![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
104 Cards in this Set
- Front
- Back
|
Iron absorption occurs in the?
|
duodenum
|
|
|
Iron consumed in two dietary forms
|
Heme form(meat) and non-heme form(vegetables)
heme form more readily absorbed |
|
|
Describe transport and storage of iron
|
1.) Absorption of iron occurs via enterocytes and their transporters.
2.) Transport into bloodstream occurs through ferroportin(iron MUST be bound while in blood to prevent free radical) 3.) Iron is then bound to transferrin in bloodstream where it is delivered to liver and bone marrow. 4.) Stored intracellular iron is bound to ferritin, which prevents free radical formation |
|
|
Causes of Microcytic Anemia
|
Iron deficiency Anemia
Anemia of Chronic Disease Sideroblastic Anemia Thalassemia Anemia of Chronic Disease - Iron locked away in macrophages |
|
|
Most common nutritional deficiency?
|
Iron deficiency
|
|
|
Most important regulatory step in iron uptake?
|
Ferroportin - transport of iron into blood. On enterocyte.
|
|
|
TIBC
|
measures amount of transferrin in blood
|
|
|
Serum Iron
|
Measure of iron in blood(this iron WILL be bound to transferrin)
|
|
|
% Saturation
|
percentage of transferrin bound to iron
|
|
|
Serum Ferritin
|
amount of iron bound in liver and macrophages
|
|
|
Lab findings in first stage of iron deficiency
|
Storage iron is depleted
This causes decreased ferritin because iron is being picked up from the liver/macrophage. Also causes increased TIBC because liver makes more transferrin to try and pick up more iron. |
|
|
Lab findings in second stage of iron deficiency
|
Serum iron is depleted
Serum iron goes down(duh). % Sat of transferrin also goes down. |
|
|
Lab findings in third stage of iron deficiency
|
Normocytic anemia
Bone marrow LIKES pretty red blood cell, but lacks the iron to make enough of them. Therefore it makes less, BUT they are normocytic! |
|
|
Lab findings in fourth stage of iron deficiency
|
Microcytic, hypochromic anemia
|
|
|
clinical features of iron deficiency
|
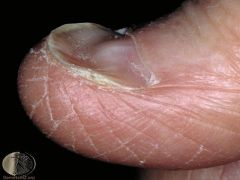
Anemia, Koilonychia, and Pica
|
|
|
Describe RDW and FEP in Iron deficiency anemia...
|
Increased RDW due to initial normocytic RBC's mixing with microcytic, so the distribution of width is larger than normal.
FEP, increased free erythrocyte protoporphyrin is due to the lack of heme to bind to it. |
|
|
Plummer-Vinson syndrome
|
Esophageal webs, atrophic glossitis(smooth tongue), and iron deficiency anemia
|
|
|
Patient presents with anemia, dysphagia, beefy red tongue.
|
Plummer-Vinson syndrome
esophageal webs atrophic glossitis(smooth tongue) iron deficiency anemia |
|
|
Anemia of Chronic Disease pathophys
|
chronic disease --> chronic inflammation --> increased acute phase reactants(Hepcidin)
Hepcidin sequesters iron in storage sites, preventing transfer of iron from macrophages to erythroid precursors. In addition, Chronic disease suppresses EPO production. |
|
|
Lab findings in anemia of chronic disease
|
increased ferritin, decreased TIBC
decreased serum iron, decreased %sat increased FEP Increased FEP - Heme = Fe+ + Protoporphyrin Since iron is reduced, but protoporphyrin is normal, there is elevated protoporphyrin. |
|
|
Sideroblastic Anemia pathophys
|
defective protoporphyrin synthesis.
Heme = Iron + Protoporphyrin If protoporphyrin is bad, then Heme is bad, which means RBC's are going to be smaller |
|
|
Rate limiting step in protoporphyrin synthesis?
|
Conversion of Succinyl-CoA to Aminolevulinic Acid(ALA) via Aminolevulinic Acid Synthetase(ALAS)
REQUIRES Vitamin B6(pyridoxine) |
|
|
Final step of protoporphyrin synthesis? Where does it occur?
|
Ferrochelatase atatches protoporphyrin to iron to make heme.
This occurs in mitochondria. |
|
|
Classic histological finding in sideroblastic anemia? What causes them?
|
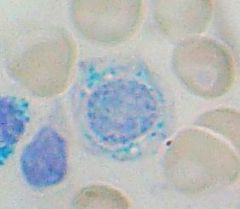
Ringed Sideroblasts
Caused by accumulation of iron in mitochondria surrounding nucleus due to the inability to leave(no functional protoporphyrin to bind to) |
|
|
What stain marks iron?
|
Prussian Blue
|
|
|
Congenital form of sideroblastic anemia caused by?
|
Enzyme defect in ALAS(Aminolevulinic Acid Synthetase) which is the rate limiting step in sideroblastic anemia.
|
|
|
Acquired form of sideroblastic anemia caused by?
|
Alcohol - mitochondrial poison
Lead poisoning - denatures ALAD and ferrochelatase Vitamin B6 deficiency - ALAS cofactor |
|
|
What drug commonly causes vitamin B6 deficiency?
|
Isoniazid treatment
|
|
|
Lab findings in sideroblastic anemia...
|
Increased iron in mitochondria of erythroblasts will cause eventual free radical damage and cellular lysis. Iron leaks out and is picked up by the bone marrow macrophages(increased ferritin, which decreases TIBC). Some iron also leaks out into blood stream, which increases serum iron and % sat.
Increased ferritin, decreased TIBC Increased serum iron, increased % sat |
|
|
What disease are people who are carriers for thalassemia protected against?
|
Plasmodium falciparum malaria
|
|
|
3 types of hemoglobin
|
alpha 2, beta 2 - adult (HbA)
alpha 2, gamma 2 - fetal (HbF) alpha 2, delta 2 - A2 (HbA2) |
|
|
genetics of alpha thalassemia...
|
4 alleles on chromosome 16
problem due to gene DELETION |
|
|
alpha thalassemia common in Asia. Africans?
|
cis-deletion of alpha allele
trans-deletion seen in Africans |
|
|
HbH
|
Beta subunit tetramer
can be seen on electrophoresis |
|
|
HbBarts
|
Gamma subunit tetramer
incompatible with life can be seen on electrophoresis |
|
|
genetics of beta thalassemia
|
problem due to gene MUTATION
|
|
|
isolated increase in HbA2
|
Beta Thalassemia Minor
(Beta/Beta+) |
|
|
Hepatosplenomegaly with evidence of hematopoiesis in facial bones and skull
|
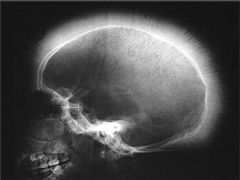
Massive Erythroid Hyperplasia
Due to Beta Thalassemia Major(Beta0/Beta0) Can also happen in Sickle Cell Anemia "Crewcut appearance of skull" Chipmunk like facial structure |
|
|
Aplastic crisis
|
due to parvovirus B19 infection in Beta Thalassemia Major patients.
|
|
|
Target cells
|
present in Beta Thalassemia Minor and Major
also, post splenectomy |
|
|
Characteristic of Beta Thalassemia Major
|
Nucleated RBC's
|
|
|
Electrophoresis findings for Beta Thalassemia Major
|
no HbA
Only see HbA2 and HbF |
|
|
Macrocytic Anemia
|
due to folate or vitamin B12(cobalamin) deficiency (megaloblastic anemia)
THF-M --> VitB12-M --> Methionine (transfer of methyl groups) |
|
|
hypersegmented neutrophils
|
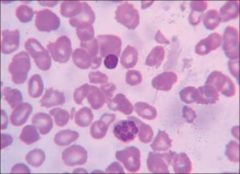
neutrophils with greater than 5 lobes
a result of folate/B12 deficiency |
|
|
Epithelial cell change in Folate/B12 deficiency
|
Megaloblastic change
|
|
|
A drug that can cause macrocytic anemia. Clinical diseases that can cause it?
|
5-Fluorauracil(also Alcohol and Liver disease)
notably, would not see hypersegmented nuclei or megaloblastic change in other cells |
|
|
Where is folate absorbed?
|
jejunum
|
|
|
Glossitis a/w?
|
Folate deficiency and Vitamin B12 deficiency
|
|
|
what kind of leukocyte is characteristic of macrocytic anemias?
|
hypersegmented neutrophils
folate deficiency lab findings macrocytic RBC's Glossitis decreased serum folate increased serum homocysteine Normal methylmalonic acid |
|
|
Methylmalonic Acid usefulness as a lab test?
|
Folate deficiency will have normal MMA because MMA to succinyl coA requires B12 and B12 is normal in folate deficiency.
|
|
|
Where is intrinsic factor made? Where are they located?
|
gastric Parietal cells located on body of stomach
|
|
|
Describe path of Vitamin B12
|
dietary form is bound to animal protein which is then cleaved by amylase from salivary glands. Salivary gland also secrete r-binder which stabilized B12 till it reaches the stomach. In the stomach pancreastic proteases cleave r-binder and intrinsic factor from gastric parietal cells attach to it. IF-B12 complex then absorbed in ileum.
|
|
|
Where is B12-IF complex absorbed?
|
ileum
|
|
|
pernicious anemia arises from?
|
autoimmune destruction of parietal cells
|
|
|
Causes of vitamin B12 deficiency?
|
Pancreatic insufficiency - inability to cleave Vit B12-R-binder complex
Damage to terminal ileum due to Crohn's disease or Diphyllobothrium latum(tapeworm) Dietary deficiency is RARE except for vegans |
|
|
deficiency in vegans
|
Vitamin B12
|
|
|
What causes Subacute combined degeneration of spinal cord. What is the presentation?
|
Vitamin B12 deficiency
degeneration of dorsal and lateral columns leading to poor proprioception and vibratory sensation(dorsal column) as well as spastic paresis(lateral corticospinal tract) |
|
|
spastic paresis occurs due to damage where in the spinal cord?
|
lateral corticospinal tract
|
|
|
proprioception and vibratory sensation dysfunction is due to damage where in the spinal cord?
|
dorsal column
|
|
|
What two reactions require Vitamin B12
|
Conversion of Methylmalonic Acid to Succinyl CoA which is important for fatty acid synthesis.
Transfer of methyl group from THF. B12-methyl then transfers to homocysteine which turns into methionine. |
|
|
Lab findings of vitamin b12 deficiency
|
decreased vitamin b12
increased methylmalonic acid increased homocysteine |
|
|
Describe reticulocyte
|
Slightly larger than normal RBC with a bluish hue due to residual RNA.
|
|
|
Reticulocyte correction in anemia
|

|
|
|
Describe digestion of RBC's by macrophages
|
Globin --> amino acids
Heme ---> Iron(recycled) + protoporphyrin protoporphyrin --> unconjugated bilirubin unconjugated bilirubin binds to albumin and is delivered to liver to be conjugated and excreted into bile |
|
|
Clinical presentation of extravascular hemolysis
|
Anemia with splenomegaly(hypertrophy due to excess digestion of RBC's)
Jaundice due to excess unconjugated bilirubin(overwhelms liver's ability to conjugate bilirubin) Increased risk of bilirubin gallstones, because the bile is super saturated with conjugated bilirubin |
|
|
Lab findings for intravascular hemolysis
|
-- Hemoglobinuria
-- Hemoglobinemia -- Hemosiderinuria - renal tubular cells pick up some hemoblobin and digest it into iron which accumulates as hemosiderin. Then when the renal cells turn over, it shows up in the urine. -- Decreased serum haptoglobin |
|
|
Most common protein defects causing hereditary spherocytosis
|
spectrin, ankyrin, band 3.1
These cause cytoskeletal defects that result in the loss of blebs leading to loss of biconcave disks. |
|
|
Describe RDW in Hereditary spherocytosis
|
SINCE the cause of the spherocytosis is destruction of blebs of membrane from the RBC's by the spleen, this means that older cells will be smaller due to loss of membrane.
Causes increased RDW |
|
|
What is MCHC? How does it change in hereditary spherocytosis?
|
Mean Corpuscular Hemoglobin Concentration. It is INCREASED in hereditary spherocytosis due to the relative increase in hemoglobin per size of RBC since the spleen is picking off blebs of the RBC.
|
|
|
Target cells in thalassemia a result of...
|
removing the Hb in the RBC. This is like "taking the air out of a basketball" and allows the center to bleb out.
|
|
|
Clinical features of Hereditary Spherocytosis
|
Splenomegaly(increased RBC destruction), jaundice with unconjugated bilirubin, increased risk for bilirubin gallstones,
increased risk of aplastic crisis with parvovirus B19 increased RDW and Mean Corpuscular Hemoglobin Concentration(MCHC) |
|
|
how do you diagnose hereditary spherocytosis
|
Osmotic fragility test
Normal cell resistant to hypotonic solution Spherocyte will lyse in osmotic solution(fragile) |
|
|
What kind of cell will you see on peripheral blood smear post-splenectomy?
|
Howell-Jowelly bodies(fragments of nuclear material in RBCs).
An example of when you might see this is Hereditary Spherocytosis treated with a splenectomy. Lack of a spleen which would usually correct these RBCs is absent. |
|
|
Sickle Cell Anemia protects against?
|
protects against Plasmodium Falciparum
|
|
|
What drug will increase levels of HbF?
|
hydroxyurea, MOA unknown
|
|
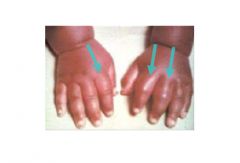
When HbS cells sickle, what happens?
|
They polymerize(NOT Covalent bonds. It is reversible)
This also causes membrane damage, which causes most of the clinical manifestations |
|
|
Clinical manifestations of Sickle Cell Disease
|
A result of RBC membrane damage. Leads to...
Extravascular Hemolysis -- anemia, jaundice(unconjugated hyperbilirubinemia), bilirubin stones risk in gallbladder Intravascular Hemolysis(minor) -- decreases free haptoglobin as it binds to Hb. Target Cells seen in Sickle Cell due to dehydration of RBC's as they get damaged. This means there is air out of basketball. |
|
|
Haptoglobin
|
Circulates in serum and binds to Hb in intravascular hemolysis
|
|
|
Most common cause of death in adults with sickle cell disease?
|
Acute Chest Syndrome -- vasoocclusion in pulmonary microcirculation. Presents with chest pain, shortness of breath and lung infiltrates. Can be precipitated by pneumonia
|
|
|
Renal Papillary Necrosis
|
Hematurea, and proteinurea due to vasoocclusion a/w Sickle Cell Disease
|
|
|
What organ is most susceptible to damage in sickle cell TRAIT individuals? What is the classic finding of long term complications related to this fact?
|
renal medulla
loss of the ability to concentrate urine |
|
|
What chemical induces sickling?
|
Metabisulfite
Will be positive in both trait and disease |
|
|
Name the types of hemoglobin present in sickle cell disease and trait
|
Sickle Cell Disease - HbS, HbF, HbA2 (NO HbA present!!)
Sickle Cell Trait - HbA, HbS, HbA2 |
|
|
Hemoglobin C high yield association?
|
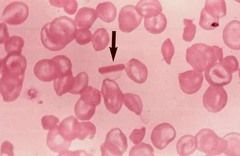
HbC Crystals seen on blood smear.
|
|
|
Paroxysmal Nocturnal Hemoglobinurea
|
ACQUIRED defect in myeloid stem cell that messes up GPI which anchor DAF and MIRL to RBC. As such, DAF and MIRL are not present on RBC which means complement can lyse them.
When you sleep, your respiratory drive decreases causing a transient respiratory acidosis which can activate complement leading to destruction of RBC. DAMMIT DAF AND MIRL WHERE WERE YOU!! |
|
|
Glutathione Reductase Pathway
|
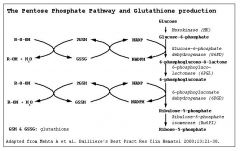
|
|
|
Variants of G6PD?
|
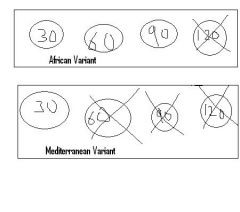
African variant -- mildly reduced half life of G6PD leading to mild intravascular hemolysis with oxidative stress
Mediterranean Variant - markedly reduced half life of G6PD leading to marked intravascular hemolysis. |
|
|
Oxidative stress?
|
Infection, Drugs(sulfa, nitryl, primaquine), and Fava beans(mediterranean diet)
|
|
|
Classic sign in G6PD?
|
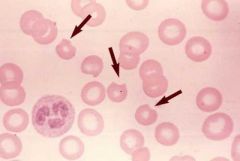
Heinz bodies -- precipitated Hb due to oxidative stress. These precipitates are bitten out of the RBC in the spleen leading to 'bite cells'
|
|
|
G6PD Clinical signs?
|
Intravascular hemolysis causing hematuria. This causes back pain since Hb is nephrotoxic
|
|
|
Normocytic anemias with Predominantly Intravascular hemolysis
|
PNH, G6PD, IHA(Immune Hemolytic Anemia), Microangiopathic Hemolytic Anemia, Malaria
|
|
|
Heinz Preparation?
|
Test for G6PD
|
|
|
Immune Hemolytic Anemia
|
IgG or IgM mediated destruction of RBC's
IgG is usually EXTRAvascular IgM is usually INTRAvascular |
|
|
IgG type Immune Hemolytic Anemia Clinical Signs
|
IgG will present with spherocytes since antibody coating of RBC will cause spleen to pick of bits and pieces of cytoplasm.
|
|
|
IgG type Immune Hemolytic Anemia associated with...?
|
extravascular hemolysis
SLE(most common), CLL, and drugs(penicillin, cephalosporin, alpha-methyldopa) penicillin can bind to RBC yielding Ab complexes or alpa-methyldopa can induce production of self-Ab to RBCs |
|
|
Treatment of IgG type Immune Hemolytic Anemia
|
IVIG -- eat IVIG instead of RBCs
Splenectomy |
|
|
IgM type Immune Hemolytic Anemia Associations
|
INTRAvascular hemolysis. A/w Mycoplasma Pneumonia(cold agluttinins) that cause complement activation in relative cold temp of extremities.
Also associated with infectious mononucleosis |
|
|
Overview of Coombs Test
|
Direct Coombs Test --- Do I have RBC's already bound to IgG?
---> add Anti-IgG Ab to solution of patient RBC Indirect Coombs Test -- Do I have Ab to RBC's in my serum? ---> Anti-IgG and test RBC's are added to patient serum |
|
|
Microangiopathic Hemolytic Anemia
|
Formation of Schistocytes due to creation of thrombi in microcirculation.
Occurs in A/w TTP(defective ADAMTS13), HUS(ETEC O157), DIC, HELLP, Prosthetic heart valves and aortic stenosis. Calcified valves shear RBCs |
|
|
'helmet' shaped RBC
|
Schistocyte
|
|
|
Malaria transmitted by?
|
Anopheles mosquito transmits the Plasmodium species of bug.
|
|
|
Plasmodium falciparum vs vivax/ovale?
|
Plasmodium falciparum -- daily fever
Plasmodium vivax/ovale -- fever every other day |
|
|
Intravascular Hemolysis can be caused by?
|
G6PD
Paroxysmal Nocturnal Hemoglobinuria Microangiopathic Hemolytic Anemia Immune Hemolytic Anemia Malaria |
|
|
Extravascular Hemolysis can be caused by?
|
Hereditary Spherocytosis
Sickle Cell Anemia |

