or web structures |
|
|
|
survival of an infectious agent depends on
|
its successful transmission to a susceptible host
its multiplication to maintain the life-cycle (aka life history) |
|
|
|
1. _______ is the portal of exit
2. ______ is the portal of entry |
1. reservoir
2. new host |
|
|
|
define a reservoir
|
Living or non-living matter
Where infectious agent normally lives and multiplies Maintains & perpetuates itself From which it can be transmitted |
|
|
|
differentiate reservoir from source of infection
|
Source of infection is where agent jumps from where it is to a susceptible host
- This is where the agent leaves - Can be a subset of reservoir Reservoir requires that the agent can survive there, multiply there, leave there. |
|
|
|
nidus
|
is center where infection settles & from where it spreads
Example: abandoned chicken coops may be a nidus for Histoplasma capsulatum |
|
|
|
source of infection
|
is an animal or substance from which an infectious agent passes directly to a susceptible host
SOI may or may not be a reservoir - Reservoir and SOI are the same for most viruses. Examples: SOI = Reservoir: Canine distemper SOI ≠ Reservoir: Equine infectious anemia (horse = reservoir; biting fly = SOI) |
|
|
|
what is a carrier
|
an infected person or animal
that sheds pathogenic/ potentially pathogenic organisms in the absence of discernible clinical disease & serves as a potential SOI Carriers are very dangerous epidemiologically b/c they have the disease & spread it but you don’t know ex: typhoid mary |
|
|
|
types of carriers
|
HEALTHY (ASYMPTOMATIC)
- Shed the agent without developing clinical disease - This state is called inapparent infection - Is a very important type of carrier for disease control - Example: Salmonella interrogans serotype typhimurium in swine. INCUBATORY - Shed agent prior to appearance of clinical signs - Example: Canine distemper and foot-and-mouth disease CONVALESCENT - Shed agent for short periods after signs have abated - Examples: - Salmonellosis in cattle / Feline panleukopenia INTERMITTANT SHEDDERS - Intermittently shed agent for a moderate periods of time after recovery from disease - Example: Toxoplasmosis in cats CHRONIC - Shed agent for extended periods of time after recovery - Example: Tuberculosis |
|
|
|
healthy (asymptomatic) carriers vs. incubatory carriers
|
Healthy (asymptomatic) carriers
- Shed agent but never show clinical signs - Hemophilia carriers - Typhoid Mary - Salmonella in snakes Incubatory carriers - Shed prior to appearance of clinical signs - Mononucleosis - Canine distemper |
|
|
|
convalescent vs. intermittent vs. chronic carriers
|
Convalescent carriers
- Shed after clinical signs have abated - Salmonellosis in cattle Intermittent shedders - Intermittenetly shed agent for moderate periods of time after recovery from disease - Toxoplasmosis in cats Chronic carriers - Able to shed for long periods of time after recovery - TB |
|
|
|
portal of exit
|
Is a place of exit of agents from the reservoir
It includes: - Respiratory tract: Nose (secretions) - Alimentary tract: Mouth (saliva, sputum); anus (feces) - Urinogenital tract: Urine, semen, eggs - Eyes: Tears (Pink eye – Haemophilus influenzae) - Body surface: Skin & Hair/ lesions & crusts/ exudates - Multiple exits: - Complicate control procedures(Q-fever & streptococci) |
|
|
|
modes of transmission
|
horizontal
- direct - indirect vertical - Hereditary - In Utero - During birth - Colostrum - Suckled milk |
|
|
|
how does direct transmission differ from indirect transmission
what is a primary vehicle |
Direct
- The agent responsible for the disease - Need HIV virus to get disease Indirect - Risky behavior/ unprotected sex indirectly lead to disease Primary vehicle - Saliva - Secretions - Etc. |
|
|
|
primary vehicle vs. secondary vehicle vs. vector
|
Primary vehicle:
- Secretions, excretions or other body fluids or tissues - Examples: saliva, feces, blood, urine Secondary vehicle - Inanimate object contaminated with the primary vehicle - Examples: water, food, grass, etc. Vectors - Are invertebrates that transmit infectious agents - Examples: fleas, ticks, flies, mosquitoes, snails |
|
|
|
define direct transmission
what 2 ways may it occur through |
Susceptible host gets infected either by contact with an infected host or host’s infected discharges
Direct transmission may occur through: Contact (contagious diseases) - ‘Fragile’ agents are generally transmitted directly Coitus (venereal diseases) - Occurs in both vertebrates and invertebrates |
|
|
|
Indirect transmission may occur in the following ways:
(4) |
Vehicle and fomites
Airborne Ingestion – food or water Vectors |
|
|
|
airborne transmission can occur by
|
Dust, expiratory droplets, droplet nuclei, vapors & gases
|
|
|
|
define a vector
what are the 2 types |
are living invertebrates responsible for the transmission of infectious agents
Mechanical - The agent doesn’t change while in the vector - No time lapse Biological vectors - There is some change in the agent (multiplication or maturation or reproduction) - Time lapse |
|
|
|
extrinsic differentiation period
|
Time from when vector picks up the agent to the time when it can infect another animal
|
|
|
|
incubation period vs. induction period
|
Incubation period
- Time between exposure and development of clinical signs Induction period - Time between exposure to chemical and development of signs of exposure |
|
|
|
________ requires an extrinsic incubation period before transmission can occur.
|
biological vector
|
|
|
|
types of biological transmission
|
Developmental transmission
- An essential devpt phase of agent occurs in vector - Example: Dirofilaria immitis in mosquitoes Propagative transmission - Multiplication of the agent occurs in the vector - Example: arboviruses in mosquitoes Cyclopropagative transmission - Both development and multiplication occur in the vector - Example: Babesia in ticks |
|
|
|
True or False: In general it is easier to control if the host range is narrow
|
True
|
|
|
|
The severity of disease caused by an agent in a susceptible host is a measure of
a. Contagiousness b. Infectivity c. Virulence d. pathogenicity |
c. virulence
|
|
|
|
One of the following does not belong to the group. Which is it
a. Immunogenicity b. Pathogenicity c. Immunological status d. Virulence e. infectivity |
c. immunological status
|
|
|
|
The extrinsic incubation period is the term used to refer to
a. Estimating the incubation period in a propogated curve b. The time between infection and the 1st clinical signs (incubation period) c. The time b/w exposure to a toxin and 1st clinical signs (induction period) d. The time before an agent can be transmitted in biological vectors |
d. The time before an agent can be transmitted in biological vectors
|
|
|
|
A localized reservoir is called a
a. Chronic carrier b. Source of infection c. Nidus d. Convalescent carrier |
c. nidus
|
|
|
|
True or False: Mechanical vectors cannot be reservoirs
|
True
The mechanical vector just transmits the agent but no change occurs (no propagation) |
|
|
|
Reasons for special review:
|
Advancement or change in rating to pay grade E-7
Detachment for PCS Detachment for intra-command reassignments if AO changes Detachment of AO who directly supervises employee Evaluee completes TAD, ADSW-RC, ADSW- AC for any length of time Also for: NJP Convicted by Court Martial |
1
|
|
|
with ________ infection can occur remotely from the vector
|
water inhabiting vectors
ex: Schistosomiasis |
|
|
|
hereditary vs. congenital
|
Hereditary
- Carried in the genome of the parent e.g. retroviruses - DNA copies of the virus carried in the hosts genome Congenital - acquired in utero |
|
|
|
WHAT IS THIS RANK IN THE ARMY?
|
E7 / SERGEANT 1ST CLASS
|
1
|
|
|
contagiousness vs. communicability
|
Contagiousness refers to spread by direct contact b/w hosts
Communicability doesn’t differentiate how the agent is spread |
|
|
|
At shore installations how is the CG ensign flown?
|
Starboard yardarm of the flag mast
|
1
|
|
|
_______ is the condition under which infection will occur
|
effective contact
|
|
|
|
seasonal vector-borne infections have a ______ effective contact.
(short or long) |
short
|
|
|
|
effective contact depends on _______
|
Portal of exit
Portal of entry Stability of the organism |
|
|
|
If an agent that is lowly infective, highly pathogenic, and highly virulent was introduced into a totally susceptible population of animals, what would you expect to see clinically
a. A few severely affected animals b. Many mildly affected animals c. A few mildly affected animals d. Many severely affected animals |
a. A few severely affected animals
|
|
|
|
The extrinsic incubation period is the term used to refer to
a. Estimating the incubation period in a propagated curve b. The time b/w infection and 1st clinical signs c. The time b/w exposure to a toxin and the 1st clinical signs d. The time before an agent can be transmitted in biological vectors |
d. The time before an agent can be transmitted in biological vectors
|
|
|
|
True or False: Mechanical vectors can be reservoirs
|
False
|
|
|
|
For long-term survival, infectious agents require:
A: Hosts with low immunity B: Hosts with moderate immunity C: Hosts with high immunity |
B: Hosts with moderate immunity
|
|
|
|
host immunity depends on
|
Nature of the agent
Challenge dose Environmental factors |
|
|
|
herd immunity
|
Resistant of the pop to infection or disease if infected
|
|
|
|
True or False: host immunity is absolute
|
false
it is relative |
|
|
|
rate of infectious disease spread in a population depends on
|
Characteristics of the infectious agent
Host immunity of the animals in the pop Pop. structure (proportion of susceptibles, resistant, etc) Pop. dynamics (Movts., social interaction, behavior, etc) Contact rate |
|
|
|
herd immunity depends on
|
Population Structure
- proportion of immune & susceptible animals - presence of alternative hosts & vectors - More alternate hosts = more chance of infection Population Dynamics - Movements, social distance, behavior, etc. - Contact Rate |
|
|
|
herd immunity vs. host immunity
|
Herd immunity is affected by population structures and population dynamics whereas host immunity doesn’t get affected by those 2
|
|
|
|
what is contact rate
how does it affect herd immunity |
Is the rate at which susceptibles interact with infected anls.
This interaction affects the degree of herd immunity. - Reducing the prob. of contact (duration, interval, effectiveness) between infected and susceptibles may slow down d’se spread Possible to control disease by changing herd immunity by manipulating herd structure without inducing host immunity |
|
|
|
endemic occurrance
|
Is used to refer to:
- the usual frequency of occurrence of a disease in the population - the constant presence of the disease in the population Endemic occurrence can be used to describe both clinical and sub-clinical disease It is applied to both infectious and non-infectious diseases When describing endemic disease, the affected population and its location should be specified |
|
|
|
hyperendemic vs hypoendemic vs. holoendemic
|
Hyperendemic occurrence - Disease is continuously present at a high level, affecting all age-groups equally.
- Rabies in Canada Hypoendemic occurrence – Disease is continuously present at a low level, affecting all age groups - TB in developed countries Holoendemic occurrence – A high level of infection begins early & affects most of the young pop resulting in a state of equilibrium so that the adult pop. show evidence of disease much less commonly. - Malaria in areas that it occurs (very young are more likely to be infected while the old have much lower incidence due to immunity) |
|
|
|
define epidemic
when does it occur |
the occurrence of disease at a level in excess of the expected (i.e. endemic) level.
Epidemic occurs when the pop is exposed to one or more factors that were not present previously |
|
|
|
point source vs. propagating epidemic
|
Point source (common source epidemic)
- All individuals infected from same source at the same time - Food borne illness Propagating - 1 individual comes in and then infects others - b/c not all infected from same source |
|
|
|
_______ is when disease occurs irregularly and haphazardly.
|
sporatic occurrence
|
|
|
|
1. _________ phase is when # of cases is going up
2. __________ phase is when # of cases is going down 3. _________ affects the amplitude of an epidemic curve |
1. progression
2. regression 3. infectivity High infectivity = increased # of individuals infected (higher amplitude) |
|
|
|
_______ makes you reach culmination point quicker (affects slope of epidemic curve)
|
Shorter incubation period
|
|
|
|
__________ has to be known before an epidemic can be recognized
|
The endemic level of the disease in a population
|
|
|
|
__________ is required to allow contact-transmitted epidemic to commence
|
A minimum (or threshold) density of susceptible animals
|
|
|
|
Factors affecting the shape of an epidemic curve
|
Incubation period of the disease
- shorter = steeper slope Infectivity of the agent - higher = increased amplitude Proportion of susceptible hosts in the population - less = lower amplitude and slower to reach culmination point |
|
|
|
how do you construct an epidemic curve
|
Identify the date of onset
Set the time Interval - 1/3 or less of IP Create X-axis lead and end periods - Should be equal to 2 incubation periods (lead and end periods) Draw tick marks & label time intervals Assign area equal to one case Plot the cases on the graph Mark Critical events on graph & add labels |
|
|
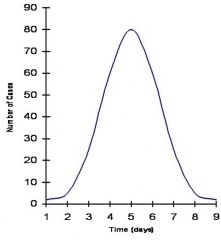
|
common source epidemic
Rapid Progression and Regression Compressed in time Approaches symmetry at average incubation period |
|
|
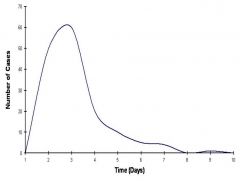
|
common source epidemic
Rapid Progression and Regression Compressed in time Approaches symmetry at average incubation period |
|
|
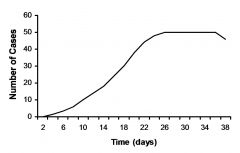
|
propagated epidemic
Initial (primary) cases infect susceptible individuals that become secondary cases Index case – Is the first identified primary case Gradual progression (ascending limb) Tendency to plateau A feature of vector & animal-to-animal spread |
|
|
|
why can you get a second peak with a propagating epidemic?
|
Introduction of susceptible animals into the area
Movt of infected animals from the epidemic area & subsequent contact with susceptible animals. |
|
|
|
temporal patterns
|
Short-term - Are exhibited in epidemics
Cyclical Trends - Result of periodic fluctuations in disease occurrence - Seasonal Trends. Are due to: - changes in host density - management practice or behavior of the host - survival of the infectious agent - vector dynamics Secular trends - are long term (several years) increases or decreases in disease rates |
|
|
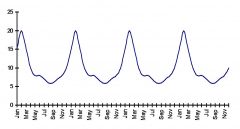
|
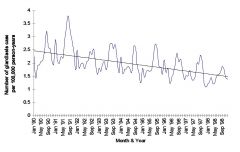
Cyclical Trends
- Result of periodic fluctuations in disease occurrence Seasonal Trends. Are due to: - changes in host density - management practice or behavior of the host - survival of the infectious agent - vector dynamics |
|
|
|
Secular trends: are long term changes where, in addition to short term ups and downs, the curve either climbs or declines more or less steadily over an extended period of time, usually years.
|
|
|
|
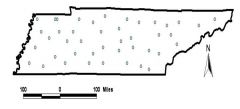
Random Spatial occurrence
are exhibited by sporadic outbreaks |
|
|
|
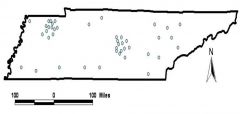
Clustering spatial pattern
Is the aggregation of disease or other events in amounts greater than would be expected by chance alone. |
|
|
|
space time clustering
|
Is an interaction between the place and time of occurrence of a disease.
An epidemic is clustering of disease over both time and space. |
|
|
|
sensitivity
|
Ability to detect disease
Se = (TP) / (TP +FN) |
|
|
|
clinical epidemiology
|
The application of the concepts of epidemiology and biostatistics to the practice of medicine on the individual patient (or herd/flock).
|
|
|
|
evidence based medicine
|
Process of systematically finding, appraising, and using research findings as the basis for clinical decisions.
|
|
|
|
what is outbreak investigation
|
a systematic procedure to identify causes and sources of epidemics
|
|
|
|
what is an outbreak
|
an epidemic that is limited to a localized increase in the incidence of a disease
|
|
|
|
sensitivity vs. specificity
|
Sensitivity: Ability of a test to accurately classify a group of patients known to have a disease
Specificity: Ability of a test to accurately classify a group of patients known to be free a disease |
|
|
|
10 steps of an outbreak investigation
|
Determine the existence of an outbreak
Confirm the diagnosis Define a “case” and count cases Describe the patterns & determine who’s at risk Determine the magnitude of the problem Develop & test hypothesis Compare the hypothesis with established facts Plan a more detailed & systematic study Prepare a written report Execute Control & prevention measures |
|
|
|
attack rate
attack rate ratio |
# sick/ total population
attack rate in exposed/ attack rate in unexposed |
|
|
|
accuracy refers to
|
What proportion of all tests have given positive results?
|
|
|
|
forage
|
search widely for food or provisions
|
|
|
|
positive vs. negative predictive value
|
Positive predictive value- the probability that a patient whose test result is positive has the disease.
Negative predictive value- the probability that a patient whose test result is negative does not have the disease |
|
|
|
1. ________ & _________ Measure the accuracy of a diagnostic test when applied to a group of patients whose disease status is known. What does it depend on?
2. _______ & _______ Measures the ability of a diagnostic test to accurately classify a patient whose disease status is unknown. What does it depend on? |
1. sensitivity & specificity
test properties 2. predictive value Depends on the sensitivity, specificity and pre-test probability (prevalence) of disease in the population from which the patient was selected. |
|
|
|
prevalance vs. point prevalance
|
Prevalence: fraction or proportion of a group that possess a clinical outcome/disease during a given period (point) in time.
Point prevalence is measured by a single survey of the group. |
|
|
|
what effect does serial testing have on sensitivity & specificity?
|
Series
- Improve specificity - Decrease sensitivity Parallel - Improve sensitivity - Decrease specificity |
|
|
|
What effect does serial testing have on sensitivity & specificity?
parallel? |
Series
- Improve specificity - Decrease sensitivity Parallel - Improve sensitivity - Decrease specificity |
|
|
|
when should you use a sensitive test?
|
When there is a penalty for missing a disease (heartworm, brucellosis in cattle, equine infectious anemia [EIA])
Early in diagnostic process to “rule out” disease Few false negative results (high NPV) Sensitive test most helpful when result is negative Useful to screen patients and eliminate those subjected to a more expensive and time consuming confirmatory test |
|
|
|
when should you use a test with high specificity?
|
When there is a penalty for false positive result (Feline infectious peritonitis, Johne’s disease)
Useful to “rule in” disease (few false positives, high PPV) Specific test most helpful when result is positive Used as follow-up test to confirm disease status on subset of patients positive on screening test. |
|
|

|
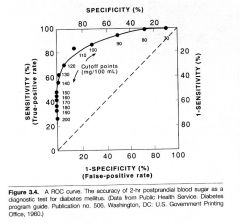
A. 100% sensitive
B. Optimal combination of sensitivity and specificity C. 100% specific |
|
|
|
the best spot is one near the top left corner
|
|
|
|
A serosurvey was conducted on a sample of dogs to determine the prevalence heartworm disease in Knox County, TN. Results 50 out of 500 dogs (10%) tested positive. This estimate is the (more than one answer may be correct):
a. Rate of heartworm infection among dogs b. Ratio of heartworm infection among dogs c. Proportion of heartworm infection among dogs d. Prevalence of heartworm infection among dogs |
c. Proportion of heartworm infection among dogs
d. Prevalence of heartworm infection among dogs |
|
|
|
The mean white blood cell count in a sample of dogs with parvovirus is 3,000/mm3 with a standard deviation of 750. Which of the following ranges would include approximately 95% of the dogs in this sample?
a. 2,250 to 3,750 b. 750 to 5,250 c. 2,500 to 3,500 d. 1,500 to 4,500 e. 2,000 to 4,000 |
d. 1,500 to 4,500
|
|
|
|
In a study to determine the effect of an new experimental treatment (antibiotic) on reproductive performance among broodmares with uterine infections, the attending veterinarian gives the new “experimental” treatment to mares she thinks are more severely affected. This action would affect the:
a. Internal validity b. External validity |
a. Internal validity
|
|
|
|
internal vs external validity
|
Internal Validity: The extent to which the design and conduct of the trial eliminate the possibility of bias.
- Are the results valid for the patients studied? - Could the results be due to bias or chance? External Validity: The extent to which the results of a trial provide a correct basis for generalization to other circumstances (= generalizability or applicability) - Are the patients in the study similar to the ones in my practice? - Are they so different that the results do not apply? - To which group of patients can I apply the results? |
|
|
|
liklihood ratio pros and cons
|
Advantages
- Probability of disease given a test result that falls within a limited range of values - Degree of measurement abnormality considered Disadvantages - Use of odds - May calculate or use nomogram to convert to probabilities - LR differs from sensitivity and specificity in that result of test is probability of disease. |
|
|
|
evidence based medicine steps
|
Formulate the problem
Search for the best evidence available Critical appraisal of the evidence Implement results in your clinical practice Evaluate our performance |
|
|
|
types of bias
|
Selection bias‑ patients in one of the treatment groups differ from those in the comparison group with regard to one or more factors that can influence the outcome being measured.
Observation bias‑ measurement of the outcome is systematically different between treatment groups. Confounding bias‑ An unknown factor (X) is associated with the suspected risk factor and the outcome (disease). |
|
|
|
measurement of characteristics
|

|
|
|
|
objective vs. subjective data
|
Objective (hard data)
Laboratory, demographic e.g., age, sex, breed Subjective (soft data) Quality of life; improvement of clinical signs, severity of lameness, assessment of pain, extent of nursing care Increasing quality of subjective (soft) data Lameness Appetite |
|
|
|
accuracy vs. precision
|
Accuracy (validity): The extent to which observations represent the truth
Precision (Reliability- repeatability): The extent to which repeated measures of the same factor are close together |
|
|
|
rate vs ratio vs proportion
|
rate:
affected/total (in a given time period) ratio: affected/unaffected proportion: affected/total (no time period) |
|
|
|
how do we define abnormality
|
Abnormal as unusual (infrequent)
- Mean + 2 SD - Extreme 2.5% at each end of distribution - BUN, WBC Abnormal as associated with disease - Cut point to determine successful passive transfer in calves. Abnormal as associated with response to treatment - Heart murmur w/o signs of congestive heart failure (CHF) dog < 30 lbs. - considered “normal” (80% of old dogs) - Older dog > 30 lbs. with a heart murmur and signs of CHF is treated |
|
|
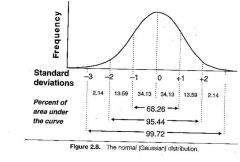
|
If this were the distribution of white blood cells in a group of dogs with parvovirus, and the mean was 3,000 with a standard deviation of 500 then 68% of dogs would have values between 2,500 and 3,500 and 95% would have values between 2,000 and 4,000.
|
|
|

|
If this were the distribution of white blood cells in a group of dogs with parvovirus, and the mean was 3,000 with a standard deviation of 500 then 68% of dogs would have values between 2,500 and 3,500 and 95% would have values between 2,000 and 4,000.
|
|

