![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
315 Cards in this Set
- Front
- Back

BRAND NAME
diazepam |
Valium
|
|
BRAND NAME
diphenhydramine HCl |
Benadryl
|
|

CLASS
diazepam |
benzodiazepine
|
|
|
BRAND NAME
charcoal, activated (without sorbitol) |
Charcola, Actidose-aqua
|
|

MECHANISM OF ACTION
diazepam |
Acts on parts of the limbic system, the thalamus, and hypothalamus producing calming effects; decreases seizures by increasing the seizure threshold: transient analgesia, amnesic, sedative.
|
|
CLASS
diphenhydramine HCl |
antihistamine, anticholinergic
|
|

INDICATIONS
diazepam |
Grand mal seizures, especially status epilepticus.
Transient analgesia/amnesia for medical procedures (eg fracture reduction, cardioversion, pacing). Delirium tremens. Treat the cause first. |
|
BRAND NAME
vasopressin |
Vasopressin
|
|

CONTRAINDICATIONS
diazepam |
Hypersensitivity (allergy).
Glaucoma, acute narrow angle (relative). |
|
MECHANISM OF ACTION
diphenhydramine HCl |
Blocks cellular histamine receptors, but does not prevent histamine release; results in decreased capillary permeability and decreased vasodilation, as well as prevention of bronchospasm.
Has some anticholinergic effects. |
|

ADVERSE REACTIONS
diazepam |
CV: Bradycardia, hypotension, cardiovascular collapse, venous thrombosis, phlebitis, vascular spasm.
RESP: Respiratory arrest; may be prolonged in neonates. CNS: Somnolence, confusion, coma, diminished reflexes; s/s may be prolonged in neonates. OTHER: Burning proximal to IV injection site; local irritation, swelling, extravasation will cause tissue necrosis. |
|
|
CLASS
charcoal, activated (without sorbitol) |
adsorbent
|
|

ADULT DOSAGE
diazepam |
2 mg increments slow IV/IO push. Administer no faster than 2 mg/min.
|
|
INDICATIONS
diphenhydramine HCl |
Anaphylaxis (2nd line)
Phenothiazine reactions (extrapyramidal symptoms) Antiemetic |
|

PEDIATRIC DOSAGE
diazepam |
IV/IO Dose: 0.2-0.3 mg/kg every 15-30 min. (max of 1.0 mg/kg); administer IV/IO over at least 3 min or until seizure activity subsides.
IV/IO Dose after rectal dose: 0.1 mg/kg with same administration instructions. RECTAL DOSE (≤ 6 years): 0.3-0.5 mg/kg rectally at IV push rate; may repeat in 15-30 min at 0.25 mg/kg. |
|
BRAND NAME
adenosine |
Adenocard
|
|

BONUS
diazepam |
Do not mix or dilute diazepam with other solutions or drugs in syringe, tubing or IV container.
Valium must be injected slowly and small veins such as those on the dorsum of the hand or wrist should be avoided. Risk of venous thrombosis, phlebitis, local irritation, swelling and vascular spasm is increased. Extreme care must be taken to avoid intra-arterial injection or extravasation. Diazepam may cause prolonged CNS depression in the neonate (30 days of age or less). |
|
CONTRAINDICATIONS
diphenhydramine HCl |
Known hypersensitivity to diphenhydramine or drugs of similar chemical structure.
Newborn or premature infants; nursing mothers. Considerable caution in patients with glaucoma, acute narrow angle; stenosing or obstructive diseases of the GI tract; bronchial asthma; hyperthyroidism; cardiovascular disease or HTN; age greater than 60 years (all relative benefit vs. risk). |
|

BRAND NAME
lorazepam |
Ativan
|
|
|
MECHANISM OF ACTION
charcoal, activated (without sorbitol) |
PHARMACOLOGICAL: Physical binding (adsorption) of toxins from GI tract.
CLINICAL EFFECTS: Prevents/reduces systemic absorption of toxins. |
|

CLASS
lorazepam |
Antianxiety, benzodiazepine, short or intermediate acting.
|
|
ADVERSE REACTIONS
diphenhydramine HCl |
CV: Hypotension, palpitations, arrhythmias, hemolytic anemia.
RESP: Anaphylaxis, thickening of bronchial secretions, tightness in chest, wheezing, nasal stuffiness. CNS: Sedation, visual disturbances, seizures. GU/GI: Urinary frequency or retention; vomiting. CHILDREN: In children, may cause paradoxical CNS excitation, seizures, palpitations, thickening of bronchial secretions. |
|

MECHANISM OF ACTION
lorazepam |
Agent binds highly to the gamma-aminobutyric acid (GABA) benzodiazepine receptor complex without displacing GABA, (GABA is the major inhibitory neurotransmitter in the brain). It exerts tranquilizing action on the CNS.
|
|
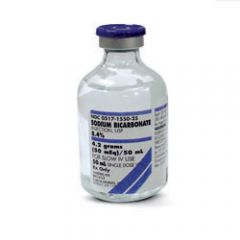
BRAND NAME
sodium bicarbonate 8.4% |
Sodium Bicarbonate 8.4%
|
|

INDICATIONS
lorazepam |
Status epilepticus
Seizure Agitation |
|
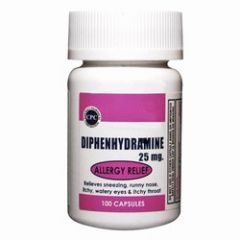
ADULT DOSAGE
diphenhydramine HCl |
ANAPHYLAXIS: 25-50 mg slow IV push or deep IM.
EXTRAPYRAMIDAL SYMPTOMS AND ANTIEMETIC: 10-50 mg IV or deep IM, dose individualized according to the needs and pt response. |
|

CONTRAINDICATIONS
lorazepam |
Known sensitivity to benzodiazepines.
Acute narrow glaucoma or myasthenia gravis. Known hypersensitivity to polyethylene glycol, propylene glycol, or benzyl alcohol. Pregnancy. |
|
|
INDICATIONS
charcoal, activated (without sorbitol) |
Sole prehospital therapy of oral ingestion of toxic substances.
Pre-lavage of oral ingestions of toxic substances. |
|

ADVERSE REACTIONS
lorazepam |
Most frequent adverse reaction is sedation.
Transient amnesia or memory impairment. Confusion Hypotension Respiratory depression Dizziness, HA |
|
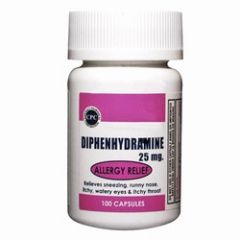
PEDIATRIC DOSAGE
diphenhydramine HCl |
1.0-1.25 mg/kg slow IV push; may also be given deep IM.
|
|

ADMINISTRATION
lorazepam |
IM: Should be administered deep into the muscle mass.
IV/IO: Do not exceed 2 mg/min or 0.05 mg/kg over 2-5 min. **Inadvertent intra-arterial injection may produce arteriospasm resulting in necrosis and potential amputation.** |
|
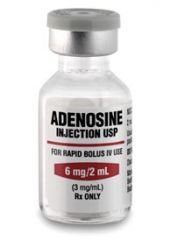
CLASS
adenosine |
antiarrhythmic, endogenous nucleoside
|
|

ADULT DOSAGE
lorazepam |
Status epilepticus: 2-4 mg IV/IO given slowly (over 2-5 min). May repeat dose in 10-15 min if needed (max 8 mg). May give IM if no IV access.
|
|
BONUS
diphenhydramine HCl |
Not used in newborn or premature infants; used in pregnancy only if clearly needed.
In anaphylaxis, used as a 2nd line treatment after epi and steroids. |
|

PEDIATRIC DOSAGE
lorazepam |
Status epilepticus: 0.05-0.1 mg/kg IV/IO over 2-5 min (max 4 mg). May repeat in 10-15 min.
|
|
|
CONTRAINDICATIONS
charcoal, activated (without sorbitol) |
Ingestion of caustics
Ingestion of hydrocarbons (relative) Oral administration to comatose patient Simultaneous administration of other oral drugs |
|

BONUS
lorazepam |
Concomitant use of CNS sedatives such as phenothiazines, narcotic analgesics, barbituates, antidepressants, and alcohol should be assessed prior to administration of IV lorazepam.
Care must be used when administering lorazepam IV to elderly patients, seriously ill patients, and those with limited pulmonary reserve as apnea and/or cardiac arrest may occur. Patients over the age of 50 years may have a more profound and prolonged sedation with IV lorazepam. All patients should be monitored for respiratory depression and hypotensive effects. Stability above room temperature has not been established. |
|
BRAND NAME
albuterol sulfate |
Proventil, Ventolin
|
|
BRAND NAME
midazolam hydrochloride |
Versed
|
|
CLASS
vasopressin |
pituitary (antidiuretic) hormone
|
|
CLASS
midazolam hydrochloride |
CNS depressant, benzodiazepine
|
|
CLASS
albuterol sulfate |
sympathomimetic, bronchodilator
|
|

MECHANISM OF ACTION
midazolam hydrochloride |
CNS effects are mediated through the inhibitory neurotransmitter gamma-aminobutyric acid (GABA).
Acts at the limbic, thalamic, and hypothalamic levels of the CNS, producing anxiolytic, sedative, hypnotic, and anticonvulsant effects. Capable of producing all levels of CNS depression, from mild sedation to coma. |
|
|
ADVERSE REACTIONS
charcoal, activated (without sorbitol) |
May provoke vomiting
May worsen overdosed induced Ileus |
|
INDICATIONS
midazolam hydrochloride |
Anti-convulsant
Sedation Management of acute agitation/treat cause first. Induction for intubation. |
|
MECHANISM OF ACTION
albuterol sulfate |
β antagonist (primarily β2); relaxes bronchial smooth muscle, resulting in bronchodilation; also relaxes vascular and uterine smooth muscle; decreases airway resistance.
|
|
CONTRAINDICATIONS
midazolam hydrochloride |
Hypersensitivity to midazolam.
Relative contraindication in: Myasthenia gravis or other neuromuscular disorders; acute alcohol intoxication, severe COPD, and acute pulmonary insufficiency. |
|
MECHANISM OF ACTION
adenosine |
Slows conduction time through the AV node; can interrupt re-entrant pathways through the AV node.
Slows sinus rate. Larger doses decrease BP by decreasing peripheral resistance. |
|
ADVERSE REACTIONS
midazolam hydrochloride |
CV: Hypotension (especially in patients premedicated with narcotics); cardiac arrest, irregular or fast heartbeat.
RESP: Apnea, respiratory depression, respiratory arrest, hyperventilation, wheezing or difficulty breathing, hiccups, coughing. CNS: Emergency delirium, muscle tremor, uncontrolled or jerky movements of the body, unusual excitement, irritability, or restlessness, dizziness, light-headedness, or feeling faint, prolonged drowsiness, HA GI: N/V |
|
INDICATIONS
albuterol sulfate |
Treatment of bronchospasm
|
|
ADULT DOSAGE
midazolam hydrochloride |
PATIENTS 14-60 YEARS OF AGE: 1-5 mg IV, titrate to effect, administer slowly in small increments of no more than 2.5 mg over at least 2 min, or 2-5 mg IM.
PATIENTS OVER 60 YEARS OF AGE: 1-3.5 mg IV, titrate to effect, administer slowly in small increments of no more than 1.5 mg over at least 2 min, or 1-3 mg IM. TOTAL DOSE: Should not exceed 20 mg. FOR EMERGENCY INTUBATION: 0.1 mg/kg up to 0.3 mg/kg with dosage limit of 20 mg. SEIZURES: 0.2 mg/kg IM for status seizures if no IV access. |
|
|
INCOMPATIBILITES/DRUG INTERACTIONS (you have to add this one)
charcoal, activated (without sorbitol) |
Ineffective for iron, lithium, heavy metals, and other ions.
May reduce the effectiveness of other treatments (Mucomyst) in pure acetaminophen OD's. Since charcoal bonds with whatever it is mixed with, flavoring with drinks reduces effectiveness. |
|
PEDIATRIC DOSAGE
midazolam hydrochloride |
Pediatric patients: 0.05-0.1 mg/kg slow IV push.
0.2 mg/kg IM for status seizures if no IV access. |
|
CONTRAINDICATIONS
albuterol sulfate |
Synergistic with other sympathomimetics.
Use caution in pts with diabetes, hyperthyroidism, and cerebrovascular disease. |
|
BONUS
midazolam hydrochloride |
Midazolam may potentiate other CNS depressants, including opiate agonists or other analgesics, barbiturates or other sedatives, anesthetics, or alcohol.
Erythromycin may double the half-life of midazolam. Flumazenil is the accepted antagonist for midazolam. |
|
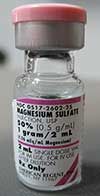
BRAND NAME
magnesium sulfate |
Magnesium Sulfate
|
|
|
BRAND NAME
morphine sulfate |
Morphine Sulfate
|
|
ADVERSE REACTIONS
albuterol sulfate |
CV: Dysrhythmias, tachycardia (with excessive use), peripheral vasodilation.
RESP: Bronchospasm (rare paradoxical with excessive use). CNS: Tremors, nervousness GI: N/V ENDOCRINE: Hyperglycemia |
|
|
CLASS
morphine sulfate |
narcotic agonist
|
|
|
ADULT DOSAGE
charcoal, activated (without sorbitol) |
30 - 60 gm (1-2 gm/kg) PO or OG/NG tube; if not in pre-mixed slurry, mix one part charcoal with four parts water.
|
|
|
MECHANISM OF ACTION
morphine sulfate |
Alleviates pain by acting on the pain receptors in the brain; elevates pain threshold.
Depresses CNS, depresses brainstem respiratory centers, decreases responsiveness to changes in PaCO2. Increases venous capacitance (venous pooling), vasodilates arterioles, reducing preload and afterload. Histamine release. |
|
ADULT DOSAGE
albuterol sulfate |
Give 2.5 mg of premixed solution for inhalation (0.083%) via SVN, or in-line with a ventilatory device. Repeat doses per medical control.
|
|
|
INDICATIONS
morphine sulfate |
Analgesia, especially in patients with burns, MI, or renal colic.
Pulmonary edema (cardiogenic). |
|
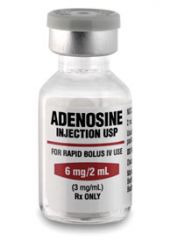
INDICATIONS
adenosine |
Conversion of SVTs with no known AFib or AFlutter
Wide complex tachycardia of uncertain origin unresponsive to lidocaine. |
|
|
CONTRAINDICATIONS
morphine sulfate |
Respiratory depression.
Head injuries. Elevated ICP. Asthma, relative. Abdominal pain, relative. |
|
PEDIATRIC DOSAGE
albuterol sulfate |
For children < 40 lbs., administer half of 0.083% premixed solution; add 1-1.5 mL NS to make 2.5-3 mL treatment administered via SVN, O2 mask or in-line with a ventilatory device. Repeat doses per medical control.
|
|
|
ADVERSE REACTIONS
morphine sulfate |
CV: Brady or tachydysrhythmias, orthostatic hypotension.
RESP: Respiratory depression or arrest. CNS: Excess sedation, seizures to coma and arrest, pupillary constriction. GI: N/V, GI spasm. DERM: Histamine release may cause local urticaria. |
|
|
PEDIATRIC DOSAGE
charcoal, activated (without sorbitol) |
0.5-1.0 gm/kg PO or OG/NG tube; if not in pre-mixed slurry, mix one part charcoal with four parts water.
|
|
|
ADULT DOSAGE
morphine sulfate |
Administer 1-3 mg increments slow IV/IO push (over 1-3 min) until desired effect.
|
|
BONUS
albuterol sulfate |
ROUTES OF ADMINISTRATION
Nebulized, mouth piece or in-line via mask Inhaler, patients own In-line with ET tube/nasotracheal tube SPECIAL NOTES Must be sulfite-free |
|
|
PEDIATRIC DOSAGE
morphine sulfate |
100-200 mcg/kg (0.1-0.2 mg/kg) slow IV/IO push.
|
|
MECHANISM OF ACTION
vasopressin |
Causes vasoconstriction (pressor effect) of peripheral, cerebral, pulmonary, and coronary vessels.
|
|
|
BONUS
morphine sulfate |
CNS side effects (including respiratory depression) can be reversed by naloxone.
Schedule II narcotic. Watch for histamine effects (wheals, urticaria) proximal to IV site, contact medical control. Correct hypotension before administration. Maximum respiratory depression 7-10 min after administration; can be reversed with naloxone, caution in patients with emphysema. IV infusions of morphine sulfate may be transported, however an infusion pump is required. |
|

BRAND NAME
dexamethasone sodium phosphate |
Decadron
|
|
BRAND NAME
succinylcholine |
Succinylcholine
|
|
|
BONUS
charcoal, activated (without sorbitol) |
Give it!
Charcoal mixture SHOULD NOT contain sorbitol; CAREFULLY read label of exchange supply. |
|
CLASS
succinylcholine |
ultra-short-acting depolarizing-type skeletal muscle relaxant
|
|

CLASS
dexamethasone sodium phosphate |
synthetic adrenocorticoid/glucocorticoid with a predominance of glucocorticoid action, anti-inflammatory.
|
|
MECHANISM OF ACTION
succinylcholine |
Combines with cholinergic receptors of the motor end plate to produce depolarization.
Hydrolyzed by acetylcholinesterase. |
|
CONTRAINDICATIONS
adenosine |
Known AFib or AFlutter.
Sick sinus syndrome, 2nd or 3rd degree AV blocks; except in pts with a functioning ventricular pacemaker. Use cautiously in patients with known asthma (has precipitated acute bronchospasm). Patients on theophylline and related methylxanthines. Patients on dipyridamole (Persantine) or carbamazepine (Tegretol). Cardiac transplant patients are more sensitive to adenosine and require only a small dose (relative). Pregnancy (no controlled studies). |
|
INDICATIONS
succinylcholine |
Endotracheal intubation requiring paralysis (RSI) by a qualified EMT-P.
|
|

MECHANISM OF ACTION
dexamethasone sodium phosphate |
Improves lung function and myocardial performance
Decrease in pulmonary edema Relaxation of bronchospasm |
|
CONTRAINDICATIONS
succinylcholine |
Muscle disorders
Personal or family history of malignant hyperthermia History of hyperkalemia Burn injured patients* Ocular injuries Patients in whom successful endotracheal intubation is doubtful. |
|
|
BRAND NAME
dextrose 50% |
Dextrose 50%, D50
|
|
ADVERSE REACTIONS
succinylcholine |
Vagal stimulation leading to bradycardia or asystole
Hyperkalemia Rhabdomyolysis Hypersalivation Elevated intraocular pressure Release of histamine |
|

INDICATIONS
dexamethasone sodium phosphate |
Reactive airway disease: Acute exacerbation of bronchial asthma.
Anaphylaxis. Cerebral edema (non-traumatic) |
|
ADULT DOSAGE
succinylcholine |
1.5 mg/kg IV push, may repeat in 2-3 min to achieve paralysis
|
|
CLASS
sodium bicarbonate 8.4% |
buffer
|
|
PEDIATRIC DOSAGE
succinylcholine |
NONE
|
|

CONTRAINDICATIONS
dexamethasone sodium phosphate |
Systemic fungal infections.
Hypersensitivity to any component of dexamethasone, including sulfites. Preterm infants. |
|
BONUS
succinylcholine |
Beta-blockers, procainamide, lithium, and quinidine prolong the effects.
Muscle paralysis lasting 4-6 min. *Succinylcholine should not be given to patients for the period from 24 hours to 21 days after significant burns or crush injury due to elevated potassium levels and potential for cardiac dysrhythmias. |
|
|
CLASS
dextrose 50% |
carbohydrate, hyperglycemic
|
|
|
BRAND NAME
nitrous oxide 50% |
Nitronox
|
|

ADVERSE REACTIONS
dexamethasone sodium phosphate |
Sodium retention, fluid retention, potassium loss, hypokalemic alkalosis, HTN, convulsions, hyperglycemia, myocardial rupture following recent MI.
|
|
|
CLASS
nitrous oxide 50% |
analgesic, inhalation
|
|
ADVERSE REACTIONS
adenosine |
CV: TRANSIENT dysrhythmias (systole, bradycardia, PVC's) occur in 55% of pts (none reported as irreversible).
Palpitations, chest pressure, CP, hypotension, transient HTN; facial flushing, sweating. RESP: Dyspnea, hyperventilation, tightness in throat, bronchospasm. CNS: lightheadedness, HA, dizziness, paresthesia, apprehension, blurred vision, neck-back pain. GI: nausea, metallic taste. |
|
|
MECHANISM OF ACTION
nitrous oxide 50% |
Centrally acting agent that produces CNS depression and elevation of the pain threshold.
|
|

ADULT DOSAGE
dexamethasone sodium phosphate |
REACTIVE AIRWAY DISEASE, ANAPHYLAXIS: 8-24 mg IV/IO or deep IM.
CEREBRAL EDEMA: 1-5 mg/kg IV/IO or deep IM. |
|
|
INDICATIONS
nitrous oxide 50% |
Moderate to severe pain from musculoskeletal trauma, burns, AMI.
|
|
|
MECHANISM OF ACTION
dextrose 50% |
PHARMACOLOGICAL: Aerobic metabolic substrate (ATP production).
CLINICAL EFFECTS: Reverses CNS effects of hypoglycemia by rapidly increasing serum glucose levels. Provides short-term osmotic diuresis. |
|
|
CONTRAINDICATIONS
nitrous oxide 50% |
Unconscious patient
Poor respiratory drive, compromise of respiratory status (ie pneumothorax) Abdominal pain unless intestinal obstruction has been completely ruled out Severe head injury |
|

PEDIATRIC DOSAGE
dexamethasone sodium phosphate |
REACTIVE AIRWAY DISEASE, ANAPHYLAXIS: 0.25-0.5 mg/kg IV/IO
CEREBRAL EDEMA: 0.5-1.5 mg/kg IV/IO |
|
|
ADVERSE REACTIONS
nitrous oxide 50% |
RESP: Worsening of pre-existing pneumothorax; may cause hypercarbia in oxygen sensitive patients (CO2 retainers).
GI: N/V, may lead to rupture of the intestine if administered to a patient with intestinal obstruction. |
|
INDICATIONS
vasopressin |
Adult shock-refractory VFib/pulseless VTach
|
|
|
ADULT DOSAGE
nitrous oxide 50% |
Self-administered and self-regulated by the patient, who must hold the mask to the face to create an airtight seal until the pain is significantly relieved or the patient drops the mask.
|
|

BONUS
dexamethasone sodium phosphate |
Dexamethasone is not compatible with benadryl or versed in IV tubing.
|
|
|
PEDIATRIC DOSAGE
nitrous oxide 50% |
Must be old enough to follow the instructions for use and large enough that the mask creates an airtight seal.
|
|
|
INDICATIONS
dextrose 50% |
Known hypoglycemia
ALOC of unknown etiology Seizures of unknown etiology Hyperkalemia |
|
|
BONUS
nitrous oxide 50% |
DOSAGE FORMS:
Nitrous oxide machine that delivers a fixed ration of nitrous oxide and oxygen (50:50) utilizing a mixing valve with separate tanks of N2O and O2 with a fail safe device (O2 tank empty, no nitrous flows) and a self-administration mask. |
|
BRAND NAME
methylprednisolone sodium succinate |
Solu-Medrol
|
|
BRAND NAME
etomidate |
Etomidate
|
|
ADULT DOSAGE
adenosine |
INITIAL: 6 mg rapid IV/IO push over 1-3 seconds
ADMINISTRATION PROCEDURE: 18 ga IV/IO AC↑, press print on monitor, use hub closest to IV catheter to push drug, immediate 20 mL flush in other hub. REPEAT: 12 mg rapid IV/IO push over 1-3 seconds (twice) if needed. Repeat only after 1-2 minutes if no response. ADMINISTRATION PROCEDURE: 2 repeat doses if needed: press print, hub closest to IV catheter to push drug, immediate 20 mL flush in other hub. |
|
CLASS
etomidate |
sedative/hypnotic agent
|
|
CLASS
methylprednisolone sodium succinate |
corticosteroid, glucocorticoid, steroid, anti-inflammatory
|
|
MECHANISM OF ACTION
etomidate |
Produces hypnosis rapidly causing CNS depression and anesthesia.
No analgesic effect |
|
|
CONTRAINDICATIONS
dextrose 50% |
Known thiamine deficiency (relative, if suspected give thiamine close to the same time).
Delirium tremens; use with caution in patients with acute alcoholism, may be ineffective without thiamine. Head injury (unless documented hypoglycemia). Intracranial hemorrhage (relative). Severe pain (paradoxical excitement may occur). |
|
INDICATIONS
etomidate |
Induction of anesthesia for rapid sequence intubation (RSI)
|
|
MECHANISM OF ACTION
methylprednisolone sodium succinate |
Enters target cells and causes many complex reactions that are responsible for its anti-inflammatory and immunosuppressive effects; thought to stabilize cellular and intracellular membranes.
|
|
CONTRAINDICATIONS
etomidate |
Hypersensitivity
|
|
CONTRAINDICATIONS
vasopressin |
Responsive patient with coronary artery disease
Hypertension |
|
ADVERSE REACTIONS
etomidate |
Transient muscle movements
Apnea Causes minimal but not clinically significant suppression of cortisol levels. May cause myoclonus and tremors that can resemble seizure activity |
|
INDICATIONS
methylprednisolone sodium succinate |
Reactive airway disease: Acute exacerbation of emphysema, chronic bronchitis, asthma
Anaphylaxis Burns potentially involving the airway |
|
ADULT DOSAGE
etomidate |
0.3 mg/kg IV over 30-60 seconds
|
|
|
ADVERSE REACTIONS
dextrose 50% |
Cerebral edema in children when given IV undiluted.
Worsening elevated ICP or cerebral edema from trauma or cerebral vascular accident. Extravasation leads to severe tissue necrosis. Slerosing effect on peripheral veins. |
|
PEDIATRIC DOSAGE
etomidate |
NONE
|
|
CONTRAINDICATIONS
methylprednisolone sodium succinate |
Preterm infants
|
|
BONUS
etomidate |
ONSET OF ACTION: 1 min
DURATION OF ACTION: 3-5 min |
|
PEDIATRIC DOSAGE
adenosine |
(Drug of choice for treating SVT in symptomatic infants and children).
INITIAL: 0.1 mg/kg as a rapid IV/IO push. ADMINISTRATION PROCEDURE: Largest IV/IO, press print on monitor, use hub closest to IV catheter to push drug, immediate 2-3 mL flush in other hub. REPEAT: If no response, dose may be doubled 1 time (0.2 mg/kg) using same administration procedure. MAX SINGLE DOSE 12 mg. INFANTS WITH SVT ASSOCIATED WITH SHOCK: Adenosine may precede cardioversion if vascular access is available, but cardioversion should not be delayed while obtaining IV access. |
|
ADVERSE REACTIONS
methylprednisolone sodium succinate |
None from a single dose.
|
|
|
ADULT DOSAGE
dextrose 50% |
HYPOGLYCEMIA, ALOC OR SEIZURES OF UNKNOWN ETIOLOGY: 25-100 mL of D50 (12.5-50 gm, 1 to 2 amps) IV/IO.
HYPERKALEMIA: 50 gm of Dextrose 50% IV total may be given over 1 hour. This is part of a comination drug therapy. See: CaCl and NaHCO3 |
|
ADULT DOSAGE
methylprednisolone sodium succinate |
REACTIVE AIRWAY DISEASE, ANAPHYLAXIS, BURNS POTENTIALLY INVOLVING THE AIRWAY: Usual dose 125 mg slow IV bolus (much larger doses can be used).
|
|
MECHANISM OF ACTION
sodium bicarbonate 8.4% |
Buffers H+ and increases pH.
|
|
PEDIATRIC DOSAGE
methylprednisolone sodium succinate |
REACTIVE AIRWAY DISEASE, ANAPHYLAXIS, BURNS POTENTIALLY INVOLVING THE AIRWAY: 2-4 mg/kg slow IV bolus.
|
|
|
PEDIATRIC DOSAGE
dextrose 50% |
(14 years and below includes infant)
Administer 05.-1 gm/kg of a dextrose 10% solution; recommended to give slowly over a 20 minute period. Dilute D50 (dextrose 50% containing 25 gm of dextrose) to a 1:4 solution. To prepare, take 50 mL out of a 250 mL NS IV bag, then add 50 mL (1 amp) of dextrose 50% into the IV bag. The resultion solution is 10% in NS or 10 gm/100 mL. |
|
BONUS
methylprednisolone sodium succinate |
ADULT DOSAGE CONTINUED - ACUTE SPINAL CORD TRAUMA: Should be within 6 hours of insult and pt meets criteria, inital bolus of 30 mg/kg IV administered over 15 minutes; bolus followed by a 45 min rest period, then a 23-hour continuous infusion of 5.4 mg/kg/hr.
SPECIAL NOTES: Use for spinal cord trauma is limited to prehospital providers that have completed a special training curriculum in accordance with their medical control authorities. PROPER administration of methylprednisolone for spinal cord trauma is imperative. INFUSIONS: An infusion pump is required for continuous infusions of corticosteroids during inter-facility transports; a minimum of microdrip tubing is required for field use if administering loading dose therapy for spinal cord trauma. |
|
BONUS
adenosine |
Adenosine is not blocked by atropine.
Dysrhythmias may recur (short half life). Second dose must be prepared and available. Check for crystallization in cold climates. |
|

BRAND NAME
phenylephrine nasal spray 0.5% |
Neo-synephrine Nasal Spray 0.5%
|
|
|
BONUS
dextrose 50% |
Determine a blood glucose level before initiating administration of dextrose.
Inducing an unnecessary hyperosmolar state during certain illness/injury states (ie head injuries, cerebral edema, intracranial bleeds, etc) may worsen neurological outcome. Additionally dextrose is very necrosing to the vascular system, will cause necrosis if infiltrated and should not be administered through small veins. |
|

CLASS
phenylephrine nasal spray 0.5% |
topical vasoconstrictor
|
|
ADVERSE REACTIONS
vasopressin |
Can increase peripheral vascular resistance and provoke cardiac ischemia and angina pectoris.
Abdominal distress. N/V Tremors. Tissue necrosis if extravasation occurs. |
|

MECHANISM OF ACTION
phenylephrine nasal spray 0.5% |
Stimulates α-receptors in the blood vessels of the nasal mucosa which causes their constriction and thereby decreases the risk of nasal bleeding.
|
|
|
BRAND NAME
glucagon |
Glucagon
|
|

INDICATIONS
phenylephrine nasal spray 0.5% |
Facilitation of nasotracheal intubation.
|
|
BRAND NAME
diltiazem |
Cardizem
|
|

CONTRAINDICATIONS
phenylephrine nasal spray 0.5% |
Known allergy to medication.
|
|
|
CLASS
glucagon |
pancreatic hormone, polypeptide, hyperglycemic agent
|
|

ADVERSE REACTIONS
phenylephrine nasal spray 0.5% |
(rare with single dose, rarely absorbed systemically from nasal instillation)
CV: HTN, palpitations CNS: Tremors |
|
CLASS
magnesium sulfate |
electrolyte, tocolytic
|
|

ADULT DOSAGE
phenylephrine nasal spray 0.5% |
2-4 sprays in each nostril before attempting tube insertion.
|
|
|
MECHANISM OF ACTIONS
glucagon |
PHARMACOLOGIC: Acts only on liver glycogen, converting it to glucose. Counteracts the effect of insulin. Relaxes GI smooth muscle causing dilation and decreased motility. Cardiac inotrope.
CLINICAL EFFECT: May reverse hypoglycemia (if patient has glycogen stored in liver) within 4-8 minutes (could be as long as 15 or more). |
|

PEDIATRIC DOSAGE
phenylephrine nasal spray 0.5% |
None.
|
|
CLASS
diltiazem |
calcium channel blocker, calcium antagonist
|
|

BONUS
phenylephrine nasal spray 0.5% |
SPECIAL NOTES:
Single patient use only. Air or strong light causes potency loss; do not use solution if brown in color or precipitate is in the bottle. |
|
|
INDICATIONS
glucagon |
Symptomatic hypoglycemia when IV access is delayed.
|
|

BRAND NAME
furosemide |
Lasix
|
|
ADULT DOSAGE
vasopressin |
V/Fib-Pulseless V-Tach: 40 Units IV/IO/ET, one time only prior to administration of 1st or 2nd dose of epi.
|
|

CLASS
furosemide |
Loop diuretic
|
|
|
CONTRAINDICATIONS
glucagon |
Known hypersensitivity
Pheochromocytoma ( a rare tumor of the adrenal gland that cause too much release of epi and norepi). Insulinoma Should not be routinely used to replace dextrose when IV access has been obtained. |
|

MECHANISM OF ACTION
furosemide |
PHARMACOLOGIC: Inhibits electrolyte reabsorption in the ascending Loop of Henle. Promotes excretion of sodium, potassium and chloride. Vasodilation increases venous capacitance and decreases afterload.
CLINICAL: Diuresis. |
|
MECHANISM OF ACTION
diltiazem |
PHARMACOLOGICAL: Inhibits calcium ion influx across cell membranes during cardiac depolarization, decreases SA & AV conduction and dilates coronary and peripheral arteries and arterioles.
CLINICAL: Slows the rapid ventricular rate associated with AFib and AFlutter, and reduces coronary and peripheral vascular resistance. |
|

INDICATIONS
furosemide |
Pulmonary edema
CHF |
|
|
ADVERSE REACTIONS
glucagon |
N/V
Generalized allergic reactions including urticaria, respiratory distress, and hypotension (made from beef/pork pancreas) Palpitations, HTN, tachycardia |
|

CONTRAINDICATIONS
furosemide |
Anuria (relative)
Hypovolemia Hypotension |
|
INDICATIONS
sodium bicarbonate 8.4% |
Pre-existing metabolic acidosis
Overdose of ASA, cyclic antidepressants (alkalinization of blood) Cardiac arrest after other interventions and ventilation is adequate |
|

ADVERSE REACTIONS
furosemide |
May exacerbate hypovolemia
Hyperglycemia (due to hemoconcentration) Hypokalemia May decrease the response to pressors |
|
|
ADULT DOSAGE
glucagon |
(children and adults greater than 20 kg or 44 lbs)
HYPOGLYCEMIA: 1 mg IM, may repeat in 7-10 min |
|

ADULT DOSAGE
furosemide |
ON ORAL FUROSEMIDE THERAPY: Consider initial dose of 2 times daily oral dose; if no effect in 20 min may double inital dose.
NOT ON ORAL FUROSEMIDE THERAPY: 0.5-1 mg/kg to a maximum of 2 mg/kg (usually 20-40 mg) IV/IO slowly. |
|
INDICATIONS
diltiazem |
Rapid ventricular rates associated with AFib and AFlutter, and for PSVT refractory to adenosine.
|
|

PEDIATRIC DOSAGE
furosemide |
1 mg/kg IV/IO slowly
|
|
|
PEDIATRIC DOSAGE
glucagon |
(for children under 20 kg or 44 lbs)
HYPOGLYCEMIA: 0.5 mg IM or a dose equivalent to 20-30 mcg/kg, may repeat in 7-10 minutes. |
|

BONUS
furosemide |
Ototoxicity and resulting deafness can occur.
Since furosemide is a sulfonamide derivative, it may induce allergic reactions in pts with sensitivity to sulfonamides (sulfa drugs). |
|
PEDIATRIC DOSAGE
vasopressin |
Not used in the pediatric population.
|
|
|
BRAND NAME
ipratropium bromide |
Atrovent
|
|
|
BONUS
glucagon |
Blood sugar should be measured rapidly before deciding upon the administration of D50 or glucagon, especially in the non-diabetic patient.
Documented hypoglycemia is a true medical emergency, IM glucagon, should be administered rapidly if IV access is delayed. In known alcoholics, administer thiamine in addition to glucagon to prevent inducing an encephalopathy in a thiamine-deficient patient. Patients with Type I diabetes do not have as great a response in blood glucose levels as Type II stable patients. For all patients having hypoglycemic episode, supplementary complex carbohydrates should be eaten within 2 hours, especially in a child or adolescent. |
|
|
CLASS
ipratropium bromide |
anticholinergic, bronchodilator
|
|
CONTRAINDICATIONS
diltiazem |
Hypotension (less than 90 mmHg systolic)
Acute MI Cardiogenic shock VT or wide-complex VT of unknown origin Second or third-degree AV block Wolff-Parkinson-White (WPW) syndrome Sick sinus syndrome Beta blocker use |
|
|
MECHANISM OF ACTION
ipratropium bromide |
Anticholinergic (parasympatholytic) agent appears to inhibit vagally-mediated reflexes by antagonizing the action of acetylcholine, the transmitter released from the vagal nerve.
|
|
|
BRAND NAME
ondansetron |
Zofran
|
|
|
INDICATIONS
ipratropium bromide |
Treatment of bronchospasm associated with COPD (emphysema and chronic bronchitis).
To be used either alone or in combination with Albuterol. |
|
CONTRAINDICATIONS
sodium bicarbonate 8.4% |
Alkalosis
|
|
|
CONTRAINDICATIONS
ipratropium bromide |
Ipratropium bromide is contraindicated in known or suspected cases of hypersensitivity to ipratropium bromide or to atropine and its derivates.
PRECAUTION: Should be used with caution in pts with narrow angle glaucoma. |
|
|
CLASS
ondansetron |
antiemetic agent
|
|
|
ADVERSE REACTIONS
ipratropium bromide |
RESP: Coughing, sputum increased
CNS: Dizziness, insomnia, tremors, nervousness GI: Nausea |
|
ADVERSE REACTIONS
diltiazem |
CV: hypotension, bradycardia, heart block, CP, asystole.
GI: N/V CNS: HA, fatigue, drowsiness |
|
|
ADULT DOSAGE
ipratropium bromide |
Give 500 mcg in 2.5 mL NS (1 unit dose vial) via SVN or in-line with a ventilatory device. Repeat according to medical control preference.
May mix one unit dose vial of ipratropium bromide with one unit dose vial of albuterol. |
|
|
MECHANISM OF ACTION
ondansetron |
Selectively blocks serotonin 5-HT3 receptors located in the CNS at the chemoreceptor trigger zone and in the peripheral nervous system on nerve-terminals of the vagus nerve
|
|
|
PEDIATRIC DOSAGE
ipratropium bromide |
Not used pre-hospital for peds.
|
|
MECHANISM OF ACTION
magnesium sulfate |
PHARMACOLOGY: Second most plentiful intracellular cation; important role in neurochemical transmission and muscular excitability (may decrease acetylcholine released by nerve impulses); decreases myocardial irritability and neuromuscular irritability.
CLINICAL: Cardiac - reduces ventricular irritability, especially when associated with hypomagnesemia; inhibition of muscular excitability. |
|
|
BONUS
ipratropium bromide |
Anticholinergics produce preferential dilation of the larger central airways, in contrast to beta agonists, which affect the peripheral airways.
May be more effective used in combination with beta agonists. Should be kept out of light in a foil pouch and avoid excessive humidity. |
|
|
INDICATIONS
ondansetron |
N/V
|
|
ADULT DOSAGE
diltiazem |
INITIAL: 0.25 mg/kg IV/IO (usually 20 mg) administered over 2 minutes
REPEAT IN 15 MIN IF RESPONSE IS INADEQUATE: 0.35 mg/kg IV/IO administered over 2 min MAINTENANCE INFUSION: 5.0-15.0 mg/hr |
|
|
CONTRAINDICATIONS
ondansetron |
Hypersensitivity
Use with caution in patients with hepatic impairment |
|
ADVERSE REACTIONS
sodium bicarbonate 8.4% |
CV: CHF, edema secondary to sodium overload.
METABOLIC: Hyperosmolarity, metabolic acidosis, hypernatremia, in cardiac arrest may cause extracellular alkalosis and intracellular acidosis. |
|
|
ADVERSE REACTIONS
ondansetron |
CNS: HA, malaise, fatigue, dizziness, fever, sedation, extrapyramidal syndrome
CV: CP, arrhythmias RESP: Hypoxia GI & HEPATIC: Diarrhea, constipation, abdominal pain, xerostomia, decreased appetite SKIN: Rash |
|
PEDIATRIC DOSAGE
diltiazem |
The safety and efficacy of this drug for use in children has not been established.
|
|
|
ADULT DOSAGE
ondansetron |
4-8 mg IV slow push over 2-5 min
8mg PO ODT or tablet |
|
ADULT DOSAGE
sodium bicarbonate 8.4% |
PRE-EXISTING METABOLIC ACIDOSIS OR ALKALINIZATION OF BLOOD: 50-100 mEq IV/IO per medical control authority.
INFUSION: 50 mEq of sodium bicarbonate in 250 mL of NS or as determined by medical control. CARDIAC ARREST: First dose usually 1 mEq/kg with subsequent doses of 0.5 mEq/kg every 10 minutes after other standard treatment (defib, CPR, intubation, ventilation and more than one trial of epi) has been used. |
|
|
PEDIATRIC DOSAGE
ondansetron |
(1 month to 12 years old)
Greater than 40 kg - 4 mg IV slow push over 2-5 min Less than 40 kg - 0.1 mg/kg IV slow push over 2-5 min 4-12 years old 4 mg PO ODT or Tablet |
|
BONUS
diltiazem |
Avoid use in pts with poison or drug induced tachycardia.
CaCl can be used to prevent hypotensive effects of this drug and treat patients with a calcium channel blocker OD. Beta blocker use. |
|
|
BONUS
ondansetron |
Do not attempt to push Zofran ODT tablets through the foil backing. With dry hands, PEEL BACK the foil backing of 1 blister and GENTLY remove the tablet. IMMEDIATELY place the Zofran ODT tablet on top of the tongue where it will dissolve in seconds, then swallow with saliva. Administration with liquid is not necessary.
Bottles: Store between 2 degrees C and 30 degrees C (36F-86F). Protect from light. Dispense in tight, light-resistant container as defined in the USP. Unit dose packs: store between 2C-30C (36F-86F) Protect from light. Store blisters in cartons. |
|
INDICATIONS
magnesium sulfate |
Torsade de pointes, drug of choice
VF/Pulseless VT refractory to lidocaine and/or amiodarone Hypomagnesemia Pre-term labor (PTL) Pregnancy-induced hypertension (PIH, toxemia of pregnancy, pre-eclampsia and/or eclampsia) Hyperreactive airway - severe asthma |
|
|
BRAND NAME
naloxone HCl |
Narcan
|
|
|
BRAND NAME
amiodarone |
Amiodarone
|
|
|
CLASS
naloxone HCl |
narcotic (opioid) antagonist
|
|
PEDIATRIC DOSAGE
sodium bicarbonate 8.4% |
1 mEq/kg IV/IO slowly, if ventilation is adequate according to medical control. Can contribute to acidosis and cause fluid overload.
|
|
|
MECHANISM OF ACTION
naloxone HCl |
Competitive inhibition at narcotic receptor sites.
Reverses respiratory depression secondary to narcotics. |
|
|
CLASS
amiodarone |
anti-arrhythmic agent
|
|
|
INDICATIONS
naloxone HCl |
ANTIDOTE FOR: Opioid overdoses.
May differentiate opioid-induced coma from other causes. |
|
BRAND NAME
acetylsalicylic acid, aspirin, ASA |
Bufferin, Anacin, APC, among others
|
|
|
CONTRAINDICATIONS
naloxone HCl |
Hypersensitivity
|
|
|
MECHANISM OF ACTION
amiodarone |
Multiple effects on potassium, sodium and calcium channels.
Prolongs action potential, refractory period. Ventricular automaticity (potassium channel blockade). Slows membrane depolarization and impulse conduction (sodium channel blockade). Negative chronotropic activity in nodal tissue, rate reduction, and antisympathetic activity (calcium channel and B-blockade). Dilates coronary arteries due to calcium channel and alpha-adrenergic blocking action. |
|
|
ADVERSE REACTIONS
naloxone HCl |
Withdrawal symptoms, especially in neonates (N/V, diaphoresis, increased HR, falling BP, tremors).
BE PREPARED FOR COMBATIVE PT AFTER ADMINISTRATION. |
|
NEONATAL DOSAGE
sodium bicarbonate 8.4% |
1 mEq/kg IV/IO of 4.2% slowly. Waste 25 mL of 8.4% solution and add 25 mL of NS, each mL contains 0.5 mEq of sodium bicarbonate.
|
|
|
ADULT DOSAGE
naloxone HCl |
IV, IM, SC, Inject SL, ET: 2.0 mg initial bolus IV or ET, may repeat every 2 minutes as necessary; titrate to effect.
INTRA-NASAL: 1.0 mg each nostril using a Mucosal Atomizer Device for a total of 2 mg. May repeat every 2 minutes as necessary. Titrate to effect. |
|
|
INDICATIONS
amiodarone |
Treatment of: DEFIB! - refractory VF/pulseless VT, polymorphic VT, and wide complex tachycardia of uncertain origin.
Control hemodynamically stable VT when cardioversion is unsuccessful. Adjunct to cardioversion of SVT and PSVT. Rate control in AFib or AFlutter. |
|
|
PEDIATRIC DOSAGE
naloxone HCl |
< 5 years or < 20 kg: 0.1 mg/kg IV, ET, inject SL, SC, IO (includes neonate)
> 5 years or > 20 kg: 2 mg IV, ET, inject SL, SC, or IO |
|
CONTRAINDICATIONS
magnesium sulfate |
Hypermagnesemia
Use cautiously in patients with impaired renal function and pre-existing heart blocks (relative). |
|
|
BONUS
naloxone HCl |
Opioid drugs include heroin, Dilaudid, morphine, meperidine, codeine, methadone, Lomotil, Darvon, Darvocet, Talwin and others.
|
|
|
CONTRAINDICATIONS
amiodarone |
Bradycardia
Second or third degree heart block unless a functioning pacemaker is present. Cardiogenic shock Hypotension Pulmonary congestion |
|
|
BRAND NAME
thiamine HCl (vitamin B1) |
Betalin
|
|
BONUS
sodium bicarbonate 8.4% |
Flush tubing before and after administration, ESPECIALLY with concurrent use of calcium chloride.
Sodium bicarbonate administration should be considered only for treatment of documented severe acidosis associated with prolonged cardiac arrest or unstable hemodynamic state, hyperkalemia, or certain overdoses (ie cyclic antidepressants, ASA, phenobarbital, etc.) SEVERE tissue necrosis will occur with extravasation. |
|
|
CLASS
thiamine HCl (vitamin B1) |
vitamin
|
|
|
ADVERSE REACTIONS
amiodarone |
CV: bradycardia, hypotension, asystole/cardiac arrest, AV block
TORSADES DE POINTES: (prolongs QT interval), CHF GI & HEPATIC: N/V, abnormal LFTs SKIN: slate-blue pigmentation OTHER: fever, HA, dizziness, flushing, abnormal salivation, photophobia |
|
|
MECHANISM OF ACTION
thiamine HCl (vitamin B1) |
Required for carbohydrate metabolism.
Deficiency leads to anemia, polyneuritis, Wernicke's encephalopathy, cardiomyopathy. Administration may reverse symptoms of deficiency, but effects are dependent upon duration of illness and severity of disease. |
|
ADVERSE REACTIONS
magnesium sulfate |
CARDIOVASCULAR: Hypotension, flushing, circulatory collapse, depressed cardiac function, heart block, asystole, smooth muscle relaxant (antihypertensive effects).
RESPIRATORY: respiratory depression and/or paralysis. This adverse reaction may occur in both mother and/or infant during or up to 24 hours after the administration. CNS: sweating, drowsiness, hypothermia, depressed reflexes progressing to flaccidity and paralysis. May occur in both mother and/or infant up to 24 hours. GI: nausea GU: mild diuretic METABOLIC: hypocalcemia, hypermagnesemia |
|
|
INDICATIONS
thiamine HCl |
Alcoholism, delirium tremens.
Coma of unknown origin, especially if alcohol or malnourishment may be involved. Suspect Wernicke or Korsakoff Syndrome Other thiamine deficiency syndromes Severe CHF |
|
|
ADULT DOSAGE
amiodarone |
VF/PULSELESS VT: 300 mg IV/IO push over 30-60 seconds, may repeat in 3-5 min with 150 mg IV/IO push
WIDE COMPLEX TACHYCARDIAS, AFLUTTER, AFIB, SVT WITH CARDIOVERSION: 150 mg IV/IO over 10 min (mix in 50 mL bag of D5W) may repeat q 10 min. MAINTENANCE INFUSION POST RESUSCITATION/CONVERSION: After successful defib, follow with 1 mg/min IV/IO infusion for 6 hours, then up to 0.5 mg/min IV/IO infusion for up to 18 hours, max daily dose is 2.2 grams. |
|
|
CONTRAINDICATIONS
thiamine HCl |
Do not give intra-arterial
|
|
ADULT DOSAGE
magnesium sulfate |
CARDIAC:
VF/Pulseless VT: 1-2 gm IV diluted in 50-100 mL NS or D5W, administered over 1-2 minutes. Torsade de pointes: 1-2 gm IV diluted in 50-100 mL NS or D5W administered over 1-2 minutes, followed by the same amount infused over 1 hour. Hypomagnesemia: 1-2 gm IV diluted in 50-100 mL NS or D5W administered IV push over 5-60 minutes. RESPIRATORY: Respiratory/severe asthma: Initial infusion (field) 2 gm magnesium sulfate mixed in 50 mL NS or D5W to be infused IV using microdrip tubing over 5-10 minutes. Stop infusion if hypotension, respiratory depression or bradycardia develops. PRE-TERM LABOR (PTL): Initial bolus 4-6 gm over 15-20 minutes (suggestion is to add 4 gm to 100 mL D5W, LR or NS. Resultant concentration is 40 mg/mL.) PREGNANCY INDUCED HYPERTENSION, PRE-ECLAMPSIA/ECLAMPSIA: Initial bolus: 3-6 gm over 10-15 minutes (suggestion is to add 4 gm to 100 mL D5W, LR, NS. Resultant concentration is 40 mg/mL). |
|
|
ADVERSE REACTIONS
thiamine HCl |
Hypotension (rare)
|
|
|
PEDIATRIC DOSAGE
amiodarone |
VF/PULSELESS VT: 5 mg/kg IV/IO push (max 300 mg single dose), may repeat x 2 q 5 min to a total max of 15 mg/kg/day.
PROBABLY VT WITH PULSE: 5 mg/kg IV/IO administered over 20 min, may repeat two more times to a total of 15 mg/kg/day. |
|
|
ADULT DOSAGE
thiamine HCl (vitamin B1) |
100 mg SIVP or Deep IM
|
|
BONUS
magnesium sulfate |
O2 should be administered to patients receiving magnesium sulfate.
Respiratory (asthma) emergencies: magnesium sulfate follows Albuterol and Atrovent SVN and administration of 0.3 IM Epi (1:1000) For IV/IO infusions (respiratory) start and stop times should be closely monitored and documented per administration guidelines of 20 minutes or greater. Transport gravid patients lying or tilted to left side to prevent restricting venous return to heart. Use cautiously in patients with impared renal function, pre-existing heart blocks and women in labor. Evaluate cardiac status and ECG assessing for prolonged PR and widened QRS intervals. |
|
|
PEDIATRIC DOSAGE
thiamine HCl (vitamin B1) |
Rarely used.
|
|
|
BONUS
amiodarone |
Half-life may exceed 40 days.
Patient must be on a cardiac monitor - monitor the HR and rhythm. |
|
|
BONUS
thiamine HCl (vitamin B1) |
In the known alcoholic patient, dextrose should not be administered without thiamine.
Thiamine has been shown to be useful in severe CHF. |
|
CLASS
acetylsalicylic acid, aspirin, ASA |
analgesic, antipyretic, anti-inflammatory
|
|
|
BRAND NAME
oxytocin |
Pitocin, Syntocin
|
|
BRAND NAME
calcium chloride |
Calcium Chloride
|
|
|
CLASS
oxytocin |
pituitary hormone, polypeptide, uterine stimulant
|
|
MECHANISM OF ACTION
acetylsalicylic acid, aspirin, ASA |
In small doses, aspirin blocks thromboxane A2, a potent platelet aggregant and vasoconstrictor.
This property has lead to its use in the acute phase of management of an MI. Decreased platelet aggregation. |
|
|
MECHANISM OF ACTION
oxytocin |
Binds to oxytocin receptor sites on the surface of uterine smooth muscles: increases force and frequency of uterine contractions.
|
|
CLASS
calcium chloride |
electrolyte
|
|
|
INDICATIONS
oxytocin |
Postpartum hemorrhage due to uterine atony
|
|
INDICATIONS
acetylsalicylic acid, aspirin, ASA |
Chest pain or other signs/symptoms suggestive of acute myocardial infarction
ECG changes suggestive of acute myocardial infarction Unstable angina Pain, discomfort, fever in adult patient only |
|
|
CONTRAINDICATIONS
oxytocin |
Hypersensitivity
|
|
MECHANISM OF ACTION
calcium chloride |
Increases extracellular and intracellular calcium levels.
Stimulates release of catecholamines. Increases cardiac contractile state (+ inotropic effect). May enhance ventricular automaticity. Inhibits the effects of adenosine on mast cells. |
|
|
ADVERSE REACTIONS
oxytocin |
CV: Shock, tachycardia, dysrhythmias
RESP: Anaphylaxis GI: N/V GU: If used prior to deliver, can cause uterine rupture, uterine spasm, lacerations, and fetal damage. OTHER: Clotting disorders, electrolyte disturbances. |
|
CONTRAINDICATIONS
acetylsalicylic acid, aspirin, ASA |
Bleeding ulcer, hemorrhagic states, hemophilia.
Known hypersensitivity to salicylates or other NSAID's that has lead to hypotension and/or bronchospasm. Children and adolescents (pre-hospital personnel should not administer ASA to this age group). |
|
|
ADULT DOSAGE
oxytocin |
POSTPARTUM HEMORRHAGE: 10-20 USP units added to 1000 mL NS or LR and run at a rate necessary to control uterine atony or 10 USP units may be given IM after delivery of the placenta.
|
|
INDICATIONS
calcium chloride |
Acute hypocalcemia
Calcium channel blocker OD Acute hyperkalemia (known or suspected) Hypermagnesemia (Magnesium OD) Pre-treatment for IV verapamil administration |
|
|
PEDIATRIC DOSAGE
oxytocin |
Not applicable.
|
|
ADVERSE REACTIONS
acetylsalicylic acid, aspirin, ASA |
Use with caution in the patient with history of asthma.
Anaphylactic reactions in sensitive patients have occured; skin eruptions. Other side effects rare with single dose. |
|
|
BONUS
oxytocin |
Post partum hemorrhage is defined as blood loss in excess of 500 mL at delivery and during the first 24 hours after delivery.
|
|
CONTRAINDICATIONS
calcium chloride |
Hypercalcemia
Concurrent digoxin therapy (relative) |
|
ADULT DOSAGE
acetylsalicylic acid, aspirin, ASA |
CARDIAC: 160-325 mg PO (2-4 pediatric chewable tabs), chew or swallow
PAIN/DISCOMFORT/FEVER: 325 mg PO (4 pediatric chewable tabs), chew or swallow |
|
ADVERSE REACTIONS
calcium chloride |
Brady-asystolic arrest
Severe tissue necrosis if solution extravasates. Use cautiously in patients on digitalis; may cause serious arrhythmias. |
|
PEDIATRIC DOSAGE
acetylsalicylic acid, aspirin, ASA |
Not recommended for pre-hospital use in children.
|
|
ADULT DOSAGE
calcium chloride |
HYPOCALCEMIA, CALCIUM CHANNEL BLOCKER OD, HYPERKALEMIA, HYPERMAGNESEMIA: 5-10 mL (0.5-1 gm) of CaCl 10% IV/IO. May repeat in 10 min.
PRE-TREATMENT FOR IV VERAPAMIL ADMINISTRATION: 3 mL of 10% CaCl. May be repeated once. |
|
BONUS
acetylsalicylic acid, aspirin, ASA |
Baby ASA is heat and light sensitive. The odor of acetic acid (vinegar-like smell) indicates degradation of product.
|
|
PEDIATRIC DOSAGE
calcium chloride |
HYPOCALCEMIA, CALCIUM CHANNEL BLOCKER OD, HYPERKALEMIA AND HYPERMAGNESEMIA: 0.2-0.25 mL/kg of a 10% solution IV/IO infused slowly.
|
|
BRAND NAME
epinephrine HCl |
Adrenalin
|
|

BRAND NAME
atropine sulfate |
Atropine Sulfate
|
|
BRAND NAME
dopamine |
Intropin
|
|

CLASS
atropine sulfate |
anticholinergic agent, antidote, antispasmodic, antiarrhythmic, antimuscarinic
|
|
CLASS
epinephrine HCl |
sympathomimetic
|
|

MECHANISM OF ACTION
atropine sulfate |
PHARMACOLOGICAL: Blocks the action of acetylcholine as a competitive antagonist at muscarinic receptor sites in smooth muscle, secretory glands, and the drying of secretions. Atropine reverses the muscarinic effects of cholinergic poisoning by primarily reversing bronchorrhea and bronchoconstriction. At high enough doses, atropine may have an effect on nicotinic receptors responsible for restlessness, hallucinations, disorientation, and/or delirium.
CV: ↑ HR (+ chronotropic effect); ↑ conduction velocity (+ dromotropic effect); ↑ force of contraction (slight)(+ inotropic effect), ↑ CO. RESP: Decreased mucus production; increased bronchial smooth muscle relaxation (bronchodilation). GI: Decreased GI secretion and motility. GU: Decreased urinary bladder tone. MISC: Mydriasis (pupillary dilation); decreased sweat production. |
|
MECHANISM OF ACTION
epinephrine HCl |
PHARMACOLOGICAL EFFECTS: Direct acting α and β agonist; α-bronchial, cutaneous, renal, and visceral arterial constriction (increased systemic vascular resistance).
β1 - positive inotropic and chronotropic actions (increases myocardial workload and oxygen requirements), increases automaticity and irritability. β2 - bronchial smooth muscle relaxation and dilation of skeletal vasculature. Other: blocks histamine release. CLINICAL EFFECTS: Cardiac arrest - increases cerebral and myocardial perfusion pressure, systolic and diastolic BPs, and electrical activity in the myocardium; can stimulate spontaneous contractions in asystole. Bradycardia - increases heart rate & BP Bronchospasm/anaphylaxis - reverse signs/symptoms. |
|

INDICATIONS
atropine sulfate |
Symptomatic bradycardia (sinus, junctional, AV blocks causing hypotension, ventricular ectopy, CP, ALOC, etc.), monitored pt only.
Asystole (after epi) monitored pt only. PEA with actual or relative bradycardia (after epi) monitored pt only. Acetylcholinesterase inhibitor poisoning (organophosphate, carbamate cholinergic poisoning). |
|
CLASS
dopamine |
sympathomimetic
|
|

CONTRAINDICATIONS
atropine sulfate |
Hypersensitivity to atropine or any component of the formulation - Belladonna alkaloid allergy.
Glaucoma, acute narrow angle (relative contraindication for pt with symptomatic bradycardia) adhesions between the iris and lens. Myasthenia gravis (unless used to treat side effects of acetylcholinesterase inhibitor). Tachycardia, asthma, thryotoxicosis, Mobitz type II block, 3rd degree heart block, hepatic disease, renal disease, obstructive uropathy. Obstructive GI disease, paralytic ileus, intestinal atony of the elderly or debilitated pt, severe ulcerative colitis, or ulcerative colitis. PREGNANCY RISK FACTORS/CONSIDERATIONS: Atropine has been found to cross the human placenta. Trace amounts of atropine can enter breast milk. |
|
INDICATIONS
epinephrine HCl |
Cardiac arrest - VF/Pulseless VT; asystole; PEA (First line pharmacologic agent for any pulseless dysrhythmia in cardiopulmonary arrest)
Severe bronchospasm, ie bronchiolitis, asthma, croup or anaphylaxis Bradycardia, refractory with profound hypotension, monitored patient only Hypotension unresponsive to other therapy, monitored patient only |
|

ADVERSE REACTIONS
atropine sulfate |
MAJOR: Tachydysrhythmias, flushing, ventricular irritability, exacerbation/initiation of angina, acute narrow glaucoma, blurred vision, mydriasis, agitation to delirium, bloating, constipation, decreased gastric emptying.
MINOR: Dry mouth/mucous membranes, loss of taste, N/V, urinary retention, neuromuscular weakness, decreased sweating, ↑ body temp. |
|
CONTRAINDICATIONS
epinephrine HCl |
No known contraindication for cardiac arrest. Hypothermia is a relative contraindication.
|
|

ADULT DOSAGE
atropine sulfate |
SYMPTOMATIC BRADYCARDIA: IV/IO - 0.5 mg IV push q 5 min. Do NOT exceed a total dose of 3 mg or 0.04 mg/kg if symptoms profound.
ASYSTOLE: IV/IO - 1 mg. Repeat q 3-5 min (generally up to 3 doses) if asystole persists. Total dose should not exceed 0.04 mg/kg. ORGANOPHOSPAHTE OR CARBAMATE POISONING: IV/IO - Initially: 1-5 mg. Doses should be doubled q 5 min until signs of muscarinic excess abate (clearing of bronchial secretions, bronchospasm, and adequate oxygenation). |
|
MECHANISM OF ACTION
dopamine |
EFFECTS ARE DOSE-DEPENDENT
1-2 mcg/kg/min: Acts on dopaminergic receptors to stimulate cerebral, renal and mesenteric vasculature to dilate; HR and BP are usually unchanged; may increase urine output. 2-10 mcg/kg/min: β1 stimulant action is primary effect (increases cardiac output and partially antagonizes the α-adrenergic-mediated vasoconstriction. Overall effect is increased cardiac output and only modest increase in systemic vascular resistance (SVR). 10-20 mcg/kg/min: α-adrenergic effects predominate resulting in renal, mesenteric and peripheral arterial and venous vasoconstriction with marked increase in SVR, pulmonary vascular resistance and further increased preload. >20 mcg/kg/min: Produces hemodynamic effects similar to norepinephrine; may increase HR and O2 demand to undesirable limits. |
|

PEDIATRIC DOSAGE
atropine sulfate |
SYMPTOMATIC BRADYCARDIA: IV/IO - 0.02 mg/kg (min of 0.1 mg), repeat q 5 min to a max total dose of 1 mg in children and 2 mg in adolescents.
MAX SINGLE DOSE: Child: 0.5 mg, Adolescent: 1 mg. ORGANOPHOSPHATE/CARBAMATE CHOLINERGIC POISONING: IV/IO - 0.03-0.05 mg/kg q 10-20 min until cholinergic symptoms minimize, then every 1-4 hours for at least 24 hours. |
|
ADVERSE REACTIONS
epinephrine HCl |
CARDIOVASCULAR: Hypertension, ventricular dysrhythmias; tachycardia; angina
CNS: Anxiety, agitation GI: Nausea/vomiting |
|

BONUS
atropine sulfate |
Administering too small of a dose or administering too slowly may result in PARADOXICAL BRADYCARDIA.
|
|
ADULT DOSAGE
epinephrine HCl |
PULSELESS ARREST: 1 mg of 1:10,000 solution IV/IO; repeat every 3-5 min OR
ET: Give 2-2.5 mg via the ET. May use 1:10,000 or dilute 1:1000 to equal 10 mL via ET tube for adult. 2 mg 1:1,000 epi with 8 mL NS in a 10 mL syringe. INFUSION FOR HYPOTENSION OR SYMPTOMATIC BRADYCARDIA: 1 mg added to 500 mL of NS administered at 1 mcg/min titrated to desired hemodynamic response (range 2-10 mcg/min); not first-line therapy. ANAPHYLAXIS & ASTHMA: Give 0.3-0.5 mg of 1:1,000 solution IM (preferred), SC, or inject SL, may repeat every 15-20 min; in extreme cases only, may be asked to use 1:10,000 solution and give 0.1 mg every 5 min IV/IO or continuous IV/IO infusion of 1-4 mcg/min to prevent multiple injections. |
|

BRAND NAME
lidocaine HCl |
Xylocaine
|
|
INDICATIONS
dopamine |
Symptomatic bradycardias
Hemodynamically significant hypotension in the absence of hypovolemia (cardiogenic or septic shock ONLY after fluid administration; assess breath sounds first) |
|

CLASS
lidocaine HCl |
antiarrhythmic, local anesthetic
|
|
PEDIATRIC DOSAGE
epinephrine HCl |
PULSELESS ARREST OR REFRACTORY BRADYCARDIA: 0.01 mg/kg of 1:10,000 IV/IO repeat every 3-5 min, maximum single dose 1 mg
ET: 0.1 mg/kg of 1:1,000; mix with NS to a total of 3-5 mL in syringe; repeat every 3-5 min, maximum single dose 1 mg ASTHMA & ANAPHYLAXIS: Use 1:1,000 solution, give 0.01 mg/kg IM (preferred), SC (maximum single dose of 0.5 mg/dose) IV INFUSION: 0.1-1 mcg/kg/min; prepare for children 0.6 x body weight in kg = mg added to NS to make 100 mL. Delivery of 1 mL/hour delivers 0.1 mcg/kg/min CROUP: 3 mg 1:1,000 mixed in 3 mL NS via SVN |
|

MECHANISM OF ACTION
lidocaine HCl |
Decreases automaticity by slowing the rate of spontaneous phase 4 depolarization.
Terminates re-entry by decreasing conduction in re-entrant pathways (by slowing conduction in ischemic tissue, equalizes conduction speed among fibers). ↑ ventricular fibrillation threshold. |
|
NEONATAL DOSE FOR FIRST 12 HOURS OF LIFE
epinephrine HCl |
INITAL AND REPEAT DOSE FOR CARDIAC ARREST / REFRACTORY BRADYCARDIA: 0.01-0.03 mg/kg IV/IO of 1:10,000 every 3-5 min
ET: 0.1 mg/kg of 1:10,000 every 3-5 min if neonate has no vascular access |
|

INDICATIONS
lidocaine HCl |
Suppression of ventricular arrhythmias (VT, VF, PVC's).
Prophylaxis against recurrence after conversion from VT or VF. Frequent PVC's (>6 min, 2 or more in a row, multiform PVC's, or R-on-T phenomenon). Pre-intubation for head trauma or suspected intracranial hemorrhage. |
|
CONTRAINDICATIONS
dopamine |
Hypovolemic shock (relative)
Pheochromocytoma MAO inhibitors, such as Marplan, Nardil, or Parnate |
|

CONTRAINDICATIONS
lidocaine HCl |
Known hypersensitivity/allergy.
Use extreme caution in pts with conduction disturbance (2nd or 3rd degree heart block). DO NOT TREAT ECTOPIC BEATS IF HR IS < 60. THEY ARE PROBABLY COMPENSATING FOR THE BRADYCARDIA; INSTEAD, TREAT THE BRADYCARDIA! |
|
ADVERSE REACTIONS
dopamine |
CV: Cardiac arrhythmias may occur due to increased myocardial oxygen demand (usually tachydysrhythmias), hypertension, hypotension at low doses
GI: nausea and vomiting GU: renal shutdown (at higher doses) OTHER: extravasation may cause tissue necrosis |
|

ADVERSE REACTIONS
lidocaine HCl |
CV: May also cause SA nodal depression or conduction problems and hypotension in large doses, or if given too rapidly.
Excessive doses in pediatric pts may produce myocardial and circulatory depression. CNS: In large doses drowsiness, disorientation, paresthesia, decreased hearing acuity, muscle twitching, agitation, focal or generalized seizures. |
|
ADULT DOSAGE
dopamine |
(dosage range 2-10 mcg/kg/min)
PREPARATION: (If premixed not carried) Add 400 mg to 250 mL NS or dextrose = 1600 mcg/mL BRADYCARDIA: Start at 5 mcg/kg/min SHOCK - CARDIOGENIC OR SEPTIC (non-hypovolemic) BP <70 systolic: Start drip at 5 mcg/kg/min BP >70 systolic: Start drip at 2.5 mcg/kg/min |
|

ADULT DOSAGE
lidocaine HCl |
PULSELESS VF/VT: Initial bolus of 1-1.5 mg/kg IV/IO push q 3-5 min to a total dose of 3 mg/kg.
An initial bolus of 1.5 mg/kg should be given for cardiac arrest situations. Following the return of a spontaneous rhythm, initiate a drip at 2-4 mg/min for maintenance infusion. ANTIDYSRHYTHMIC OR RHYTHMS WITH A PULSE: Initial boluses can be given as 1-1.5 mg/kg IV/IO push and additional boluses can be given as 0.5-0.75 mg/kg q 5-10 min to a total dose of 3 mg/kg. Following the return of a spontaneous rhythm, initiate a drip at 2-4 mg/min. MAINTENANCE INFUSION: Started after return of spontaneous rhythm for either indication above. Add 1-2 gm to a 250 mL NS or 5% dextrose solution or use premixed solution (2 gm in 500 mL) and initiate a drip at 2-4 mg/min according to concentration. Patients > 70 years or with hepatic, renal disease or poor perfusion state - reduce maintenance infusion by half. Pre-intubation for head trauma or suspected intracranial hemorrhage (consider administration of 1 mg/kg IV bolus 1-2 min prior to intubation.) |
|
PEDIATRIC DOSAGE
dopamine |
2-20 mcg/kg/min for circulatory shock or shock unresponsive to fluid administration
To prepare infusion for small children: 6 x body weight in kg = mg added to NS to make 100 mL. With this mixture 1 mL/hour delivers 1 mcg/kg/min; titrate to effect |
|

PEDIATRIC DOSAGE
lidocaine HCl |
INITIAL BOLUS DOSES: 1 mg/kg, may repeat 1 time in 3-5 min for VF/Pulseless VT or in 15 min if used for refractory dysrhythmias with a pulse (VT with pulse, significant ventricular ectopy).
INFUSION WITH RETURN OF SPONTANEOUS RHYTHM, OPTIONAL: 20-50 mcg/kg/min; prepared by adding 120 mg (3cc) of 1 gm/25 mL (40 mg/mL) solution to 97 mL NS, yielding 1200 mcg/mL. 1 mL/kg/hr delivers 20 mcg/kg/min. 2.5 mL/kg/hr delivers 50 mcg/kg/min. |
|
BONUS
dopamine |
Always monitor drip rate, NEVER run wide open.
An infusion pump is required for interfacility transports; a minimum of microdrip tubing is required for field use. It is important to remember that even in low dose ranges dopamine elevates pulmonary artery occlusive pressure and may induce or exacerbate pulmonary congestion despite a rise in cardiac output. |
|

BONUS
lidocaine HCl |
Decrease maintenance infusion by 50% in cases of CHF, shock, or liver disease.
|
|
BRAND NAME
nitroglycerin |
Nitrostat, Tridil
|
|
BRAND NAME
verapamil HCl |
Isoptin, Calan, Verelan
|
|
CLASS
nitroglycerin |
vasodilator, organic nitrate, antianginal
|
|
CLASS
verapamil HCl |
calcium channel blocker
|
|
MECHANISM OF ACTION
nitroglycerin |
Smooth muscle relaxant acting on vascular, uterine, bronchial, and intestinal smooth muscle.
Reduces workload on the heart by causing blood pooling (decreased preload). Arteriolar vasodilation (decreased afterload). Coronary artery vasodilation. Increases blood flow to myocardium. Decreases myocardial O2 demand. |
|
MECHANISM OF ACTION
verapamil HCl |
Blocks calcium ion influx into cardiac and smooth muscle cells causing a depressant effect on the contractile mechanism resulting in negative inotropy.
Reduces contractile tone in vascular smooth muscle resulting in coronary and peripheral vasodilation. Slows conduction and prolongs refractory period in the AV node due to calcium channel blocking. Slows SA node discharge. In summary, decreases myocardial contractile force and slows AV conduction. |
|
INDICATIONS
nitroglycerin |
Angina
MI CHF with pulmonary edema |
|
INDICATIONS
verapamil HCl |
SVT
AFib & AFlutter with RVR |
|
CONTRAINDICATIONS
nitroglycerin |
Hypovolemia
Increased intracranial pressure |
|
CONTRAINDICATIONS
verapamil HCl |
AV block, sick sinus syndrome, any wide QRS complex tachycardia.
Shock Severe CHF |
|
ADVERSE REACTIONS
nitroglycerin |
CV: Hypotension, reflex tachycardia, bradycardia, decreased coronary perfusion at high doses (secondary to hypotension), headache secondary to dilation of meningeal vessels.
|
|
ADVERSE REACTIONS
verapamil HCl |
Extreme bradycardia
Asystole AV Block Hypotension CHF |
|
ADULT DOSAGE
nitroglycerin |
SL FOR CHEST PAIN: 1/150 gr (0.4 mg) tablet or one full spray, may repeat x 3.
SL FOR PULMONARY EDEMA: 1-2 of the 1/150 gr (0.4 mg) tablets may be given SL every 5-10 minutes as long as the systolic BP is greater than 90-100 systolic. |
|
ADULT DOSAGE
verapamil HCl |
2.5-5 mg SLOW IV/IO push over 2-3 min. May rebolus in 15-30 min with 5-10 mg IV/IO push until a max dosage of 20 mg.
|
|
PEDIATRIC DOSAGE
nitroglycerin |
Not used.
|
|
PEDIATRIC DOSAGE
verapamil HCl |
Not used.
|
|
BONUS
nitroglycerin |
INCOMPATIBILITIES/DRUG INTERACTIONS:
Other vasodilators Viagra, Cialis, and Levitra SPECIAL NOTES: NTG is heat and light sensitive; stock rotation assures fresh supply. SL: Cautiously administer NTG to a patient who has never received it, consider establishing an IV prior to administration. Closely monitor vital signs, cardiac rhythm. Bradydysrhythmias and hypotension usually respond to Trendelenburg position; atropine and vasopressors may be administered if needed. |
|
BONUS
verapamil HCl |
Not compatible with IV beta-blockers.
Vagal maneuvers may be tried first. Avoid verapamil in pts with wide-QRS tachycardia unless it is KNOWN WITH CERTAINTY to be supraventricular in origin. |

