![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
42 Cards in this Set
- Front
- Back
|
Add H2O to alkene
|
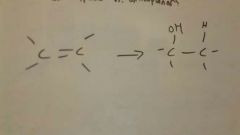
Mark
|
|
|
Reactivity of Electrophiles
|
Best have:
1. Electrophilic carbon 2. Good leaving group |
|
|
Strength of nucleophile rules
|
1. Negative charge is good
2. Opposite of electronegativity 3. Polarizability is good BUT NOT FOR BASES 4. Hindered is bad; OPPOSITE FOR BASES |
|
|
Leaving group rules
|
1. Electron withdrawing
2. Stable after leaving (not a strong base) 3. Polarizable (to stabilize transition state |
|
|
SN2 Stereochemistry
|
Yes;
Walden Inversion |
|
|
SN1 Stereochemistry
|
Could do retention or inversion (50/50)
|
|
|
E2 Stereochemistry
|
Anti-coplanar, syn-coplanar stuff;
Anti-coplanar is favored |
|
|
Add H2, Pt/Pd/Ni to alkene
|
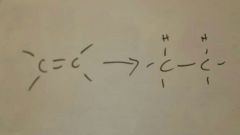
|
|
|
Add HOOH to alkene
|
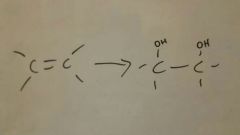
|
|
|
Oxidative Cleavage (O2)
|
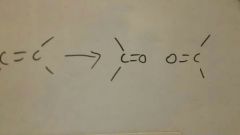
|
|
|
Add R-COOOH to alkene
|
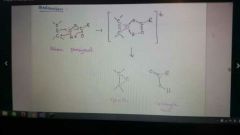
If you then add H3O+, makes a glycol (anti dihydroxylation)
|
|
|
Add X2 to alkene
|
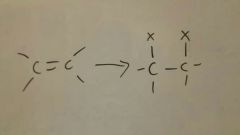
Stereochemistry: anti
|
|
|
Add X2, H2O to alkene
|
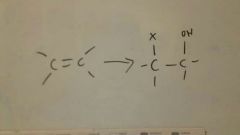
Stereochemistry: anti
Mark on respect to OH |
|
|
Add HX to alkene
|
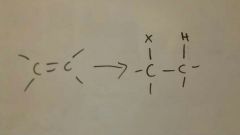
|
|
|
Cyclopropanation
|
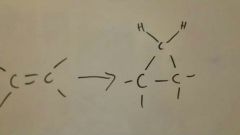
|
|
|
Free Radical Addition of HBr (mechanism)
|
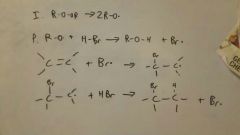
Anti-Mark (because that puts the free radical on the more stable carbon)
|
|
|
How to get more E2, less SN2?
|
Use bulky base
|
|
|
How to get more Hoffmann product in Alkene synthesis
|
Use bulky base
|
|
|
Add Hg (OAc)2, H2O, NaBH4 to alkene
|
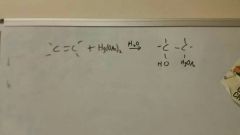
Also works with alcohols
|
|
|
Add BH3 to alkene
|
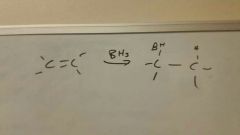
Anti-Mark
Then add O (usually in form of H2O2/-OH/H2O) to make anti-Mark alcohol |
|
|
Adding Br to a solution to test for DB
|
No DB = color stays (no rxn)
DB = Clear (rxn) |
|
|
Addition of carbenes to alkenes (:CR2)
|
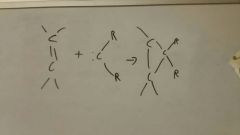
|
|
|
Making :CH2 from diazomethane
|
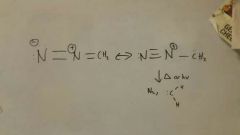
|
|
|
Simmons-Smith Reaction
|

Does addition of carbene
|
|
|
McPBA
|
Metachlorobenzoic acid;
Epoxidates alkenes; It with H3O+ works like cold, dilute KMnO4 (syn dihydroxylation) |
|
|
Add OsO4, H2O2 to alkene
|
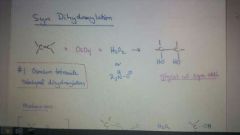
|
|
|
Add KMnO4 to alkene
|
1. Cold, dilute = syn dihydroxylation
2. Warm, concentrated = cleaves into two ketones |
|
|
Add O3, Me2S to Alkene
|
Oxidative Cleavage
|
|
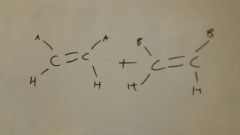
|
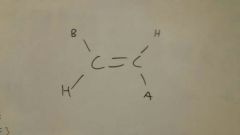
Olefin metathesis
|
|
|
Make acetylide from R-CC-H
|
Add NaNH2
|
|
|
Acetylide + carbonyl group (ketone)
|
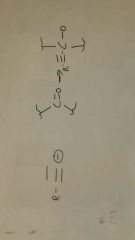
Add another H+ to make a propanyl alcohol
|
|
|
Synthesis of Alkyne?
|
Eliminate vicinal or geminal dihalides using a base
1. KOH at 200 for internal 2. NaNH2 at 150, then H2O/H+ for terminal |
|
|
Add A-B to alkyne
|
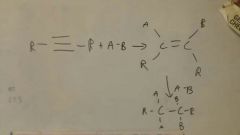
|
|
|
Add H2, Pt to alkyne
|
becomes alkane
|
|
|
Add H2, Pd/BaSO4 to Alkyne
|
Becomes cis alkene because it's poisoned
|
|
|
Add NaNH3 to alkyne
|
makes trans alkene
|
|
|
Add X2 to alkyne
|
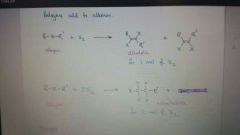
|
|
|
Add HX to alkyne
|
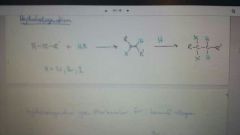
Mark for terminal alkynes
|
|
|
Add HgSO4, H2SO4, H2O to alkyne
|

|
|
|
Add R2BH to alkyne
|

|
|
|
Add KMnO4 to alkyne
|
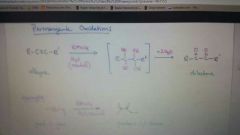
|
|
|
Add KMnO4 and base to alkyne
|

|

