![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
20 Cards in this Set
- Front
- Back
|
Explain the major responsibilities of a physician in prescribing a drug
|
a. Mechanism of action
b. Indications- When to use. Is this particular use approved for this drug by FDA? c. Contraindications d. Adverse effects (side effects)- Common cause for non-compliance |
|
|
Explain the interrelationship between pharmacodynamics and pharmokinetics
|
Pharmacodynamics is the study of the effects of drugs on tissues.
Pharmacokinetics is the study of the effects of tissues on drugs. |
|
|
List patient factors that can effect drug actions
|
a.Age
b.Body weights c.Pregnancy d.Pathological conditions e.Pharmacogenetics- polymorphic proteins can result in idosyncratic drug reactions |
|
|
Tolerance
|
repeated administration at a given dose results in decreased effects
|
|
|
Pharmacodynamic tolerance
|
reduction in normal reactivity of the drug receptor
|
|
|
Pharmacokinetic tolerance
|
drug reduces its own absorption and/or elimination
|
|
|
Tachyphylaxis
|
very rapid, acute tolerance
|
|
|
Additive effect-
|
(+) when combined effect of two different drugs equals the sum of their individual effects (typical with opiods)
|
|
|
Potentiation (or synergy)
|
(+) when combined effect is greater than the sum of the individual effects of two drugs
|
|
|
Pharmacological antagonism
|
(-) a drug blocks a receptor that another drug activates
|
|
|
Physiological antagonism
|
(-) two drugs act at different sites to produce opposite effects (norep at adrenergic and hist at other receptors)
|
|
|
Biochemical antagonism
|
(-) a drug reduces the effective concentration of another drug (warfarin and phenobarbitol)
|
|
|
Chemical antagonism
|
(-) two drugs combine to form an inactive product (lead poisoning treated with chelator)
|
|
|
Explain the concept of bioavailability
|
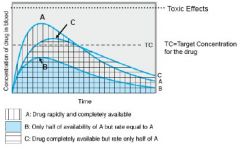
rate and extent of drug absorption into the bloodstream
|
|
|
Explain the differences between generic and proprietary drugs, and describe the process of naming drugs
|
proprietary drugs are names copyrighted by a single manufacturer and one drug can have several trademark/proprietary names, whereas one drug can have only one generic name.
|
|
|
Phase I
|
Is it safe?
i. Pharmokinetics ii. Non-toxic |
|
|
Phase II
|
Does it work in patients?
i. Efficacy |
|
|
Phase III
|
Does it work, double blind?
i. Higher numbers of patients |
|
|
Phase IV
|
Post-marketing surveillance
i. Critical period ii. Look for rare adverse effects iii. Really high numbers of patients |
|
|
List the responsibilites of the FDA
|
a. Must prove that a drug is both safe and effective before approving it
b. Must specify the conditions for which the drug is approved c. Approval of all medical devices d. Post-marketing reviews of adverse effects e. CANNOT regulate herbals and nutritional supplements |

