![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
55 Cards in this Set
- Front
- Back
|
|
|
|
|
|

|
|
|

|

|
|

|
|
|

|
|
|
|

|
|

|

|
|
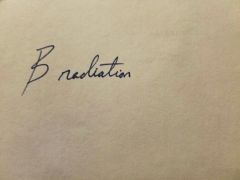
|
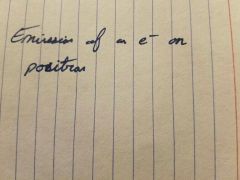
|
|

|
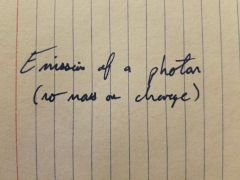
|
|

|

|
|
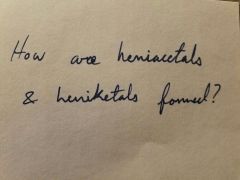
|
|
|
|

|
|
|

|
|
|

|
|
|

|
|
|
|
|
|
|
|
|

|
|
|

|
|

|

|
|

|

|
|
|

|
|

|
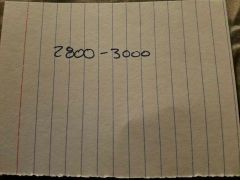
|
|

|

|
|
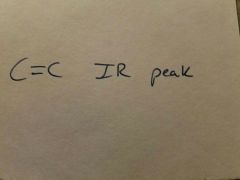
|
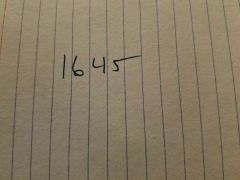
|
|
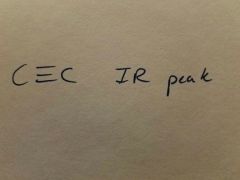
|
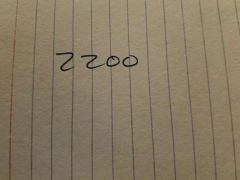
|
|
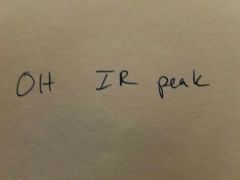
|
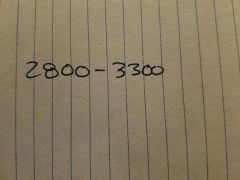
|
|
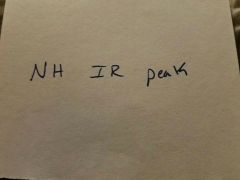
|

|
|

|

|
|
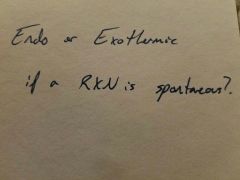
|

|
|

|

|
|
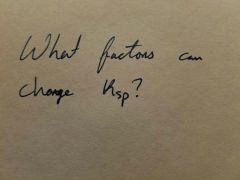
|

|
|

|

|
|
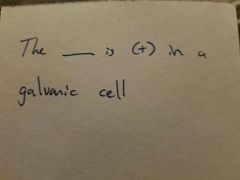
|

|
|

|

|
|
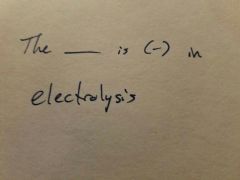
|

|
|
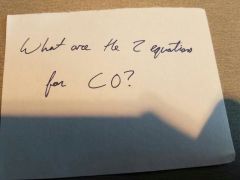
|
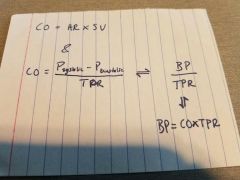
|
|

|
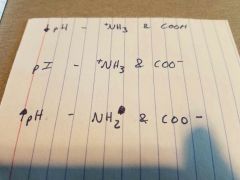
|
|

|

|
|

|

|
|

|

|
|
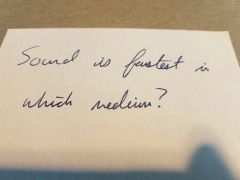
|

|
|

|
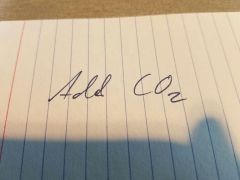
|
|

|

|
|
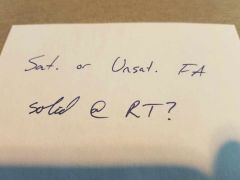
|

|
|

|

|
|
|
Steric Hinderance |
Slow RXN rate due to blockage of R groups |
|
|
R vs. S |
If 4 is in the back, R is CW, S is CCW If 4 is in the front, R is CCW, S is CW |
|
|
Deuterium |
2H - H thats 2x heavier |
|
|
What do enzymes stabilize? |
The transition state, which lowers the activation energy |
|
|
What do ideal gases assume? |
Individual volumes and IMFs are negligible |
|
|
What do lipases do? |
hydrolyze fatty acids |
|
|
How do you tell if entropy (S) is increased or decreased in a RXN? |
Look at the element state (liquid, gas, solid) in reactants vs. products |
|
|
When E^o is < 0, is the RXN spontaneous or non-spontaneous? |
Non-spontaneous |

