![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
26 Cards in this Set
- Front
- Back
|
Dominant minerals
|
1. Cations + (CO3)2-
2. Most common cations - Larger Ca2+ - Smaller Mg2+ |
|
|
Dominant minerals: Aragonite
|

1. Open orthorhombic structure
2. Can accomodate cation substitution 3. Unstable 4. CaCO3 |
|
|
Dominant minerals: Calcite
|

1. More closed rhombohedral structure
2. High Mg calcite (>5% Mg) 3. Low Mg calcite 4. CaCO3 |
|
|
Dominant minerals: Dolomite
|

CaMg(CO3)2
|
|
|
Carbonate deposition controls
|
1. Temperature
2. Pressure 3. Agitation 4. Organic activity 5. Minor siliclastics 6. Light |
|
|
Temperature
|
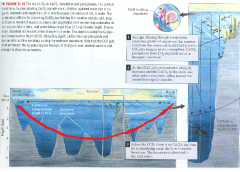
1. Precipitates in warm water
2. Dissolves in cold water 3. CCO diagram (carbonate concentration depth) |
|
|
Agitation
|
1. Currents and waves allow CO2 to escape
2. Promotes carbonate precipitation |
|
|
Organic activity
|
1. Many organisms precipitate CaCO3 from seawater
2. Many organisms remove CO2 from water - Promotes precipitation of CaCO3 |
|
|
Absent/limited presence of Siliclastics
|
Abundant silicalastics will overwhelm carbonate production
|
|
|
Light
|
1. Photosynthesis requires light
2. Nearly all carbonate production occurs at depths where light can penetrate (<20 m) 3. Carbonate not produced inmuddy water |
|
|
Distribution though time: Precipitation of Modern Cabonates
|
1. Inorganic
- Caves (travertine) - Springs (tufa) 2. Organic - Reefs * Shallow marine * Low silicates * Warm water 40 degrees North or South of Equator |
|
|
Distribution thought time: Precipitation of Ancient Carbonates
|
Abundant Proterozoic-Phanerozoic
- Controlled by: * Tectonics * Orogenic evolution |
|
|
Modern Carbonates: Lithification of Carbonates
|
1. Non-marine
- Shallow carbonate lacustrine mud 2. Shallow marine - Continental shelves where siliclastics are low 3. Deep marine - Abyss above the carbonate compensation 4. Others - Limestone rubble - K-horizon in soil (caliche) |
|
|
Ancient Carbonates: Lithification of Carbonates
|
1. Non-marine
- Rare (preservation?) 2. Shallow marine - Abundant Proterozoic-Mesozoic - Broad shallow seas that covered continents 3. Rare - Not preserved - Not abundant carbonate plankton 4. Others - Rare (preservation?) |
|
|
Limestone components
|
1. Allochems
- Skeletal - Non-skeletal 2. Orthochems |
|
|
Allochems: Skeletal clasts
|

Fossil fragments (biocasts)
|
|
|
Allochems: Non-skeletal clasts
|
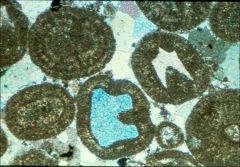
1. Coated grains (Ooids/ooliths)
2. Chemical or biochemical 3. Intrabasinal |
|
|
Allochems: Non-skeletal clasts
|

1. Peloids
- Sand-sized clasts of micro-crystalline carbonate - Intra-basinal origin * Fecal pellets * Recrystallization as allochem |
|
|
Allochems: Non-skeletal clasts
|
Grain aggregates
- Weakly held together by microbial mats - Local intrabasinal |
|
|
Allochems: Non-skeletal clasts
|
Limestone clasts
- Ripped up and transported * Intrabasinal * Estrabasinal - Equal siliciclastic rock fragments |
|
|
Orthochems
|

1. Micrite (carbonate mud)
- equals silicicclastic matrix 2. Spar (carbonate cement) |
|
|
Dunham Classification
|
1. Empasizes texture
- Allochems or grains - Mud of any compostion - Primary spar cement * Grain supported - Secondary spar * Recrystallization of micrite * Not grain supported |
|
|
Dunham Classification: Limestones
|
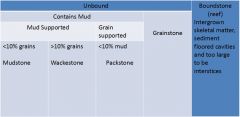
1. Unbound
A. Contains mud - Mud supported * Mudstone (<10% grains) * Wackestone (>10% grains) - Grain supported * Packstone (<10% mud) B. Grainstone 2. Boundstone (reef) |
|
|
Depositional texture not recognizable
|
1. Crystalline carbonate
2. Allochems NOT grain supported floating in spar |
|
|
Dolomite
|
1. CaMg(Co3)2
2. Forms naturally in unrealistic conditions 3. Penecomtemporanous replacement of Mg for Ca * High ph environments * Precipitation of gypsum, anhydrite, to use Ca cation * Mg becomes abundant in brine and replaces Ca 4. Secondary replacement of Ca by Mg from high Mg fluid |
|
|
Dolomite
|
1. Ancient dolomite widespread, modern dolomite limited
- Past environments were different than today - Most dolomite is by recrystallization over long time spans |

