![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
391 Cards in this Set
- Front
- Back
|
How are dietary proteins broken down?
|

|
|
|
What aa residue of Ub does Ub attach on?
|
K48
|
|
|
What does E1 do?
|
adenylates Ub
|
|
|
What does E2 do?
|
carries Ub
|
|
|
What does E3 do?
|
transfers Ub from E2 to target protein
|
|
|
Velcade (bortezomib)
|
proteasome inhibitor for myeloma and lymphoma
|
|
|
What is the first step in amino acid degradation?
|
transamination
|
|
|
What is the second step in amino acid degradation?
|
oxidative deamination
|
|
|
What are AST and ALT used for?
|
aspartate aminotransferase and alanine aminotransferase
used for heart attack diagnosis |
|
|
What coenzyme do aminotransferases use?
|
pyridoxal phosphate
|
|
|
What coenzyme does H decarboxylase use?
|
pyridoxal phosphate
|
|
|
What coenzyme does glycogen phosphorylase use?
|
pyridoxal phosphate
|
|
|
What is frequently produced in transamination reactions?
|
E from alpha-ketoglutarate
|
|
|
What determines if an amino acid is non-essential?
|
If the alpha keto acid derivative can be synthesized
|
|
|
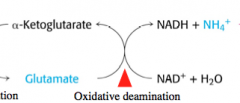
What happens to the N transferred to the alpha ketoglutarate?
|
glutamate dehydrogenase uses NADPH to release ammonium
|
|
|
What regulates glutamate dehydrogenase?
|
GTP inhibits formation of TCA intermediates
ADP activates |
|
|
Where does the glutamate dehydrogenase reaction work?
|
liver
|
|
|
What does the alanine cycle do?
|
Carries nitrogen from extrahepatic E to the liver where it is deaminated by alpha ketoglutarate to allow urea synthesis
|
|
|
What does glutamine synthase do?
|
adds ammonium to E using ATP so N can be carried through the blood
|
|
|
What is a major source of ammonia?
|
AMP -> IMP using adenylate deaminase
|
|
|
What is the most common cause of metabolic myopathy?
|
adenylate deaminase deficiency
|
|
|
What does adenosine deaminase deficiency cause?
|
severe combined immunodeficiency
|
|
|
What 3 amino acids are important to ammonia conversion?
|
A, E, Q
|
|
|
What happens to A as it arrives to the liver?
|
deaminated by A aminotransferase and the resulting glutamate is deaminated to produce ammonium
|
|
|
What happens to Q as it arrives to the liver?
|
deaminated by glutaminase to produce E which is deaminated to produce more ammonium
|
|
|
Where does the urea cycle occur?
|
liver
|
|
|
What is the first step in the urea cycle?
|
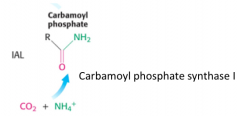
in mitochondria
uses 2 ATP |
|
|
How does carbamoyl phosphate exit the mitochondria?
|
combines with an ornithine via ornithine transcarbamoylase that enters the mt to form citrulline, which exits the mt
|
|
|
What do citrulline and D form?
|
argininosuccinate via argininosuccinate synthase
uses ATP |
|
|
What does argininosuccinase do?
|
cleaves fumarate from argininosuccinate to produce R
|
|
|
What does arginase do?
|
Cleaves urea from R to produce ornithine
|
|
|
What enters the urea cycle?
|
ammonia and aspartate
|
|
|
What exits the urea cycle?
|
fumarate and urea
|
|
|
How are the urea and TCA cycles linked?
|
argininosuccinase produces fumarate
|
|
|
What does glutamate produce in the brain?
|
decarboxylated to GABA
|
|
|
How does hyperammonemia affect glutamine?
|
increase
|
|
|
How does high ammonia affect pyrimidine levels?
|
increase
|
|
|
What is the most common urea cycle deficiency?
|
ornithine transcarbamoylase
x-linked |
|
|
What happens with argininosuccinate synthetase deficiency?
|
causes citrulline buildup, need R supplementation
|
|
|
What happens in argininosuccinase deficiency?
|
argininosuccinate buildup, need R supplementation
|
|
|
What happens in arginase deficiency?
|
CNS disorder
R exclusion diet |
|
|
Which 2 amino acids are solely ketogenic?
|
L K
|
|
|
Which 4 amino acids are keto and glucogenic?
|
FYIW
|
|
|
Which amino acids are essential?
|
MLK HIV WTF
|
|
|
What is the recommended daily allowance of protein?
|
50 g
|
|
|
What causes positive nitrogen balance?
|
growth, pregnancy
|
|
|
How is S synthesized?
|
3-PG (glycolytic intermediate) is oxidized by NAD+, aminated by glutamate, then dephosphorylated
|
|
|
How is G synthesized?
|
S -> G by serine hydroxymethyltransferase
produces 5-10-methylene-THF |
|
|
What cofactor does serine hydroxymethyltransferase use?
|
THF
|
|
|
What disorders is folate deficiency associated with?
|
megaloblastic and pernicious anemia, neural tube defects, heart disease, cancer
|
|
|
How much folate do you need per day?
|
400 micrograms
|
|
|
What is the main biochemical function of folate?
|
one C transfer
|
|
|
What processes is folate necessary for?
|
nucleotide biosynthesis
S, G, H biosynthesis methionine regeneration |
|
|
Which atoms of THF are reactive?
|
N5 and N10
|
|
|
What 5 one C units are transferred by THF?
|
methyl, methylene, formyl, formimino, methenyl
|
|
|
sulfanilamide
|
antibiotic against folic acid synthesis enzyme
antimetabolite of PABA |
|
|
What is the role of polyglutamylation of folate?
|
folate polyglutamates are retained in the cell
|
|
|
What is the active form of folate?
|
THF
|
|
|
What are the 3 major single C donors to folate?
|
G, S, H
|
|
|
What is the main route of G metabolism?
|
THF -> 5,10-methylene-THF
|
|
|
How many molecules of 5,10-methylene-THF are formed from serine?
|
2
|
|
|
What is methyl-THF used for?
|
methylate homocysteine to methionine
uses B12 |
|
|
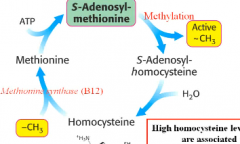
Why is methionine regeneration necessary?
|
for SAM production, which is involved in methylation
|
|
|
Rank the contribution of amino acids into the folate pool
|
S > G >> H
|
|
|
What does DHFR do?
|
NADPH-mediated reduction of folate to THF
catalyzes twice |
|
|
aminopterin and amethopterin
|
methotrexate
inhibit DHFR |
|
|
raltitrexed
|
inhibitor of thimidylate synthase
|
|
|
pemetrexed
|
inhibits thimidylate synthase, DHFR, GARFT (de novo purine biosynthesis)
|
|
|
What does thimidylate synthase do?
|
methylates dUMP to dTMP
|
|

name these:
|
uracil thymine
|
|
|
5-FU
|
suicide inhibitor of thymidylate synthase
|
|
|
How is SAM synthesized?
|
ATP is hydrolyzed to bind methionine + adenosine
|
|
|
What does DNA methylation do?
|
Protects it from restriction enzymes and silences gene tx
|
|
|
What catalyzes methionine regeneration?
|
methionine synthase
|
|
|
How is cysteine produced?
|
homocysteine + serine by cystathionine beta-synthase
|
|
|
What cofactor does cystathionine beta-synthase use?
|
PLP
|
|
|
What 3 enzyme deficiencies cause homocystinuria?
|
cystathionine synthase, methionine synthase, methylene-THF reductase
|
|
|
How does folate deficiency cause ds DNA breaks?
|
uridylate is misincorporated into DNA instead of thymidylate
uracil is excised out if two are excised close too each other |
|
|
What intermediate is formed when H donates a C to THF?
|
forminino-THF
|
|
|
formaldehyde + THF
|
methylene-THF
|
|
|
What does homocystinuria cause?
|
mental retardation
|
|
|
Hyperhomocysteinemia causes what disease?
|
vascular
|
|
|
Folate trap
|
When cobalamine is deficient, methionine synthase cannot use methyl-THF so all the folate funnels to it.
|
|
|
What 2 symptoms make up pernicious anemia?
|
CNS defecit
megaloblastic anemia |
|
|
What causes pernicious anemia?
|
cobalamine deficiency
|
|
|
Which symptoms of cobalamine deficiency can be fixed with folate supplementation?
|
megaloblastic anemia
|
|
|
How is histamine formed?
|
H reacted with H decarboxylase
|
|
|
What cofactor does H decarboxylase use?
|
pyridoxal phosphate
|
|
|
What do histamines do in the stomach?
|
secrete stomach acid
|
|
|
cimetidine
|
histamine receptor antagonist
|
|
|
PKU
|
phenylalanine hydroxylase deficiency
|
|
|
What is the coreductant for F oxidation to Y?
|
tetrahydrobiopterin
|
|
|
Albinism
|
Lacking melanin pigments, synthesized from Y
|
|
|
How is NO produced?
|
R -> citrulline using NO synthase, O2 and NADPH
|
|
|
Maple syrup urine disease
|
oxidative decarbox of alpha-ketoacids from V, I, and L are blocked
|
|
|
Salvage pathway for nucleotides
|
base attached to activated ribose (PRPP)
|
|
|
PRPP synthetase
|
catalyzes ribose 5 P activation using a pyrophosphate from ATP
|
|
|
What is the committed step in de nove purine biosynthesis?
|
PRPP amination to 5-phosphoribosylamine by Q phosphoribosyl amidotransferase
|
|
|
What molecules are used in de novo purine biosynthesis?
|
D, CO2, G, 2x Q, ribose-P, 10-formyl-THF
|
|
|
What is the initial product of de novo purine biosynthesis?
|
IMP
|
|
|
What inhibits the first reaction of de novo purine biosynthesis?
|
pemetrexed
|
|
|
What high energy substrate is used to catalyze IMP -> AMP?
|
GTP
|
|
|
How many enzymes in IMP synthesis have folate derivatives?
|
2 (10-formyl-THF)
|
|
|
What 2 precursors contribute C atoms to pyrimidine biosynthesis?
|
carbamoyl phosphate, aspartate
|
|
|
What enzyme catalyzes the first step in de novo pyrimidine biosynthesis?
|
carbamoyl phosphate synthetase II
|
|
|
What are the main subunits of cabamoyl phosphate synthetase?
|
glutamine hydrolysis to produce ammonia
bicarbonate phosphorylation to produce caboxyphosphate carbamic acid phosphorylation to product carbamoyl phosphate |
|
|
What make up CAD?
|
CPSII, aspartate transcarbamoylase, dihydroorotase
|
|
|
Know generally the steps from carbamoyl phosphate to orotate
|
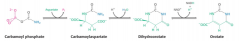
|
|
|
leflunomide
|
inhibits de novo synthesis of pyrimidine nucleotides at dihydroorotate dehydrogenase
|
|
|
What is PRPP?
|
activated ribose that can accept nucleotide bases
|
|
|
orotate + PRPP =
|
orotidylate
|
|
|
What makes up UMP synthase?
|
orotate phophoribosyltransferase and orotidylate decarboxylase
|
|
|
hereditary orotic aciduria
|
anemia, growth retardation
deficient UMP synthase treat with exogenous uridine |
|
|
What inhibits CPSII?
|
UTP
|
|
|
What converts nucleoside monophosphates to diphosphates?
|
specific nucleoside monophosphate kinases
|
|
|
How to get CTP from UTP?
|
amination using Q and ATP
|
|
|
thymidylate synthase
|
one C transfer from 5,10-methylenetetrahydrofolate to dUMP, results in DHF, NADPH mediated
|
|
|
How to go from UMP to dUMP?
|
ribonucleotide reductase
|
|
|
What inhibits thymidylate synthase?
|
5 fluorouracil via suicide inhibition
|
|
|
What is the in vivo version of fuorouracil?
|
fluorodeoxyuridylate
|
|
|
What enzyme is important in salvage pathway of nucleotide biosynthesis?
|
phosphoribosyltransferase
|
|
|
dihydroorotate dehydrogenase
|
dihydroorotate to orotate
|
|
|
hypoxanthine-guanine phosphoribosyltransferase
|
HGPRTase can act on guanine or hypoxanthine to form GMP/IMP
|
|
|
What do salvage reactions do?
|
Form nucleotides from free bases
|
|
|
Erythrocytes salvage which pyrimidines?
|
orotate, uracil, thymine but not cytosine
|
|
|
HGPRT deficiency
|
Lesch-Nyhan syndrome
compulsive self injury, mental deficiency hyperuricemia and high PRPP |
|
|
What are NAD+, FAD derivatives of?
|
ATP
|
|
|
pellagra
|
low nicotinate and W
dermatitis, dementia, diarrhea |
|
|
What is the common feature of NAD, FAD, CoA biosynthesis?
|
AMP transfer
|
|
|
What is the substrate for ribonucleotide reductase?
|
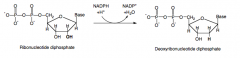
|
|
|
What catalyzes the committed step in purine nucleotide biosynthesis?
|
glutamine phosphoribosyl amidotransferase catalyzes conversion of PRPP into phosphoribosylamine
|
|
|
What inhibits glutamine phosphoribosyl amidotransferase?
|
AMP, GMP, IMP
|
|
|
Where does AMP/GMP inhibit the de novo pathway?
|
PRPP amidotransferase
|
|
|
Describe regulation after inosinate production?
|
Inosinate can become either AMP or GMP, both of which are feedback inhibited
|
|
|
What inhibits ribonucleotide reductase?
|
dATP by allostery, ATP reverses the inhibition
|
|
|
What are the regulatory steps in pyrimidine synthesis?
|
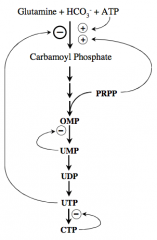
|
|
|
nuclease
|
nucleic acid to nucleotide
|
|
|
How is AMP degaded?
|
AMP -> adenosine deaminated by adenosine deaminase to inosine -> lose ribose to hypoxanthine -> oxidized by xanthine oxidase to xanthine -> oxidized again to uric acid/urate
|
|
|
What is the benefit of having high urate?
|
ROS scavenging
|
|
|
What does adenosine deaminase deficiency cause?
|
severe combined immunodeficiency
|
|
|
gout
|
urate deposition disease
sodium urate crystals form in joints and cause arthritis |
|
|
allopurinol
|
inhibits xanthine oxidase to decrease urate
|
|
|
How to differentiate between hyperuricemia from overproduction of purines or excessive cell death/kidney disease?
|
by radiolabeling protein synthesis and seeing if new urate has the radiolabel
|
|
|
What is a positive effector for glutamine-PRPP amidotransferase?
|
PRPP
|
|
|
What biochemical effect does decreased HGPRTase have?
|
increased intracellular PRPP, decreased IMP/GMP, allows hypoxanthine/xanthine to be scavenged to IMP/XMP
|
|
|
What are pyrimidines broken down to?
|
beta amino acids, ammonium
|
|
|
What process produces beta-aminoisobutyrate?
|
thymine degradation
|
|
|
What patients have high beta-aminoisobutyrate?
|
cancer patients undergoing chemotherapy
|
|
|
What are the two alcohol metabolizing systems?
|
liver alcohol dehydrogenase
liver microsomal ethanol oxidizing systems |
|
|
What are some acute effects of alcohol?
|
hypoglycemia
ketoacidosis lacticacidosis |
|
|
What are some long term effects of alcohol?
|
liver disease
acetaldehyde adducts free radical damage |
|
|
What other alcohols are metabolized?
|
methanol
ethylene glycol |
|
|
What is ethylene glycol in?
|
antifreeze
brake fluid |
|
|
What happens when you ingest ethylene glycol?
|
acidosis
kidney damage |
|
|
Where is ethanol metabolized?
|
<5% in upper GI
90% liver up to 10% excreted by lungs/kidney |
|
|
What is the toxic intermediate of ethanol metabolism?
|
acetaldehyde
|
|
|
Draw ethanol metabolism from ethanol to CO2:
|
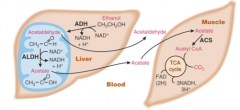
|
|
|
What coenzyme is used by alcohol dehydrogenase?
|
NAD+
|
|
|
How is acetaldehyde metabolized?
|
acetaldehyde dehydrogenase takes it to acetic acid
|
|
|
How do the two ALDH isoforms differ?
|
ALDH2 is mitochondrial and catalyzes 80% of acetaldehyde oxidation
ALDH1 is cytosolic and oxidizes multiple aldehydes |
|
|
What is significant about the ADH2*2 allele?
|
ADH catalyzes ethanol too quickly to acetaldehyde that ALDH cannot keep up and acetaldehyde build up.
|
|
|
Where are Class I ADH expressed?
|
liver
|
|
|
Where are Class IV ADH expressed?
|
upper GI tract
|
|
|
What happens with excess acetaldehyde?
|
nausea and vomiting
|
|
|
What is significant about ALDH2*2?
|
ALDH has little activity
acetaldehyde accumulates |
|
|
Disulfiram
|
ALDH inhibitor
|
|
|
What happens to the acetate that is produced from ALDH?
|
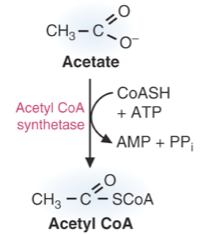
|
|
|
Where is MEOS found?
|
microsomal ethanol oxidation system
liver |
|
|
What is the main enzymes in MEOS?
|
CYP450 enzyme CYP2E1
|
|
|
When is MEOS used?
|
at high alcohol concentration
|
|
|
What is the metabolic flaw of MEOS?
|
produces acetaldehyde too fast
byproduct of free radicals uses up NADPH which is needed to regenerate reduced glutathione |
|
|
Why is taking acetaminophen dangerous for alcoholics?
|
CYP2E1 converts acetaminophen to NAPQI an unstable oxidant
|
|
|
Why is taking phenobarbital and ethanol dangerous?
|
Phenobarbitals induce CYP2B1/2, which metabolize phenobarbital, but ethanol inhibits these enzymes and phenobarbital can accumulate to toxic levels.
|
|
|
What is the energy yield for ethanol using ADH?
|
+5 from 2x NADH produced in etOH -> acetate
-2 from acetate -> acetyl CoA +10 from acetyl CoA net +13 |
|
|
What is the energy yield of etOH using MEOS?
|
-2.5 from NADPH use of CYP2E1
+2.5 from acetaldehyde -> acetate -2 from acetate -> acetyl CoA +10 from acetyl CoA net +8 |
|
|
empty calories
|
metabolic energy with few nutrients
like liquor, processed foods |
|
|
How does alcoholism cause vitamin deficiency?
|
alcohol damages cells lining GI system
damages liver and its ability to store nutrients especially thiamine (B1), riboflavin (B2), pyidoxine (B6), ascorbic acid, folic acid |
|
|
What does alcohol-related thiamine deficiency cause?
|
Wernicke-Korsakoff syndrome
encephalopathy psychosis |
|
|
What does thamine do?
|
vit B1 -> TPP
cofactorfor: PDH, alpha KG DH, branched chain alpha-ketoacid DH, transketolase |
|
|
List 6 reversible metabolic effects of etOH:
|
inhibition of FA oxidation
activation of TAG synthesis fatty liver ketoacidosis lactic acidosis hypoglycemia |
|
|
List 7 irreversible effects of etOH:
|
acetaldehyde adducts
free radicals inflamed liver with cell death disrupted blood flow cirrhosis loss of liver function hepatic failure |
|
|
What %age of autopsies have alcohol-induced cirrhosis?
|
10%
|
|
|
What is the peak incidence age of etOH toxicity?
|
40-55
|
|
|
What 3 forms of liver disease occur from alcohol?
|
fatty liver
hepatitis cirrhosis |
|
|
What is the main metabolic side effect of etOH metabolism?
|
increased NADH/NAD+ ratio
|
|
|
What consequences does high NADH/NAD+ have?
|
inhibits FA oxidation
inhibits TCA cycle FA synthesis ketoacidosis lactic acidosis low GNG due to blocking pyruvate formation from alanine, lactate |
|
|
What does acetaldehyde do in the liver?
|
- prevents protein secretion from by forming adducts with proteins and microtubules
- binds reduced glutathione - lipid peroxidation |
|
|
How does ROS affect acetaldehyde?
|
ROS inhibits eTC and limits NAD+ formation, to increase acetaldehyde
|
|
|
What hepatic enz are measured in the serum to detect hepatocyte damage?
|
A aminotransferase ALT
D aminotransfer AST |
|
|
What is the main problem following liver acetaldehyde adduct formation?
|
The products are denatured and cannot be secreted, causing osmotic imbalance and subsequent swelling of the liver, causing portal hypertension and esophageal verices.
|
|
|
What is the main source of free radicals in etOH metabolism?
|
CYP2E1
|
|
|
Why is CYP2E1 a good source for free radicals?
|
leakage from FMN and heme
|
|
|
What free radicals does CYP2E1 produce?
|
hydoxyethyl radical, superoxide
|
|
|
What is the main target of free radicals?
|
membrane lipids, especially on the inner mitochondrial membrane
|
|
|
cardiomyopathy from alcohol
|
acetaldehyde and malondialdehyde adducts target heart proteins and weaken it, highly dependent on energy production
|
|
|
What are the products of meOH metabolism?
|

|
|
|
What are the products of ethylene glycol metabolism?
|

|
|
|
Why is ethylene glycol metabolism so toxic?
|
It produces intermediates with 2 hydroxyls
acidemia oxalate crystals -> kidney damage |
|
|
4-MP
|
fomepizole, antizol
inhibitor of ADH |
|
|
Why is oxalic acid dangerous?
|
Calcium oxlate can precipitate in kidneys and cause kidney stones
Oxalate chelates calcium |
|
|
Why is it important to screen kidney stones for calcium oxalate?
|
some people need to avoid oxalate foods because of its kidney stone forming tendencies
|
|
|
Why doesn't muscle glycogen buffer blood glc?
|
muscles lack glc 6 phosphatase
|
|
|
When is glycogen especially important to cardiac muscle?
|
myocardial infarction
|
|
|
What organ has the highest concentration of glycogen?
|
liver
|
|
|
What tissue has the highest mass of glycogen?
|
skeletal muscle
|
|
|
How are glc molecues attached in glycogen?
|
alpha 1,4 in straight chain
alpha 1,6 in branch |
|
|
Where does glycogen breakdown or synthesis begin?
|
non-reducing end
|
|
|
What is the purpose of glycogen branching?
|
Increase solubility, allow for multiple glycogen breakdown sites
|
|
|
How many glc residues b/w branches?
|
10
|
|
|
What is the advantage to high MW glycogen?
|
Doesn't contribute to osmtoic pressure
|
|
|
What enzyme catalyzes glycogen breakdown?
|
glycogen phosphorylase
|
|
|
What enzyme catalyzes glycogen synthesis?
|
glycogen synthase
|
|
|
Draw glycogenesis:
|
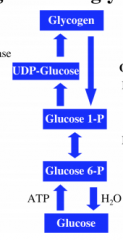
|
|
|
What transports glc into liver? muscle?
|
liver - GLUT2
muscle - GLUT4 |
|
|
What enzyme interconverts glc 1 P and glc 6 P?
|
phosphoglucomutase
|
|
|
What is the predominant product of glc6P in muscle?
|
lactate
|
|
|
gluco/hexokinase?
|
Glucokinase is found in the liver and beta cells and has a lower affinity for glc than hexokinase. Glc6P inhibits hexokinase.
|
|
|
MODY
|
maturity onset diabetes in the young
glucokinase disorder inadequate insulin secretion upon glc stimulation |
|
|
What is the first step in glycogenesis?
|
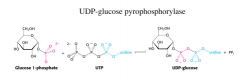
glc must be activated as UDP glc
|
|
|
What enzyme catalyzes the elongation of glycogen chains?
|

|
|
|
Where is ATP used in glycogen synthesis?
|
Converting UDP -> UTP to produce UDP-glc
|
|
|
glycogenin
|
primer for glycogen synthesis
once enough glc's are added, it becomes cleaved off to start anew |
|
|
What is the minimum glc residues inb/w branches?
|
4
|
|
|
What is the mechanism of glycogen branching?
|
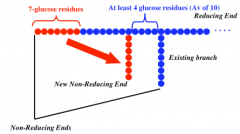
Once a 7 glc residue is free, a branching enzyme moves it to an alpha 1,6 spot
|
|
|
What effect does insulin have on glycogenesis?
|
increase
|
|
|
What effect do glucocorticoids have on glycogenesis?
|
decrease
|
|
|
lysosomal alpha 1,4-glucosidase
|
small quantity of glycogen is broken down by lysosomes
|
|
|
How does glycogen debranching work?
|
phosphorylase cleaves glc no closer than 4 glc from a branch
debranching enzyme (alpha 1,6 glucosidase) moves 3 glc units and hydrolyzes 1 glc |
|
|
What is the normal glc1P:glc ratio?
|
10:1 because there are usually 10 glc b/w branches and nonP glc is produced from branch points
|
|
|
How to go from glc1P to glc?
|
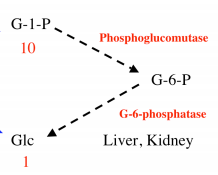
|
|
|
What effect do glucagon and epinephrine have on glycogen metabolism?
|
GPCR/cAMP/PKA in liver to activate glycogenolysis by phosphorylase a and inhibiting glycogen synthase b. It also inhibits protein phosphatase 1 which reveres PKA.
|
|
|
What activates protein phosphatase 1?
|
Insulin
|
|
|
Is dephosphorylated glycogen synthase b active or inactive?
|
inactive
|
|
|
How does activated protein phosphatase 1 affect glycogen synthase b and phosphorylase b?
|
dephosphorylate both of them to activate GS and deactivate phosphorylase
|
|
|
How do glucocorticoids affect glycogen metabolism?
|
induce glycogen breakdown
|
|
|
What are downstream effects of glucocorticoids?
|
induced: GNG, glycogen synthase, proteolysis by Ub
repressed: glycolysis, glucokinase, glycogen phosphorylase |
|
|
How is phosphorylase kinase activated?
|
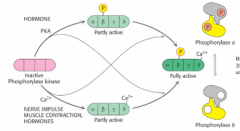
hormones/PKA or Ca activate glycogenolysis
|
|
|
How does glc affect phosphorylase in hepatocytes?
|
allosteric inhibition
|
|
|
phosphorylase a/b, which is active?
|
a
|
|
|
von Gierke disease, type I
|
glc-6-phosphatase deficiency
liver is overloaded with glycogen hyperlipidemia, lactic acidosis |
|
|
Pompe disease, type II
|
lysosomal alpha 1,4-glucosidase deficiency
glycogen accumulation in lysosomes |
|
|
Cori disease, type III
|
debranching enzyme deficiency
hypoglycemia due to inefficient glycogen usage affects muscle, liver |
|
|
Andersen disease, type IV
|
branching enzyme deficiency
affects liver long glycogen branches cause inefficient storage and usage of glycogen |
|
|
What are the basic functions of the pentose phosphate pathway?
|
glc oxidation to produce NADPH
produce ribose 5 P interconvert pentose sugars |
|
|
What is NADPH used for?
|
Reductive biosynthesis of FA, nt's, NTS, cholesterol
Detox - regenerating glutathione and CPY450 |
|
|
What does ribose 5 P produce?
|
nucleic acids, ATP, NADH, FAD, CoA
|
|
|
What do the two phases of the pentose phosphate pathway do?
|
oxidative starts with glc6P to produce NADPH
the nonoxidative phase interconverts different sugars |
|
|
How many NADPH per glc 6 P?
|
2
|
|
|
What is the first rxn in the pentose phosphate pathway?
|
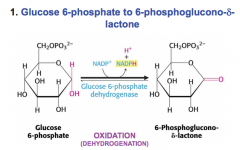
NADPH producing
|
|
|
Does glc 6 P dehydrogenase ever reduce NAD+?
|
no
|
|
|
What happens to 6-P glucono-delta-lactone?
|
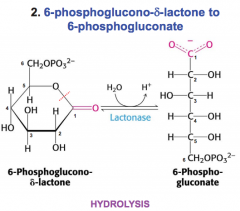
hydrolyzed by lactonase
|
|
|
What happens to 6 P gluconate?
|
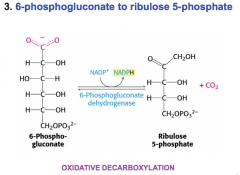
oxidative decarboxylation to produce more NADPH
|
|
|
What is the key intermediate in the pentose phosphate pathway?
|
ribulose 5 P
|
|
|
In order to produce nucleotides, what intermediate does ribulose 5 P convert to?
|
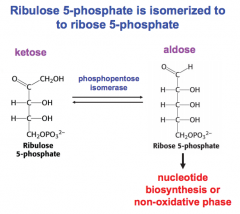
ribose 5 P
|
|
|
What is ribulose 5 P epimerized to?
|
xylulose 5 P
|
|
|
Fundamentally, what does transketolase do?
|
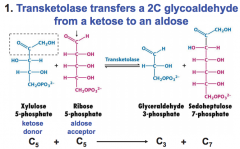
transfers 2C glycoaldehyde from a ketose to an aldose
|
|
|
What coenzyme does transketolase use?
|
TPP
|
|
|
What intermediates do xylulose 5 P and ribose 5 P produce?
|
glyceraldehyde 3 P and sedoheptulose 7 P
|
|
|
What enzymes use TPP?
|
transketolase
PDH alpha KG DH |
|
|
Fundamentally, what does transaldolase do?
|
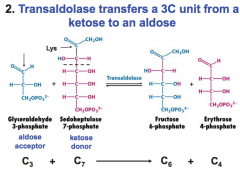
transfers 3C unit from ketose to aldose
|
|
|
What amino acid residue is involved in the transaldolase mechanism?
|
K
|
|
|
What two intermediates can ribulose 5 P go to?
|
ribose 5 P or xylulose 5 P
|
|
|
What do erythrose 4 P and xylulose 5 P produce?
|
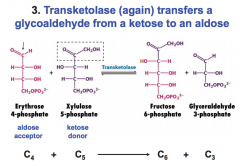
fructose 6 P and glyceraldehyde 3 P
|
|
|
What is the net product of the nonoxidative phase?
|
in: ribose 5 P, 2x xylulose 5 P
out: 2x fructose 6 P, glyceraldehyde 3 P |
|
|
How to diagnose thiamine deficiency?
|
Measure RBC transketolase activity +/- thiamine, should be no change if the diet is adequate in thiamine
|
|
|
What disease does thiamine deficiency cause?
|
beri-beri
|
|
|
What step of the pentose phosphate pathway is controlled? What controls it?
|
glc 6 p dehydrogenase
NADP+ availability, NADPH can outcompete it |
|
|
What forms of glc 6 p dehydrogenase are active?
|
dimer and tetramer
monomer is inactive |
|
|
What does NADP+ binding to glc 6 p dehydrogenase do?
|
dimer and tetramer formation
|
|
|
What are the four pathways glc 6 p can go in?
|
glycolysis
pentose phosphate pathway glycogenesis blood glc (liver) |
|
|
When is ribose 5 p needed more than NADPH?
|
during DNA replication (G1 phase)
|
|
|
What happens to glc 6 p during G1?
|
glycolysis because DNA replication demands more ribose 5 p
|
|
|
When are both ribose 5 p and NADPH needed?
|
Concurrent nt and FA synthesis in mitotic cells
|
|
|
What is the fate of glc 6 p during nt and FA synthesis demand?
|
Just the oxidative phase produces 2x NADPH and ribose 5 P
|
|
|
When do you require NADPH but not ribose 5 p?
|
detox, free radical crisis
|
|
|
What happens to glc 6 p during free radical crisis?
|
Both the oxidative and nonoxidative pathways are on so NADPH can keep being reproduced alongside glycolysis and GNG
|
|
|
When is NADPH and ATP required?
|
cholesterol or FA synthesis
|
|
|
What is the fate of glc 6 p during cholesterol synthesis?
|
oxidative and nonoxidative ppp, along with glycolysis to produce pyruvate/ATP
|
|
|
What is reduced glutathione used for?
|
defense against oxidative stress
electron donor maintains reducing conditions in rbc |
|
|
How much reduced glutathione exists in animal cells?
|
5 mM
|
|
|
What is the ratio of reduced:oxidized glutathione?
|
500:1
|
|
|
What is the reactive part of glutathione?
|
sulphydryl group
|
|
|
What is oxidized glutathione?
|
2 glutathione molecules that are connected via disulfide bond
|
|
|
What is the molecular mechanism of glutathione reduction by glutathione reductase?
|
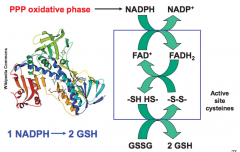
NADPH electrons are transfered to FAD+ to cysteines to glutathione
|
|
|
What cells really need reduced glutathione?
|
RBC
maintain reduced Hb and ferrous Fe |
|
|
What does defective pentose phosphate pathway cause in rbc's?
|
denatured Hb in rbc (Heinz bodies)
|
|
|
What do glutathione peroxidases do?
|
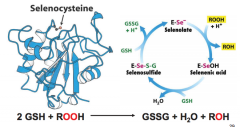
reduces lipid hydroperoxides to alcohols
reduces hydrogen peroxide to water |
|
|
What is the active residue in glutathione peroxidase?
|
selenocysteine
|
|
|
What does misfolding cause glutathione peroxidases to do?
|
aggregate via disulfide bond formation
|
|
|
How do Heinz bodies affect rbc's?
|
Increase likelihood of lysis
|
|
|
list 4 causes of haemolytic anemia
|
rbc membrane defects (hereditary spherocytosis)
abnormal Hb (thalassemia) abnormal glycolytic enzymes including glc 6 p DH (non-spherocytic hemolytic anemia) oxidative stress |
|
|
How common is glc 6 p DH mutation?
|
7%, commonly in Africa, South Asia, and Mediterranean
|
|
|
Why is glc 6 p DH good?
|
protects against malaria by Plasmodium falciparum
|
|
|
What does glc 6 p DH cause?
|
hemolytic anemia
|
|
|
What induces hemolytic anemia in glc 6 p DH mutant patients?
|
antimalarial drugs, sulfa drugs, fava beans, infections
|
|
|
What does a Beutler fluorescent test measure?
|
Detects deficiency in glc 6 p DH via rbc NADPH production by UV (365 nm)
|
|
|
What patients produce a false positive Beutler fluorescent test?
|
Actively hemolytic patients produce high fluorescence even though they are deficient
|
|
|
What causes MetHb?
|
antimalarial drugs - pamaquine, primaquine
antibiotics - trimethoprim, sulphonamides, dapsone |
|
|
What is MetHb?
|
Fe 3+ oxidized Hb
|
|
|
How do fava beans cause hemolytic anemia?
|
Produces vicine which damages rbc membranes and produces peroxide
|
|
|
What treatments are used for glc 6 p DH deficiency?
|
vaccination to reduce infections
self limiting avoiding certain drugs blood transfusions splenectomy/bone marrow transplant for extreme cases |
|
|
Why does glc 6 p DH cause hemolytic anemia?
|
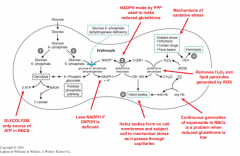
less NADPH to regenerate reduced glutathione
Heinz bodies form on rbc membranes continued generation of superoxides and lipid peroxides |
|
|
What is another name for tylenol/acetaminophen?
|
Paracetamol
|
|
|
What is the most common cause of acute liver toxicity?
|
tylenol overdose
|
|
|
What is produced in excess during tylenol overdose?
|
NABQI -> free radical damage to liver
|
|
|
How to treat acetaminophen overdose?
|
N-acetylcysteine to restore reduced glutathione
|
|
|
How is acetaminophen metabolized?
|
glucuronidation and sulfation produce excretable products
CYP-mediated hydroxylation produces toxic NAPQI which conjugates with nucleic acids, proteins, and lipid membranes |
|
|
What is the basic structure in porphyrins?
|
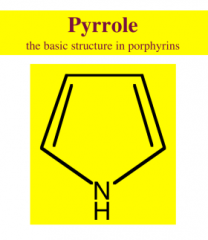
pyrrole
|
|
|
What is bilirubin broken down to?
|
urobilins in urine
stercobilin in feces |
|
|
What is the major porphyrin in humans?
|
heme
|
|
|
What proteins use heme?
|
Mb, Hb, ER and mitochondrial cytochromes, catalase
|
|
|
What is a prosthetic group?
|
a tight-bound non-peptide component of proteins
|
|
|
What is the first tetrapyrrole ring that is formed in heme synthesis?
|
uroporphyrinogen III
|
|
|
What rxn is catalyzed by ALA synthase?
|
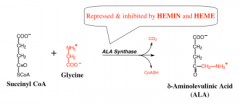
succinyl CoA and glycine decarboxylation to ALA
|
|
|
What coenzyme does ALA synthase need?
|
PLP
|
|
|
What is the rate-limiting step in porphyrin synthesis?
|
ALA synthase
|
|
|
What allosterically inhibits ALA synthase?
|
hemin and heme
|
|
|
What is hemin?
|
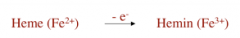
oxidized heme
|
|
|
Can hemin carry oxygen?
|
no
|
|
|
What vitamin is PLP derved from?
|
B6 (pyridoxine)
|
|
|
What are the major anaplerotic molecules for the TCA cycle?
|
glc, alanine, lactate, citrate, branched amino acids
|
|
|
How many succinyl CoA's are req'd for one heme?
|
8
|
|
|
What rxn is catalyzed by ALA dehydrase?
|
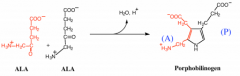
2 ALA molcules to porphobilinogen containing a pyrrole ring
|
|
|
What side chain is important for ALA dehydrase?
|
sulfhydryl
|
|
|
What metal is used by ALA dehydrase?
|
Zn
|
|
|
What inhibits ALA dehydrase?
|
Pb
|
|
|
How do ALA and porphobilinogen affect the brain?
|
neurotoxin
|
|
|
What rxn is catalyzed by porphobilinogen deaminase
|
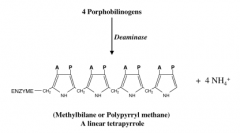
linear tetrapyrrole formation from 4 porphobilinogens to methylbilane or polypyrryl methane
|
|
|
How are the acetate and propionate side chains of porphobilinogen oriented?
|
symmetric
APAPAPAP |
|
|
What rxn is catalyzed by uroporphyrinogen synthase?
|
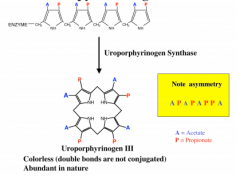
uroporphyrinogen III tetrapyrrole ring formation
|
|
|
What is the acetate and propionate orientation of uroporphyrinogen III?
|
asymmetry
APAPAPPA |
|
|
What is uroporphyrinogen I?
|
symmetric uroporphyrinogen produced spontaneously with a genetic defect in uroporphyrinogen synthase
|
|
|
What rxn is catalyzed by uroporphyrinogen decarboxylase?
|
decarboxylation of sidechains of uroporphyrinogen III to coproporphyrinogen III (acetyl -> methyl)
|
|
|
How is protoporphyrin IX synthesized?
|
Coproporphyrinogen III is propionyl side chain-decarboxylated to vinyl and ring-oxidated
|
|
|
What catalyzes the last rxn in heme synthesis?
|
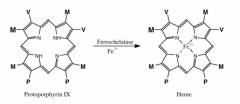
ferrochelatase
|
|
|
Ohter than heme, what is another metaloporphyrin?
|
cabalamin (B12)
|
|
|
What substitutions does active cobalamin have?
|
5'-deoxyadenosine or methyl
|
|
|
How is heme synthesis compartmentalized?
|
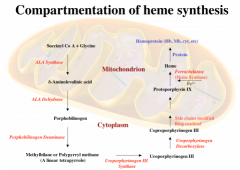
ALA leaves the mitochondria and coproporphyrinogen III enters back in
|
|
|
How do heme and hemin regulate heme synthesis?
|
ALA synthase precursos synthesis, transport into the mitochondria, and activity
|
|
|
What is one of the most difficult environmental problems to control?
|
lead poisoning
|
|
|
How does lead poisoning manifest itself in adults?
|
fatigue, abdominal pain, arthralgia
|
|
|
How does lead poisoning manifest itself in children?
|
encephalopathic crisis
|
|
|
What biochemical effects does lead poisoning have?
|
inhibits ALA dehydrase and ferrochelatase
forms zinc protoporphyrin |
|
|
What do most porphyrias share?
|
photosensitivity
|
|
|
Which porphyrias do not induce photosensitivity?
|
ALA dehydrase and AIP
|
|
|
What symptoms come with porphyrias?
|
psychiatric problems and abdominal pain
|
|
|
What causes AIP?
|
Partial loss (50%) of porphobilinogen deaminase
|
|
|
What is the problem in AIP?
|
Accumulation of ALA and porphobilinogen
|
|
|
What symptoms are seen in AIP?
|
ab pain
seizures agitation mental disturbance insomnia constipation tachycardia high BP parasthesia dark red urine |
|
|
What are porphyria patients often misdiagnosed with?
|
psychiatric disorder
|
|
|
How ti diagnose AIP?
|
elevated urine PBG and ALA
porphobilinogen deaminase in rbc's no photosensitivity porphyrins not elevated |
|
|
How do some drugs worsen AIP?
|
CYP450 inducing drugs drain the heme supply and build up ALA and PBG
|
|
|
How to treat AIP?
|
withdraw CYP450 inducing drugs
administer hematin to inhibit ALA synthase (hemin) carb rich diet inhibts ALA synthase |
|
|
What causes congenital erythropoietic porphyria?
|
Mutated uroporphyrinogen synthase
|
|
|
What is bad about congenital erythropoietic porphyria?
|
methylbilane -> uroporphyrinogen I and its metabolites accumulate
|
|
|
What symptoms come with congenital erythropoietic porphyria?
|
skin blisters
ulcerating vesicles scarring hemolytic anemia spleen enlargement red urine |
|
|
Where is heme catabolized?
|
spleen (70%)
liver bone marrow |
|
|
What is the first enzyme in heme catabolism?
|
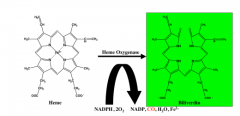
heme oxygenase
|
|
|
What byproducts does heme oxygenase produce?
|
iron, carbon monoxide, NADP+
|
|
|
Why is CO toxic?
|
binds to Hb and complex IV of eTC
|
|
|
What is the function of bilirubin?
|
antioxidant
While bound to albumin in blood each molecule can destroy two hydroperoxy radicals |
|
|
What is oxidized bilirubin?
|
biliverdin
|
|
|
How is biliverdin reduced back to bilirubin?
|
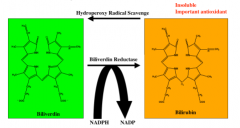
biliverdin reductase
|
|
|
What free radical-related processes depend on NADPH?
|
glutathion reduction
heme oxygenase biliverdin reductase |
|
|
What are the different colors of bruises?
|
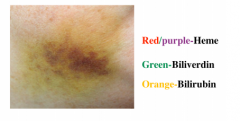
|
|
|
How is bilirubin made water soluble?
|
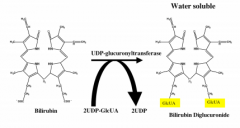
conjugation with 2x glucuronic acid on proprionate side chains to form bilirubin diglucuronide
|
|
|
Where does bilirubin conjugation take place?
|
liver
|
|
|
What happens to bilirubin in case of liver damage?
|
water insoluble bilirubin accumulates in blood and fatty tissue like subcutaneous fat
jaundice |
|
|
What are synonyms of soluble bilirubin?
|
bilirubin diglucuronide
conjugated bilirubin direct bilirubin |
|
|
What are synonyms of insoluble bilirubin?
|
bilirubin
unconjugated bilirubin indirect bilirubin |
|
|
How is bilirubin excreted?
|
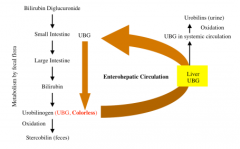
UBG -> oxidized in blood -> urobilins
UBG -> oxidized in large intestine -> stercobilin |
|
|
What are possible causes of yellow skin in jaundice?
|
beta-carotene
lycopene bilirubin |
|
|
What causes prehepatic jaundice?
|
excessive hemolysis
|
|
|
What causes hepatic jaundice?
|
defective transport
lack of UDP-glucuronyltransferase liver dysfunction |
|
|
What causes posthepatic jaundice?
|
biliary obstruction
|
|
|
Lactate threshold
|
Point at which blood lactate accumulates above resting levels
|
|
|
OBLA
|
onset of blood lactate accumulation, point at which exercise intensity is such that lactate cannot be cleared at the rate it is produced
|
|
|
What are mean energy req's?
|
40 kcal/kg body
|
|
|
What are the recommend macronutrient guidelines?
|
60% carb
15% protein 25% fat |
|
|
What is the max amount of protein that should be consumed?
|
No more than 1 g per lb body weight
|
|
|
How much Ca per day?
|
1200 - 1500 mg
|
|
|
How high can sweat rates be?
|
2 L / hr
|
|
|
What are the benefits to sodium for athletes?
|
maintains plasma osmolarity
reduces urine output induces thirst |
|
|
What is classic carb loading?
|
2 exhaustive sessions to blank glycogen
fat/protein diet for 3 d while training high carb diet for 3 d with rest |
|
|
What is modern carb loading?
|
regular diet with 75% VO2 max/90'
high carb diet with light exercise 2d high carb diet with 1d rest |
|
|
When does plasma caffeine peak?
|
40 - 60'
|
|
|
What is caffeine's half-life?
|
3 - 5 hr
|
|
|
What metabolizes caffeine?
|
liver
|
|
|
How quickly is caffeine absorbed?
|
90% within 20'
|
|
|
What effects does caffeine have?
|
mobilize intracellular Ca
augment catecholamine spare adenosine by blocking adenosine receptors enhance lipolysis stimulate contractility |
|
|
What adverse effects does caffeine cause?
|
restless
nervous insomnia tremors hyperesthesia diuresis anorexia |

