![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
7 Cards in this Set
- Front
- Back
|
test 1 |
blue precipitate green ppt and goes brown at surface |
|
|
test 2 |
brown ppt white ppt blue ppt |
|
|
a) reaction |
cu2+ (AQ) + 2OH-(AQ) --> Cu(OH)2(S) precipitation |
|
|
b) ion diagram |
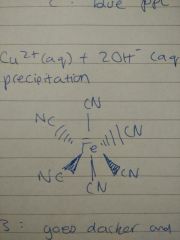
|
|
|
part 3 equilibrium |
goes darker and Eq pos goes right |
|
|
4ai) EDTA |
colourless solution forms : the EDTA complex is more stable |
|
|
4aii) equtation |

|

