![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
29 Cards in this Set
- Front
- Back
|
Particle Concept |
Short EM waves such as X-rays predominantly react with matter as if they were particles rather than waves At high frequencies electrons interact with EM radiation as if the EM radiation were an energy bundle. These particles are actually discrete bundles of energy and each of these bundles is called a quantum or photon |
|
|
Atomic Structure |
X-ray photons may interact either with orbital electrons or with the nucleus. In the diagnostic energy range, the interactions are always with orbital electrons The most important factor is the atomic make up of a tissue and not its molecular structure |
|
|
Atomic Structure |
Electrons – negatively charges Protons – positively charged Neutrons - Neutral K shell : 2 electrons L shell : 8 electrons Each shell has a specific binding energy & closer the shell is to the nucleus, the tighter it is bound to the nucleus. The electrons in the outermost shell are loosely bound to the nucleus & are hence called “free electrons”. |
|
|
Interaction of x-ray photons with matter |
When a beam of x-ray photons passes through matter, its intensity (energy – or number of x-ray photons - flowing per second) is reduced the beam has been attenuated, photon energy has been removed from the beam |
|
|
Interaction of x-ray photons with matter Image |
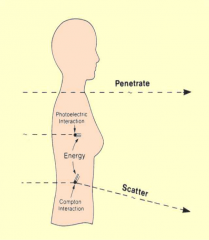
|
|
|
Absorption |
Some or all of the x-ray photon energy may be absorbed when energy is transferred to matter The energy deposited per unit mass of matter is called absorbed dose Units: 1 joule per kg = 1 gray (Gy) The energy deposited as absorbed dose causes ionisation and subsequent chemical changes that may result in biological effects |
|
|
Scatter |
Some x-ray photons are partially absorbed and their remaining energy scattered: deflected from their original path At low photon energies in diagnostic imaging, scatter occurs in all directions and may contribute to loss of image quality by increasing the overall density of the image receptor Scattered x-ray photons contribute to the patient’s dose (by internal scatter) and exposure of staff |
|
|
Scatter Image |
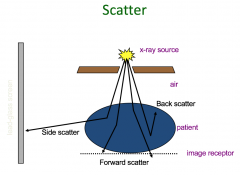
|
|
|
Scatter Explained |
Direction of scatter is forward when hits body it may scatter Low energy = the more scatter and can bounce straight back 180 degrees The higher energy photon the more likely it will still travel forward but scatter and deviate slightly |
|
|
Attenuation is exponential… |
Attenuation increases with: - Physical density - Atomic number - Electron density Decreases with: - Beam energy more attenuation with the larger mass of patient less attenuation with a small mass lead attenuates as it has a high atomic number |
|
|
Exponential Attenuation |
The linear attenuation coefficient (μ) gives the fractional reduction in intensity of a homogenous beam of x-ray photons per cm thickness of matter For example, the linear attenuation coefficient of soft tissue for 100 keV x-ray photons is approximately 0.2 (20%) (Bushong: table 29.1 in 6th ed)20% of photon energy is attenuated in each cm thickness of soft tissue; 80% is transmitted |
|
|
Exponential Attenuation Image |
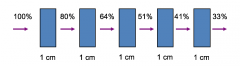
|
|
|
Interaction Processes |
There are various interaction processes of x-ray (and gamma ray) photons, that may occur alongside each other: -Coherent (elastic) scatter (negligible) -Compton (inelastic) scatter -Photoelectric absorption -Pair production -Photodisintegration |
|
|
Coherent Scattering |
Radiation only undergoes change in direction. No change in wavelengththat's why sometime called “ unmodified scattering Coherent scattering of X-rays is an interaction of the wavetype in which the X-ray is deflected. Coherent Scattering occurs mainly at low energies. |
|
|
Coherent Scattering Image |
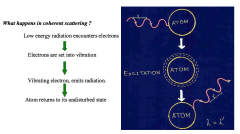
happens at such low energy levels that not relevant Where x-ray is scattered and photon does not lose any energy just put off tract |
|
|
Relative importance of each interaction process in water |
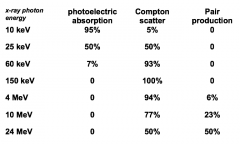
|
|
|
Photoelectric absorption |
Occurs when an x-ray photon interacts with a bound electron, in the inner shells of an atomOnly occurs if the energy of the x-ray photon exceeds the binding energy of the shell The x-ray photon disappears by transferring all its energy to the bound electron This energy is used to overcome the binding energy of the electron, which then escapes the atom as a photoelectron carrying kinetic energyThe photoelectron loses its KE via ionisation of surrounding atoms |
|
|
Photoelectric absorption Image |
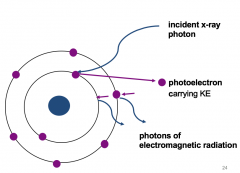
|
|
|
Photoelectric absorption Explained |
Photoelectric absorption happens at inner shell Causes the photon to completely disappear and gives it energy to the electron giving it more kinetic energy which causes it to move very fast. Photo electron is removed from the shellPhoton wave (X-ray) hits the electron transferring all pf its energy and photon ceases to exist. This forms a photon electron with enough kinetic energy will fly off leaving a gap. |
|
|
Photoelectric absorption - The gap in shells? |
the gap in the second shell caused by the removal of the photon electron will cause an electron from the outer shell to fall into the gap as the atom likes to remain stable Cascade of electrons as more electrons drop down to fill gaps until it stops at the outer most shell where no more can drop down and creates an ion and is now positively charged. As the electron drops down it loses energy and there is a known energy gap between the cells this creates photons of electromagnetic radiation containing less energy. Photon considered as a wave. |
|
|
When will Photoelectric absorption occur? |
is more likely to occur: in beams of low energy when the average x-ray photon energy is < 25 keV n dense matter with atoms of higher proton number eg bone, metal (shielding, filters), positive contrast media (barium sulphate, iodine) With a bound electron in an atom where the x-ray photon energy is just above the binding energy |
|
|
Compton Scatter (Occurs at outer shell) |
Occurs alongside photoelectric absorption An x-ray photon interacts with a bound electron, whose binding energy is negligible in comparison with the x-ray photon energy Some of the x-ray photon energy is transferred to the electron, to overcome its binding energy. It escapes the atom as a Compton electron carrying kinetic energy The Compton electron loses its KE via ionisation of surrounding atoms The x-ray photon with reduced energy is deviated, or scattered, from its original path and will interact again until all its energy is lost and it disappears. |
|
|
Compton scatter Image |
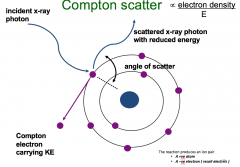
|
|
|
Compton scatter Explained |
happens alongside other processes photon interacts with an outer shell electron The electrons in the outer shell is very weak and easily removed e.g. Compton scatterThe photon does not disappear when it interacts with an electron in an atom. Is not removed just loses some of its energy and deviates on its path hence it’s called scatter. Photon bounces off with reduced energy The electron will bounce off as a Compton electron carrying KEProportion to electron density Atom has lost electron in the outer shell and becomes a positively charged ionThere is also a negatively charged electron flying around the body causing damage. |
|
|
When will Compton Scatter occur? |
In higher energy x-ray beams (average energy >25 keV) when electron binding energies in the attenuating material are negligible in comparison As photon energy increases, the proportion of forward scatter increases. In materials containing atoms with a high electron density eg hydrogen. |
|
|
Pair production |
high energy x-ray photon interacts with a nucleusThe energy is converted into an electron and a positron (anti-matter)The positron annihilates with an electron; the two particles are converted back into energy – two gamma ray photons of 511 KeVPair production only becomes important in very high energy beams of photons above 10 MeV used in PET (positron emition tomography) scanning |
|
|
Pair production Image |
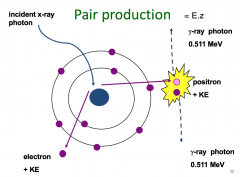
|
|
|
Photodisintegration |
A photon with extremely high energy ( 7-15 MeV), interacts directly with the nucleus of an atom. May eject a neutron, proton or on rare occasions even an alpha particle. No diagnostic importance ( We rarely use radiation >150 KeV in diagnostic radiology. |
|
|
Video |
https://www.youtube.com/watch?v=4p47RBPiOCo |

