![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
183 Cards in this Set
- Front
- Back
|
litmus paper turns red in ? |
acid
|
|
|
litmus paper turns blue in?
|
bases
|
|
|
bases turn litmus paper?
|
blue
|
|
|
arrhenius def of acid and base
acid is? base is? |
acid prod H+
base prod OH- |
|
|
what is the shortcoming of arrhenius def?
|
only desc a and b in aq media, not in non aqueous
|
|
|
bronst lowry a and b
a is b is? |
acid donate h+
bases accepts H+ |
|
|
t or f
NH3 and CL- are bronst lowry bases but not arrhenius bases |
t
|
|
|
t or f
a bronsted lowry defin of an a or a b is limited to aq solutions |
f
|
|
|
the ______________ defin of an a or a b is limited to aq solut
|
arhhenius
|
|
|
bronst lowry acid and b occur in pairs called?
|
conjugate a b pairs
|
|
|
bronst lowry
2 members of a conj pair are related by a transf of? |
a proton
|
|
|
H3O+ is the conjugate acid of the base?
|
H2O
|
|
|
NO2 - is the conjugate b of ?
|
HNO2
|
|
|
lewis def of a and b
acid base? |
acid is electr pair accept
base is electr pair donator. |
|
|
AlCl3 is a _______________ acid but not a ________________ acid
|
lewis but not a bronst lowry
|
|
|
AlCl3 can accept a?
|
electron pair
|
|
|
name of an acid is related to its?
|
parent anion
|
|
|
anion ending in -ide will become ______________ acid
|
hydro ic
|
|
|
flouride bec acid?
|
hydrofluoric acid
|
|
|
bromide becomes ________________ acid
|
hydrobromic acid
|
|
|
HF is?
|
hydrofluoric acid
|
|
|
HBr is ?
|
hydrobromic acid
|
|
|
hydrofluoric acid is?
|
HF
|
|
|
MnO4- is what anion?
|
permanganate, even though there are no manganate or manganite ions
|
|
|
acids from oxyanions are called?
|
oxyacids
|
|
|
anion ends in ite it will be __________ acid
|
ous acid
|
|
|
anion ends in ate, it will be ____________ acid
|
ic acid
|
|
|
CLO-
|
hypochlorite
|
|
|
ClO2-
|
chlorite
|
|
|
ClO3-
|
chlorate
|
|
|
ClO4-
|
perchlorate
|
|
|
NO2-
|
nitrite
|
|
|
NO3-
|
nitrate
|
|
|
HClO
|
hypochlorous acid
|
|
|
hypochlorite
|
CLO-
|
|
|
chlorate
|
CLO3-
|
|
|
perchlorate
|
ClO4-
|
|
|
nitrite
|
NO2-
|
|
|
nitrate
|
NO3-
|
|
|
HClO2
|
chorous acid
|
|
|
chlorous acid
|
HClO2
|
|
|
HClO3
|
chloric acid
|
|
|
perchloric acid
|
HClO4
|
|
|
HNO2
|
nitrous acid
|
|
|
HNO3
|
nitric acid
|
|
|
nitric acid
|
HNO3
|
|
|
ph=
|
-log[H+]=log(1/[H+])
|
|
|
pOH is=
|
-log[OH-]= log (1/[OH-])
|
|
|
in any aq solut the H2O does what?
|
dissociates slightly
|
|
|
Kw is the?
|
water dissociation constant
|
|
|
Kw= [H+][OH-]=
|
10-14
|
|
|
Kw=
|
[H+][OH-]=10-14
|
|
|
ph + pOH=?
|
14
|
|
|
math reminder
log(xy)=? |
logx + logy
|
|
|
pure water _____________ is equal to ________________
|
H+ is equal to OH-
|
|
|
pH below 7 is
|
acidic
|
|
|
pH above 7 indicates an excess of?
|
OH- ions
|
|
|
how can you estimate a log
n x 10-m = |
log(n x 10-m)= -m + logn
the negative log is m-logn |
|
|
m-logn
since n is a number betw 1 and 10 its log will be a fractoin betw ? |
0 and 1
|
|
|
m-logn
since n will be a number betw 0 and 1. m-logn will be between m-1 and ? |
m-0
|
|
|
m-logn
the larger n is the larger ? |
the fraction logn will be the answer will be closer to m-1
|
|
|
If Ka = 1 .8 x 10-5, then pKa =
estimate please |
5 - log 1.8. Since 1 .8 is small,
its log will be small, and the answer will be closer to 5 than to 4. (The actual answer is 4.74.) |
|
|
strong acids and bases are those that?
|
completely dissociate into their component ions in aq solution
|
|
|
when NaOH dissoc in water, why can you normally ignore the contribution of OH- from H2O?
|
as long as the amount of OH- or H+ is greater than 10-7 then you can ignore contribution of water.
|
|
|
when cant you ignore the contrib of water to acid and base calculations?
|
when it is a very weak acid or base whose H+ or OH- concentr is close to 10-7
|
|
|
1 x 10-8 M HCL solution
Kw=(x+ 1 xlO-8)(x)=1.O x 1O-14 molecules). what is X? |
where x=[H+]=[OH-] (bothfrom
the dissociation of water |
|
|
1 x 10-8 M HCL solution.
Kw=(x+ 1 x lO-8)(x)=1.O x 1O-14 molecules). when do use this kind of equation. ? |
when you have to calcul the H+ concentr with a weak acid.
you could also use the equation for a weak base |
|
|
name some strong acids
|
HClO4- perchloric acid
HNO3 nitric acid H2SO4 sulfuric acid HCl hydrochloric acid |
|
|
sulfuric acid is?
|
H2SO4
|
|
|
HCl is?
|
hydrochloric acid
|
|
|
HNO3 is?
|
nitric acid
|
|
|
name some strong bases
|
NaOH sodium hydroxide
KOH potassium hydroxide other soluble hydroxides of gr IA and IIA metals |
|
|
weak acids and bases only partly?
|
dissociate in aq sol
|
|
|
Ka measures?
|
the degree to which an a dissoc
|
|
|
the degree to which an an a dissoc is measur by?
|
the acid dissoc const Ka
|
|
|
t or f
Ka=[H3O][A-]/[HA] |
t
|
|
|
Ka=
|
=[H3O][A-]/[HA]
|
|
|
Note that as a weak acid or
base, the effect on pH will always be _______________ of a strong acid or base of the same concentration. |
less than that
|
|
|
weaker the acid, the smaller the?
|
Ka
|
|
|
the weaker the acid, the _____________ the Ka
|
smaller
|
|
|
Kb is the
|
base dissociation constant
|
|
|
the weaker the base the ____________ the Kb
|
smaller
|
|
|
a conjugate base is formed when?
|
an a loses a proton
|
|
|
HCO3-/CO3-
which is the conjugate a and b? |
conjugate a HCO3-
conjugate b CO3- |
|
|
To find the Ka of the conjugate acid HC03, the reaction with __________
must be considered. |
water
|
|
|
HC03- (aq) + H20 (I) ---> H3O+ (aq) + C03 2- (aq)
this reaction is used to calculate the ? |
Ka of HCO3-
|
|
|
Ka X Kb=Kw=l x10-14
this is for an acid and its? |
conjugate base
|
|
|
what can we calculate for an acid and its conjugate base?
|
Ka X Kb=Kw=l x10-14
|
|
|
Ka and Kb are _____________ related
|
inversely
|
|
|
Ka and _______ are inversely related
|
Kb
|
|
|
if Ka is large then Kb?
|
will be small
|
|
|
if the conjugate a is strong then the conjugate base?
|
will be weak
|
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5) what should you do first?
|
write the equilibrium reaction
|
|
|
CH3COOH (Ka=1.8 X 10-5)
write the equilibrium react |
CH3COOH (aq)---> H+ (aq) + CH3COO- (aq)
|
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5) what do you do after writing the equil react?
|
write the express for the acid dissoc const
|
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5)
write the acid dissoc const? |
Ka=[H+][CH3COO-]
------------ = 1.8 X 10-5 [CH3COOH] |
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5)
what is the concentr of acetic acid at equil equal to? |
its initial concentr 2.0 M- X, the amount dissociated
|
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5)
[H+] is=? |
=[CH3COO-]=x
|
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5)
How can you rewrite the Ka expression? |
Ka= [X][X]/[2.0-X]=1.8 X 10-5
|
|
|
to calculate the concentration of H+ in a 2.0 M aq sol of acetic acid CH3COOH (Ka=1.8 X 10-5)
what can you approximate? |
that 2.0-x is approx 2.0 bec acetic acid is a weak acid
|
|
|
Ka= [X][X]/[2.0]=1.8 X 10-5
what did you approximate? |
that 2.0-x is approx = to 2.0
|
|
|
Ka= [X][X]/[2.0-X]=1.8 X 10-5
if when you solve for X it is close the the original concentr of acetic acid (2.0 M) what do you need to do? |
you cant approx that 2.0-x is =2.0
instead you have to use the quadratic equation |
|
|
a neutrialization react is when?
|
an a and a base reat w each other, forming a salt and usually water
|
|
|
equation for neutralization
|
HA + BOH ---> BA + H2O
|
|
|
HA + BOH ---> BA + H2O
this is the equatrion for? |
neutralization
|
|
|
t or f
neutralization reactions usually go to completion |
t
|
|
|
salt ions reacting with water to give back the acid or base is ?
|
hydrolysis
|
|
|
hydrolysis (in terms of a and b) is?
|
reverse react of neutralization, when salt ions react w water to give back the a or b
|
|
|
what combinations of a and b are possible?
|
1. str a and str b
2. str a and weak b 3. weak a and str b 4. weak a and weak b |
|
|
this is an ex of?
HClO + NaOH ---> NaClO + H2O |
weak acid w a str base
|
|
|
prod of a react betw equal concentr of a str acid and str base are?
|
salt and water, a and b neutr each oth so ph is 7
|
|
|
prod of a react betw str a and weak b are?
|
SALT, but usually no water is formed bec weak b are usually not hydroxides
|
|
|
weak bases are usually not?
|
hydroxides
|
|
|
str a w a weak b
the cation of the salt formed will do what? |
will react w the water solvent, reforming the weak b
|
|
|
what are the react for HCL reacting with NH3?
|
HCl (aq) + NH3 (aq) --> NH4+ (aq) + Cl-(aq) Reaction I
NH4+ (aq) + H20 (aq) --> NH3 (aq) + H3O+(aq) Reaction II |
|
|
str acid with a weak base the resulting solut will be?
|
acidic
|
|
|
weak a with a str b the resulting solut is?
|
basic.
|
|
|
weak a with a str b why is the result solut basic?
|
bec of hydrolysis of the salt to reform the aci and format of OH- from hydrolyzed water molecules.
|
|
|
ph of a weak a and a weak b depends on?
|
relative strenght of the reactants
|
|
|
the acid HClO has a Ka = 3.2 X 10-8,
and the base NH3 has a Kb = 1.8 x 1O-5. what will the resulting solut be? acidic, basic, or neutral? |
an aqueous solution of HCIO
and NH3 is basic since Ka for HCIO is less than Kb for NH3 |
|
|
an a equiv is equal to ?
|
one mole of H+
|
|
|
a b equiv is equal to?
|
one mole of OH- ions
|
|
|
H2SO4 is
a. monoprotic b. diprotic c. triprotic |
b. diprotic
|
|
|
H2S04(aq) --> H+(aq) + HS04(aq)
HS04(aq) --> H+(aq) + S042-(aq) what is this reaction showing? |
that H2SO4 is diprotic
|
|
|
what is the equation for the dissoc of H2SO4?
|
H2S04(aq) --> H+(aq) + HS04(aq)
HS04(aq) --> H+(aq) + S042-(aq) |
|
|
one mole of H2SO4 can prod ______ acid equiv
|
2
|
|
|
2M H3PO4 would have be _________ N
|
6
|
|
|
how do you calcul equival weight?
|
divide the gram molecular weight by how many moles of H+ it liberates
|
|
|
what is equival weight of H2SO4?
|
98 g/mol is molec weight
since each mole liberates 2 acid equiv, the gram equiv of H2SO4 is 98/2 or 49 g. |
|
|
name 3 polyvalent acids?
|
H2SO4
H3PO4 H2CO3 |
|
|
amphoteric means?
|
can act as an acid or a base
|
|
|
a substance that can act as an a or a b is?
|
amphoteric
|
|
|
bronsted lowry
an amphoteric species can? |
gain or lose a proton
|
|
|
t or f
water is amphoteric |
t
|
|
|
the partially dissoc conjug base of a polyprotic acid is?
|
usually amphoteric
|
|
|
the hydroxides of Al, Zn, Pb, and Cr are?
|
amphoteric
|
|
|
the hydroxides of which metals are usually amphoteric?
|
Al, Zn, Pb, and Cr
|
|
|
t or f
species that can act as either oxidiz or reducing agents are considered to be? |
amphoteric
|
|
|
t or f
spec that can act as red or oxidiz agents are consid to be amphoteric |
t
|
|
|
oxid or reduc agents act amphoteric when they ?
|
accept or donate electr pairs, acting like Lewis a or bases
|
|
|
titration is used to ?
|
determ the molarit of an a or a b
|
|
|
in titration
you react a ______ volume of a solution of unknown concentrat with a _______ volume of a sol of known concentrat |
known vol
known vol o |
|
|
equival point is when the?
|
number of acid equival equals the number of base equival
|
|
|
t or f
the equival point is always at ph of 7 |
f
only for str a with a str base |
|
|
when titrating polyprotic a or b there are several?
|
equivalence points
|
|
|
you estimate the equival point in titrat by what 2 methods?
|
1. graphical method- use ph meter
2. watching for a color ch |
|
|
indicators are?
|
weak organic a or bases that have differ colors in their undissoc or dissoc states
|
|
|
why dont indicators change the equival point
|
bec they are in low concetr
|
|
|
point at which the indicat ch color is called the?
|
end point
|
|
|
the _______ of the indicator and the __________ of the a and b should be close.
|
end point
equivalence point |
|
|
what formula do yo use to calculate the volume added to reach the endpoint?
|
NaVa=NbVb
|
|
|
no, bec the end point has a pretty different ph than the equival point of the titration
|

accord to this graph, has the best indicator been chosen?
|
|
|
acidic species
|

in the early part of the curve, the __________ species
|
|
|
it will only change the ph a little
|

in the early part of the curve, the acidic spec dominates so small amounts of base will do what to the ph?
|
|
|
near the equivalence point. this is where the ph the most
|

the addit of base most ch the concentr of H+ and OH- where on the graph?
|
|
|
weak acid
|
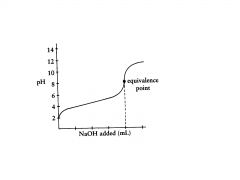
this is a titration of a ____________ with a strong base
|
|
|
t
|
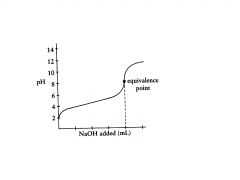
t or f
the titration of any monoprotic a with a str b will give a similar curve to this |
|
|
basic
|
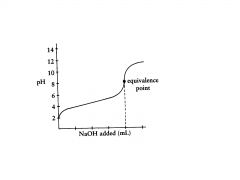
the equival point is in the _______ range
|
|
|
a buffer consists of a weak acid or a weak base and?
|
its salt
|
|
|
give 2 examples of buffers?
|
examples of buffers are: a solution of acetic acid (CH3COOH) and its salt,
sodium acetate (CH3COO-Na+); and a solution of ammonia (NH3) and its salt, ammonium chloride (NH4+Cl-). |
|
|
why are buffers useful?
|
Buffer solutions have the useful
property of resisting changes in pH when small amounts of acid or base are added. |
|
|
Consider a buffer solution of acetic acid and sodium acetate:
CH3COOH <---> H+ + CH3COO- what happens when NaOH is added to the buffer? |
When a small amount of NaOH is added to the buffer, the OH- ions
from the NaOH react with the H+ ions present in the solution; subse- quently, more acetic acid dissociates (equilibrium shifts to the right), restoring the [H+]. Thus, an increase in [OH-] does not appreciably change pH. |
|
|
Consider a buffer solution of acetic acid and sodium acetate:
what happens when a small amount of HCl is added? |
H+ ions from the HCl react with the acetate ions to form acetic acid. Thus
[H+] is kept relatively constant and the pH of the solution is relatively unchanged. |
|
|
the henderson-Hasselbalch equation is used for?
|
The Henderson-Hasselbaich equation is used to estimate the pH ot
a solution in the buffer region where the concentrations of the species and its conjugate are present in approximately equal concentrations. |
|
|
Henderson- Hasselbach equation is?
|
pH = PKa + log [conjugate base]/[weak acid]
|
|
|
henderson-hasselbach
pH = PKa + log [conjugate base]/[weak acid] when the concentr of conj base equals the conc of the weak acid then? |
ph=pKa, bec log1=0
|
|
|
henderson-hasselbach
pH = PKa + log [conjugate base]/[weak acid] when will the concentr of conj base equals the conc of the weak acid ? |
in a titration, half-way to the equivalent point
|
|
|
you can use the henderson-hasselbach equation to make a buffer at any?
|
pH. by carefully choosing a weak acid and its salt
|
|
|
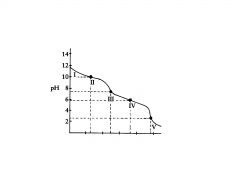
polyprotic base
|
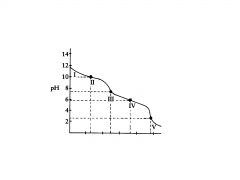
this is titration of a __________ base with an acid
|
|
|
H2C03
|
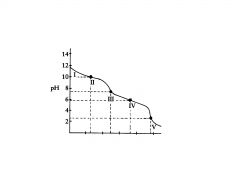
This the the titration of
Na2CO3 with HCI in which the polyprotic acid is the ultimate prod- uct. |
|
|
C032-
|
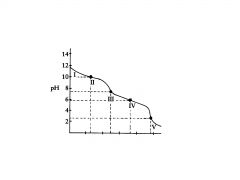
In region I, little acid has been added and the predominant species is
|
|
|
C032- and HC03,
first buffer region |
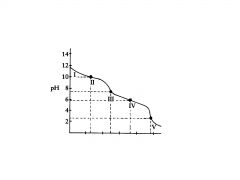
In region Il, more acid has been added and the predominant
species are ______________, in relatively equal concentrations. The flat part of the curve is the ______________, corresponding to the PKa of HC03 |
|
|
equivalence point
|
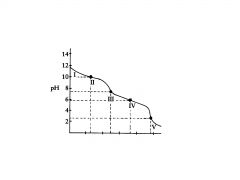
Region Ill contains the _______________ , at which all of the C032 is
titrated to HC03. As the curve illustrates, a rapid change in pH occurs at the ____________ |
|
|
C032 is
titrated to HC03. |
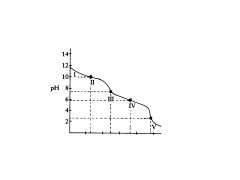
Region Ill contains the equivalence point, at which all of the ?
|
|
|
H2C03 and HC03
|
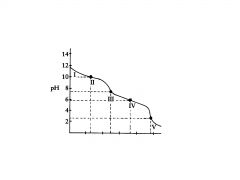
In region IV, the acid has neutralized approximately half of the HC03,
and now _____________are in roughly equal concentrations. |
|
|
second buffer region of the titration curve.
|
In region IV the flat
region is the? |
|
|
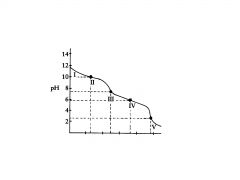
H2C03
|
In region V. the
equivalence point for the entire titration is reached, as all of the HCO3-, is converted to?. |
|
|
equivalence point
|
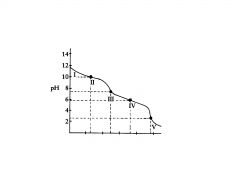
In region V. the
___________________for the entire titration is reached, as all of the HCO3-, is converted to H2C03. Again, a rapid change in pH is observed near the equivalence point as acid is added. |
|
|
Blood pH is maintained in a relatively small range (slightly
above 7) by a |
bicarbonate buffer
system. This homeostasis can be upset, leading to a condition known as acidosis. |
|
|
in a body you have the bicarbonate buffer
system. This homeostasis can be upset, leading to a condition known as ?. |
acidosis
|
|
|
What volume of a 3 M solution of NaOH is required to titrate 0.05 L
of a 4 M solution of HCl to the equivalence point? what equat should you use? |
NaVa=NbVb
|
|
|
At equilibrium, a certain acid, HA, in solution yields 0.94 M [HA] and
0.060 M [A-]. Calculate Ka. |
HA ---> H+ + A-
The molar ratio of A- to H+ is 1:1, so [H+] must also be 0.060 M at equilibrium. It follows, then, that: Ka [A-][H+]/[HA] = (0.060)(0.060)/(0.94) = 3.8 X 10-3 |
|
|
13. Which of the following combinations would produce a buffer solution
of pH = 4? (Ka HN02=4.5 X 10-4) A. 0.30 M HNO2, 0.22 M NaNO2 8. 0.22 M HNO2, 0.30 M NaNO2 C. 0.11 M HNO2, 0.50 M NaNO2 D. 0.50 M HNO2, 0.11 M NaNO2 |
pH = PKa + log [A-/[HA]
4 = 3.35 + log [A-]/[HA] 0.65 = log [A-]/[HA] [A-]/[HA] = 4.5 Only Choice C fulfills this criterion as 0.50/0.11 = 4.5. |

