![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
96 Cards in this Set
- Front
- Back
|
Hemostasis |
Process by which bleeding is stopped |
|
|
3 Stages of Hemostasis |
1) Vascular injury 2) Platelet plug formation 3) Coagulation |
|
|
Vascular Injury |
*Damage to blood vessels
*Secretion and synthesis of vasoconstrictors and platelet recruiting/activating molecules (thromboxane, ADP, thrombin) |
|
|
Platelet Plug Formation |
*Initiated when platelets exposed to collagen on damaged blood vessels-->platelets adhere-->platelet activation-->platelets aggregate to form plug |
|
|
Glycoprotein IIa/IIIb Receptor |
Forms a bridge with fibrinogen to cause platelet aggregation |
|
|
Coagulation |
*Process that leads to production of fibrin |
|
|
Fibrin |
*Protein that stabilizes platelet plug *Temporarily stops bleeding
|
|
|
2 Coagulation Pathways |
1) Intrinsic (contact activation) 2) Extrinsic (tissue factor) |
|
|
Common Pathway |
Intrinsic and Extrinsic pathways converge at Xa which is the final common pathway leading to fibrin formation |
|
|
Blood Clot |
Can form in blood vessels or the heart |
|
|
2 Types of Blood Clots |
1) Arterial thrombosis 2) Venous thrombosis |
|
|
Arterial Thrombosis |
*Clot that forms in high blood flow environment of the arteries *Called "white clot" *May lead to MI *Localized injury |
|
|
Venous Thrombosis |
*Clot that forms in slow blood flow environment of the veins *Called "red clod" *May lead to DVT or PE *Localized and secondary injury |
|
|
3 Risk Factors for Thrombosis (Virchow's Triad) |
1) Circulatory Stasis 2) Vessel wall injury 3) Hypercoagulability |
|
|
Circulatory Stasis |
*Atrial fibrillation * LV dysfunction *Immobility or paralysis *Venous insufficiency or varicose veins *Venous obstruction from tumor, obesity, or pregnancy |
|
|
Vessel Wall Injury |
*Trauma or surgery * Venepuncture *Chemical irritation *Heart valve disease or replacement *Atherosclerosis *Indwelling catheters |
|
|
Hypercoagulability |
*Malignancy *Pregnancy and peri-partum period *Estrogen therapy *Trauma or surgery of lower extremity, hip, abdomen or pelvis *Inflammatory bowel disease *Nephrotic syndrome *Sepsis *Thrombophilia |
|
|
Antithrombotic Drugs |
1) Anticoagulants 2) Antiplatelet drugs 3) Fibrinolytic agents (thrombolytic) |
|
|
Anticoagulants |
*Drugs that reduce fibrin formation
|
|
|
2 Mechanisms of Anticoagulants |
1) Inhibits activity of clotting factors -heparin and its derivatives -factor Xa inhibitors -direct thrombin inhibitors
2) Inhibits synthesis of clotting factors -warfarin |
|
|
5 Types of Anticoagulants |
1) Unfractionated heparin (UFH) 2) Low molecular weight heparin (LMWH) 3) Factor Xa inhibitors 4) Director thrombin inhibitors 5) Vitamin K antagonists |
|
|
Unfractionated Heparin (UFH) |
-Long polysaccharide chain -Heavier than LMWH -Complexes with AT3 leading to more efficient inactivation of thrombin and factor Xa -Prevents conversion of fibrinogen to fibrin |
|
|
Unfractionated Heparin MOA |
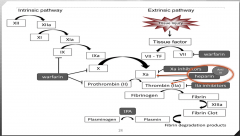
|
|
|
Heparin Indications |
*Prevention and treatment of venous thrombosis (PE & DVT) *Prevention of coagulation in devices of extracorporeal circulation *DIC *Preferred agent during pregnancy *Dose in UNITS (not mL) |
|
|
Heparin Pharmacokinetics |
*Administered IV or SQ *Onset: immediate (IV),30 mins (SQ) *Metabolized hepatically, renal excretion *Half life: 90 mins |
|
|
Heparin Monitoring |
*Routine *aPTT *Goal: 1.5 or 2x the baseline *Normal value is 40 or less |
|
|
Heparin Adverse Events |
*Bleeding (reverse with protamine) *Hypersensitivity reactions *Heparin-induced thrombocytopenia (HIT) *Contraindications: uncontrolled bleeding, thrombocytopenia, previous hypersensitivity |
|
|
Heparin-Induced Thrombocytopenia (HIT) |
*Serious immune-mediated reduction in platelets *Antibody formation against heparin-platelet complex *Platelets decrease *Develops after 5-10 days of therapy *Paradoxical increase in thrombosis *Treatment: discontinue heparin, start alternative coagulation since pt still at risk for clotting |
|
|
Low Molecular Weight Heparin (LMWH) Drugs |
1) Enoxaparin (Lovenox)-most common 2) Dalteparin (Fragmin) 3) Tinzaparin (Innohep) |
|
|
LMWH Chemistry |
*Short polysaccharide chain (aka lower weight) |
|
|
LMWH MOA |
*Complexes with AT3 leading to inactivation of factor Xa *Decreased inactivation of thrombin due to smaller size *Prevents conversion of fibrinogen to fibrin |
|
|
LMWH MOA Image |
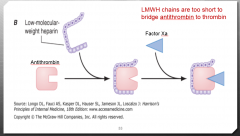
|
|
|
LMWH Indications |
*Prevention and treatment of venous thrombosis (PE & DVT) *Prevention of ischemic complications in acute coronary syndromes *Dosed in mL or IU |
|
|
LMWH Pharmacokinetics |
*Administered SQ *Half-life: up to 6 times longer than standard heparin *Renally excreted |
|
|
LMWH Adverse Events |
*Bleeding and HIT *Black box warning (epidural and spinal hematomas) *Requires adjustment for renal impairement *Incomplete reversal with promatin (60-75%) |
|
|
LMWH Monitoring |
*No routine monitoring necessary *More predictable anticoagulant activity *Signs of bleeding, CBC, serum creatinine *Anti-Xa levels in special populations |
|
|
Factor Xa Inhibitor Drugs |
1) Fondaparinux (Arixtra) 2) Apixaban (Eliquis) 3) Rivaroxaban (Xarelto) |
|
|
Fondaparinux (Arixtra) Pharmacokinetics |
*Pentasaccharide *Selective inhibition of factor Xa indirectily via AT3 *Administered SQ *Indications: Prevent and treat venous thrombosis *100% bioavailable, lasting 17-21 hours |
|
|
Fondaparinux (Arixtra) Adverse Events |
*Bleeding *Not reversed by protamine (NON REVERSIBLE) *Black box warning (epidural and spinal hematomas) *Does not lead to HIT |
|
|
Fondaparinux (Arixtra) Monitoring |
*Signs of bleeding *CBC, serum creatinine *Routine monitoring not necessary
|
|
|
Apixaban (Eliquis) Pharmacokinetics |
*Direct and selective inhibition of factor Xa *Administered orally *Half-life: 8-12 hours with normal renal function
|
|
|
Apixaban (Eliquis) Indications |
*Prevention of stroke/systemic embolism in afib *Initial treatment of VTE, extended treatment of VTE to reduce risk of recurrance and DVT/PE |
|
|
Apixaban (Eliquis) Adverse Events |
*Bleeding *Caution: epidural/spinal hematomas *Contrindications: severe renal/liver dysfunction, active bleeding |
|
|
Apixaban (Eliquis) Monitoring |
*Route monitoring not necessary *CBC, renal and liver function, signs of bleeding *May affect anti-Xa activity, PT/PTT/INR |
|
|
Rivaroxaban (Xarelto) MOA |
*Direct and selective inhibition of factor Xa |
|
|
Rivaroxaban (Xarelto) Pharmacokinetics |
*Adminstered orally *Half-life: 5-9 hrs with normal renal function *Indications: prevention of VTE after hip/knee replacement, treatment of VTE, prevention of stroke/systemic embolism in afib |
|
|
Rivaroxaban (Xarelto) Adverse Effects |
*Bleeding (possibly reversed by PCC or factor VIIIa) *Black box warning: epidural and spinal hematomas *Contraindications: several renal/liver dysfunction, active bleeding |
|
|
Rivaroxaban (Xarelto) Monitoring |
*Routine monitoring not necessary *CBC, renal and liver function, signs of bleeding *May affect anti-Xa actiity, PT/PTT/INR |
|
|
Direct Thrombin Inhibitor Drugs |
1) Hirudin (natural agent) 2) Argatroban 3) Bivalirudin 4) Lepirudin (removed from market) 5) Dabigatran (oral agent) |
|
|
Argatroban Indications |
*Prophylaxis and treatment of thrombosis in patients with HIT |
|
|
Argatroban Pharmacokinetics |
*Immediate onset *Half-life: 45 mins *Metabolized in liver *Not available in bolus dosing |
|
|
Argatroban Adverse Events |
*Bleeding *Allergic reactions |
|
|
Argatroban Monitoring |
*Goal: 1.5-3x baseline value *Gives FALSE ELEVATION of INR |
|
|
Bivalirudin Indications |
*Alternative to heparin in patients undergoing coronary angiography |
|
|
Bivalirudin Pharmacokinetics |
*Immediate onset *Half-life: 25mins *Renally eliminated *Available in bolus dosing |
|
|
Bivalirudin Adverse Events |
*Bleeding (less than heparin) |
|
|
Bivalirudin Monitoring |
*ACT *Dosing and ACT monitoring depends on stage of surgery |
|
|
Dabigatran MOA |
Oral prodrug that undergoes rapid conversion to active drug |
|
|
Dabigatran Indications |
Prevention of stroke/systemic embolism in afib |
|
|
Dabigatran Pharmacokinetics |
Half-life: 13-18 hours |
|
|
Dabigatran Adverse Events |
*Bleeding *Dyspepsia (indigestion) *Non reversal |
|
|
Dabigatran Monitoring |
*Routine monitoring not necessary *May affect PT/PTT/INR *CBC, real function, signs of bleeding |
|
|
Vitamin K Antagonists: Warfarin MOA |
Inhibits vitamin K-epoxide reductase resulting in decreased production of vitamin K dependent clotting factors (2,7,9,10) as well as protein C and S (natural anticoagulants) |
|
|
Warfarin Indications |
*Prevention and treatment of VTE *Prevention of stroke/systemic embolism in afib *Preferred oral anticoagulant in patient with mechanical valves |
|
|
Warfarin Pharacokinetics |
*Rapid and complete absorption *99% bound to albumin *Peak at 4 hours *Onset: 3-7 days *Half-life: 2 days *Metabolism: hepatic via CYP 450 |
|
|
Warfarin Monitoring |
*PT used to assess anticoagulant effects *Goal INR: 2-3 (2.5-3.5 for mechanical valves) *Monitored daily during initiation (1-4 weeks once stable) *CBC, signs of bleeding |
|
|
Warfarin Adverse Effects |
*Bleeding *Teratogenic (can't be given to preg pts!) *Skin necrosis (rare) |
|
|
Warfarin Reversal |
*Vitamin K *FFP *Fresh whole blood *Prothrombin complex concentrates (PCC) *Recombinant factor 7a |
|
|
Warfarin Drug Interations |
1) Increase anticoagulant effects 2) Promote bleeding 3) Decrease anticoagulant effects (increase clotting risk) |
|
|
Warfarin Drug Interactions Chart |

|
|
|
Half-Life of Clotting Factors |
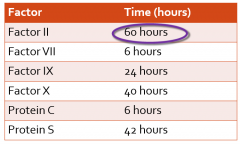
Important because new factors inhibited but existing ones already active |
|
|
INR Calculation |

|
|
|
4 Types of Antiplatelets |
1) Aspirin 2) Adenosine diphosphate receptor antagonists 3) GP 2b/3a receptor antagonists 4) Other: dipyridamole, cilostazol |
|
|
Aspirin MOA |
Irreversible inhibitor of cyclooxygenase (required for thomboxane synthesis) and suppresses platelet aggregation |
|
|
Aspirin Indications |
*Primary/secondary prevention of MI *Primary/secondary prevention of stroke
*Dose: 81 or 325 mg daily |
|
|
Aspirin Adverse Events |
*GI bleeding *Hemorrhagic stroke |
|
|
ADP Receptor Antagonist Drugs |
1) Clopidogrel (plavix) 2) Prasugrel (effient) 3) Ticlopidine (ticlid) 4) Ticagrelor (brilinta) |
|
|
ADP Receptor Antagonist MOA |
*Clopidogrel, Prasugrel, Ticlopidine: irreversible ADP receptor antagonists
*Ticagrelor: reversible ADP receptor antagonists |
|
|
ADP Receptor Antagonist Indication |
*Prevent restenosis of coronary stents *Reduce thombotic complications in ACS
|
|
|
ADP Receptor Antagonist Adverse Events |
*Bleeding *Ticlopidine (rarely used): neutropenia/agranulocytosis |
|
|
GP 2b/3a Receptor Antagonist Drugs |
1) Abciximab 2) Tirofiban 3) Eprifibatide |
|
|
GP 2b/3a Receptor Antagonist Pharmacokinetics |
*Most effective antiplatelet agents available *Administered IV |
|
|
GP 2b/3a Receptor Antagonist Indications |
*Prevention of ischemic events during percutaneous coronary intervention |
|
|
GP 2b/3a Receptor Antagonist Adverse Events |
*Bleeding |
|
|
Other: Dipyridamole MOA |
*Prevents platelet aggregation by increasing plasma levels of adenosine |
|
|
Other: Dipyridamole Adverse Events |
*GI upset *Bleeding |
|
|
Other: Cilostazol MOA |
Inhibits phosphodiesterase 3 in platelets and blood vessels causing decreased aggregation and vasodilation |
|
|
Other: Cilostazol Indication |
*Intermittent claudication (pain in legs while walking) |
|
|
Other: Cilostazol Adverse Events |
*Headache *Diarrhea *Palpitations *Edema |
|
|
Thrombolytics |
Used to break down a currently existing clot |
|
|
Thrombolytic Agents |
1) Streptokinase 2) Tenecteplase 3) Alteplase (tPA)-used at Rush 4) Reteplase |
|
|
Thrombolytic MOA |
Promotes conversion of plasminogen to plasmin which is an enzyme that degrades fibrin (clots) |
|
|
Thrombolytic Indications |
*Acute coronary thrombosis *DVT *Ischemic stroke |
|
|
Thrombolytic Pharmacokinetics |
*Rapid onset *Short half-life *Several contraindications to administration |
|
|
Thrombolytic Adverse Events |
*Bleeding |
|
|
Thrombolytic Comparison Chart |
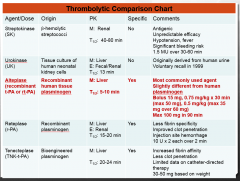
|

