![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
50 Cards in this Set
- Front
- Back
|
Valence
|
number of bonds an atom usually forms
C = 4, tetravalent N = 3, trivalent P = 3, trivalent O = 2, divalent S = 2, divalent H & halogens = 1, monovalent |
|
|
Formal charge
|
number of electrons in the isolated atom, minus # of electrons assigned to the atom in lewis structure
sum of formal charges for each atom in molecule or ion represents total charge on molecule or ion |
|
|
Fischer projection
|
vertical lines are assumed to be oriented into the page
horizontal lines are assume to be oriented out of page |
|
|
Newman projection
|
view straight down axis of 1 of sigma-bonds
both intersecting lines and large circle are assumed to be C atoms |
|
|
Index of Hydrogen Deficiency AKA degree of unsaturation
|
# of pairs of H a compound requires in order to become a saturated alkane
saturated alkane contains (2n + 2) # of H, where n = # C A double bond and ring each counts as one IHD. A triple bond counts as two IHD. count halogens as H, ignore O, count N as 1/2 H Hydrocarbons (CxHy): IHD = (2x + 2 - y)/2 (where x and y stand for # of C and H respectively.) index of H deficiency of saturated alkane = zero |
|
|
Electrostatic force
|
force between electrons and nuclei that creates all molecular bonds
2 electrons are required to form a bond electrons are at lowest energy level when they form a bond because they have minimized their distance from both nuclei each bonded nuclei can donate a single electron to the bond |
|
|
Coordinate covalent bond
|
one nucleus donates both electrons to the bond
|
|
|
Sigma-bond
|
forms when bonding pair of electrons are localized directly between 2 bonding atoms
lowest energy, most stable form of covalent bond, strong always 1st type of covalent bond to formed between any 2 atoms single bond must be a sigma-bond any double or triple bond, contains 1 sigma-bond |
|
|
Pi-bonds
|
additional bonds that form between 2 sigma-bonded atoms
orbital of 1st Pi-bond forms above and below sigma-bonding electrons because sigma-bond leaves no room for other electron orbitals directly between atoms 1 pi-bond = double bond (orbital above and below sigma-bond) 2 pi-bonds = triple bond (orbital on either side of sigma-bond) weaker and more reactive than sigma-bond, but strengthen and shorten overall bond C, N, O & S form pi-bonds pi-bonds prevent rotation |
|
|
Bond energy
|
energy necessary to break a bond
If a bond is weaker, it has a lower bond energy, and it takes less energy to break the bond |
|
|
Atomic orbitals
|
s, p, d and f orbitals
The orbitals have subshells (s has 1, p has 3..) |
|
|
hybrid orbital types:
|
1. sp
2. sp^2 3. sp^3 |
|
|
Character
|
superscripts indicate the character as follows:
sp^2 = 1s + 2p = 33% s + 66% p hybrid orbitals resemble in shape and energy the s and p orbitals from which it is formed to the same extent that s or p orbitals are used the more s character a bond has, the more stable, stronger, shorter the bond becomes |
|
|
hybridization, bond angles and shape
|
sp = 180 = linear
sp^2 = 120 = trigonal planar sp^3 = 109.5 = tetrahedral, pyramidal or bent sp^3d = 90, 120 = trigonal-bypyramidal, seesaw, T-shaped or linear sp^3d^2 = 90 = octahedral, square pyramidal or square planar |
|
|
Resonance structure
|
2 or more lewis structures representing molecules with delocalized electrons
weighed average of these structures represents the real molecule (lower energy than lewis structures) |
|
|
4 rules for resonance structures
|
1. atoms must not be moved (move electrons, not atoms)
2. # of unpaired electrons must remain constant 3. resonance atoms must lie in same plane 4. only proper lewis structures allowed |
|
|
2 conditions exist for resonance structures to occur
|
1. a species must contain an atom either with a p orbital or an unshared pair of electrons
2. that atom must be single bonded to an atom that possesses a double or triple bond (Conjugated unsaturated systems) |
|
|
Criteria for an aromatic compound (Huckel's rule)
|
The molecule is cyclic (a ring of atoms)
The molecule is planar (all atoms in the molecule lie in the same plane) The molecule is fully conjugated (p orbitals at every atom in the ring) 4n + 2 = # of pi-electrons and n should equal any integer or 0. |
|
|
Polar molecule or bond
|
molecule or bond with dipole moment
results from differences in electronegativity of its atoms molecules with polar bond may or may not have a dipole moment |
|
|
Induced dipoles
|
weaker than permanent dipoles
dipole moment is momentarily induced in an otherwise nonpolar molecule or bond by a polar molecule, ion or electric field |
|
|
Instantaneous dipole moment
|
electrons in bond move about orbital, and at any given moment may not be evenly distributed between 2 bonding atoms
very short lived and weaker than induced dipoles can act to induce dipole in neighboring atom |
|
|
Intermolecular attractions
|
attractions between separate molecules
occur solely due to dipole moments must be weaker than covalent forces (1% as strong) attraction between molecules in proportional to their dipole moments |
|
|
Hydrogen bond
|
intermolecular bond formed when H is attached to a highly electronegative atom (F, O, or N) it creates a large dipole moment leaving H with strong partial positive charge. When H approaches N, O or F on another atom, intermolecular bond is formed
|
|
|
London Dispersion Forces
|
weakest dipole-dipole force
between 2 instantaneous dipoles very weak, but are responsible for phase changes of nonpolar molecules |
|
|
Isomers
|
2 molecules are isomers if they have same molecular formula but are different compounds
|
|
|
Conformational isomers (conformers)
|
not true isomers
different spatial orientations of the same molecule simplest way to distinguish between conformers is with newman projections (eclipsed, staggered) |
|
|
Structural isomer/constitutional isomer
|
simplest form of isomer
same molecular formula but different bond-to-bond connectivity ex: isobutane and n-butane, both are C4H10, but have different structures |
|
|
Stereoisomers
|
2 molecules with same molecular formula and same bond-to-bond connectivity that are not same compound
unless geometric isomers, stereoisomers much each contain at least 1 chiral center in same location 2 types: 1. enantiomers 2. diastereomers |
|
|
Chirality
|
handedness of a molecule
chiral molecules differ from their reflections achiral molecules are exactly the same as their reflections Chiral compounds have absolute configs: R or S |
|
|
Absolute configuration
|
physical description of orientation of atoms about a chiral center (such as a chiral carbon)
Determined by R (right) & S (left): 1. atoms attached to chiral center or #ed from higher (higher atomic weight) to lowest priority (smaller atomic weight) 2. substituents on double and triple bonds are counted 2 or 3 times, respectively 3. lowest priority group faces away 4. circle is drawn from lowest to higher priority 5. clockwise (R) and counter-clockwise (S) 6. mirror image always has opposite absolute configuration |
|
|
Relative configuration
|
not related to absolute configuration
2 molecules have the same relative configuration about a C if they differ by only 1 substituent and other substituents are oriented identically about C |
|
|
Observed rotation
|
direction and degree that electric field in plane-polarized light rotates when it passes through a compound. Measured using a polarimeter.
Clockwise rotation: + or D Counterclockwise rotation: - or L |
|
|
Polarimeter
|
screens out photons with all but one orientation of electric field
resulting light consists of photons with their electric fields oriented in same direction |
|
|
Plane-polarized light
|
white light that has passed through a polarimeter and now has photons all oriented in same direction
|
|
|
Optically inactive
|
may be compounds with no chiral centers or contain equal amounts of both stereoisomers
compound does not rotate light no single molecular orientation is favored, so there is no rotation of plane of electric field. |
|
|
Racemic mixture
|
optically inactive compound
contains equal amounts of both stereoisomer therefore no rotation of light is observed |
|
|
Optically active
|
compound that rotates light, orientation of electric field is rotated
racemic mixture is separated, resulting in compound containing molecules with no mirror images if rotates plane-polarized light clockwise = + or d (right) if rotates plane-polarized light counter-clockwise = - or l (left) |
|
|
Specific rotation
|
a standardized form of observed rotation
calculated from observed rotation and experimental parameters |
|
|
Enantiomers
|
same molecular formula, same bond-to-bond connectivity, not same molecule, mirror images of each other
opposite absolute configurations (R and S) at each chiral C C-C C-C Enantiomers R S S R C-C C-C Enantiomers R R S S rotate plane-polarized light in opposite directions to an equal degree Enantiomers have same physical and chemical properties except: 1. reactions with other chiral compounds 2. reactions with polarized light make racemic mixture, when mixed together in equal concentrations |
|
|
Resolution
|
separation of enantiomers
|
|
|
Diastereomers
|
same molecular formula, same bond-to-bond connectivity, not same molecule, NOT mirror images of each other
C-C C-C Diastereomers R S R R C-C C-C Diastereomers R S S S |
|
|
Geometric isomer
|
type of diastereomer
A type of diastereomer that exists due to hindered rotation about a bond (ring structure, double or triple bond) (cis or trans) different physical properties 1. 2 substituents on each C are prioritized using atomic weight 2. higher priority substituent for each C on opposite sides = E 3. higher priority substituent for each C same side = Z |
|
|
Cis-isomers
|
diastereomers. geometric isomers. molecules with same side substituents
have dipole moment stronger intermolecular forces leading to higher boiling points (due to dipole moment) do not form crystals as readily leading to lower melting points (due to lower symmetry) steric hindrance (substituents crowd each other) of cis isomers produce higher energy levels resulting in higher heats of combustion |
|
|
Trans-isomers
|
diastereomers. geometric isomers. molecules with opposite-side substituents
do not have dipole moment |
|
|
Maximum # of optically active isomers that a compound can have?
|
2^n
n: # of chiral centers |
|
|
Meso compounds
|
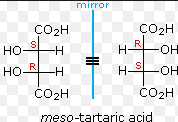
2 chiral centers in a single molecule that offset each other creating an optically inactive molecule
plane of symmetry through their centers which divides them into 2 halves that are mirror images to each other are achiral and therefore optically inactive |
|
|
Epimers
|
Diastereomers that differ at only 1 chiral C
when ring closure occurs at epimeric C, 2 possible diastereomers may be formed |
|
|
Anomers
|
distinguished by orientation of substituents (in glucose, if the hydroxyl on the anomeric carbon is axial it is alpha, if equitorial it is beta)
|
|
|
Anomeric carbon
|
the chiral C of an anomer
|
|
|
When are carbon compounds gaseous?
|
Any carbon compound with less than 4 carbons is probably a gas. Greater than that is liquid/solid
|

