![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
89 Cards in this Set
- Front
- Back
|
GIT barriers |
pH bile prevents enveloped virues villi mononuclear cells survey gut (macro) M cells deliver antigens via transcytosis |
|
|
Norovirus characters, genome |
Caliciviridae ssRNA, non env single capsid prot |
|
|
Norovirus is easily transmissible |
contagious rapid and prolifically shed constant evolve limited immunity moderately virulent - maintain pool of susceptible hosts - symbiosis of gut flora may faciliate persistence |
|
|
Norovirus RdRp |
contributes to evolution error prone may change structure of VP1 recomb during coinfection for reassortment |
|
|
Norovirus Spread |
spread by contaminated food and water worldwide distribution |
|
|
MNV |
cannot culture human tissue, use a mouse model MNV
transient expression of CD300lf receptor may sustain infection |
|
|
Norovirus IR |
dsRNA > MDA5 on mito > IRF > T1 IFN T1 IFN > JAKSTAT path > ISG |
|
|
Rotavirus |
Reoviridae segmented dsRNA multilayer capsid
|
|
|
Rota VP |
VP4 - spiked for recepter interaction, cleaved by trypsan into VP5/8 to increase infectivity. VP4 (P sero) and 7 (G sero) elicit netralising ab VP6 - elicit ab inhibibting infection during IgA transcytosis |
|
|
Detect Rota |
ELISA w mab to det sero nested RTPCR to det geno EM seroconversion IgA conversion in stool |
|
|
Rota symptoms |
Severe dehydration due to blunting of villi, decrease absorbative capacity Base of villi contain IgA producing cells |
|
|
Rota diversity |
gene reassortment fro coinfection |
|
|
Rota infection cofacters |
cofacters = pH, ionic strength binding is low aff, high av binding to terminal SA = susceptible to sialidase. resistance by binding branched SA. VP8 structural change can change glycan class, |
|
|
Rota binding |
binds glycans and integrins
Upon binding, struct change to adapt to diff viral receptors |
|
|
Rotavirus and diabetes |
increase of ab ass with diabetes progression beta cells prod insulin in pancreas. virus taken up by DC, produce type 1 IFN and activates bystander autoreactive islet sp CD8 cells to kill the beta cells. |
|
|
Rota vaccine |
live vaccines atten through passage monovalent human strain bovine reassortment vaccine |
|
|
Bats |
only flying mammals variable thermoregulation low rate of tumorogenesis long life span |
|
|
Hendra |
paramyxo - ssRNA, non segmented encodes fusion and attachment prot, matrix, NC bat-horse-human, no HH |
|
|
Nipah |
related to hendra bat-pig-human - high PP Bangldesh has yearly emergence w no intermediate host and HH specific due to conserved surface recept |
|
|
SARS |
+RNA spike prot used for both fusion and att. human spike binds ACE2, bats doesnt MERS = Sars like, high mortality low transmission. uses diff receptor, true reservoir unknown |
|
|
Melaka |
reoviridae, dsRNA has envelope and fusogenic in human not bats
|
|
|
Why bats? |
flying = high metab = ox stress = efficient DNA repair systems = no cancer, harbour virus w/o disease high basal expression of IFNa MHC1 groove is varied, flexible binding No M2 macro different post-transl mod of NLRP3/caspase |
|
|
Epidemiology |
study of distribution and determinants of health related states or evens and its application to the control of diseases and other health problems |
|
|
Endemic |
present at all times at low frequency, ongoing age incidence is det by duration of immunity, Whooping cough - IR drops off w age |
|
|
Epidemic |
sudden severe outbreak of a region or group |
|
|
Pandemic |
widespread epidemic may be novel, highly susceptible, must be highly transmissable |
|
|
Cross over events |
spread of dieases from animals, ranked by degree of severity |
|
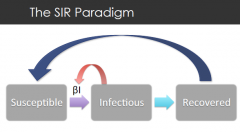
The SIR paradigm |
susceptible - infected - recovered recovery leads to removal of virus from pop rate of infection det by number of infected = the transmission parameters x prevalence |
|
|
Host determinants of susceptibility |
age immune status - naive, vaccinated, prior inf underlying risk pregnancy ethnicity |
|
|
Virus determinants of natural history |
latency infectiousness - sympt, duration, shed/spread induction of IR - temporary, strain sp |
|
|
Population determinants of spread |
birth rate household size crowding population density connectedness mobility |
|
|
Environmental risk factors |
season sanitation proximity to vector/reservoirs natural disaster |
|
|
Surveillance |
layers of severity = diff burdens for intervention year by year comparisons threshold detection algorithms serosurveillance to determine at risk |
|
|
Intervention |
non pharma pharma vaccines, antivirals mitigate severe outcomes by ID vulnerable groups to reduce risk of critical care requirements |
|
|
RNA structure |
complex, can self anneal and pair noncoding RNA contains sequenc sepcific structural info
dsRNA/shRNA initiate plants silencing, cleaved into siRNA pre miRNA initiates mammalian silencing |
|
|
Plants RNA silencing |
- long dsRNA from infection or exp intrinsically -processed by Dicer/RNAse3 into siRNA w overhangs - RISC produced targets complementary mRNA and cleaves into fragments |
|
|
siRNA |
short dsRNA important for genetic immunity 21nt, perfectly duplexed double helix with 3' overhangs |
|
|
Dicer |
processed long dsRNA, seperate strands guide strans is incorp into RISC, has less tightly paired 5' |
|
|
RISC |
RNA induced silencing complex has argonaut proteins to inhib gene exp argo1 = silencer, binds NA argo2 = endonucl, cleaves target RNA |
|
|
Human RNA silencing - PKR |
long dsRNA causes the ISG PKR to autophos, leading to phos of ELF2a to block protein synth |
|
|
miRNA for silencing |
in humans not perfectly duplex, has sequence mismatches |
|
|
Human silencing - RISC |
triggered by small 21nt RNA DNAP produces pre miRNA shRNA enters nucleus, interact with DICER RISC represses translation |
|
|
cellular miRNA silences exp of viral RNA |
- primate foamy virus - huan miRNA inhib replication by binding to complementary sequence - Hep C requires miRNA expressed in hepatocytes for replication = tropism
|
|
|
Viral miRNA inhibits host mRNA |
- Adeno Pol3 can bind to inhib PKR and dicer - Herpes LATS prod miRNA = targets apoptosis and CTL killing - HIV Tat suppresses prod of miRNA, supports persistance and latency |
|
|
viral miRNA can inhibit viral mRNA |
- Adeno SV40 late genes prod miRNA that degrades early gene = reduces susceptibility to LT specific CTL |
|
|
miRNA for therapeutic approaches |
make lentivirus vector to produce miRNA and target specfic genes to alter expression
embed miRNA sequence into an intron on a plasmid |
|
|
Cellular restriction factors |
intrinsic antiviral proteins that block RT, uncoating or budding Tetherin prevents enveloped virus from budding APOBEC3 induces lethal G-A hypermutations on ssDNA |
|
|
cancerous agents
|
chemical carcinogens UV and ionising radiation viruses |
|
|
Tumour |
growth produced by abnormal cell proliferation. Leukemia = circulating cells Lymphoma = solid cells |
|
|
Transformation |
introduction of inheritable change to a cell that causes a change in growth phenotype and immortalisation |
|
|
cell cycle |
G1 = synth proteins required for DNA prod S = replication M = mitosis controlled by cyclans that stim/inhib |
|
|
Tumour stimulation genes |
c-onc aka protooncogenes = genes encoding proteins that give GO sigals for exp of proteins. normal products of a cell v-onc - viral version of the c-onc, may be slightly diff |
|
|
Tumour suppressor genes |
give stop signals for negative regulation of growth |
|
|
classes of oncogene |
growth factors for prolif eg. IL2 growth factor receptors intracellular signal transducers TF e.g. NFkb |
|
|
RNA tumour viruses |
non lytic, do not lead to cell death integration is essential v-onc is not unique, there is a c-onc counterpart stimulate activators |
|
|
Classes of retrovirus oncogenesis |
-endogenous - transmitted in germline
|
|
|
retrovirus classes of tumour prod |
transducing - introduce v-onc cis activating - insertional mutagenesis trans activating - gene regulation |
|
|
Transducing retroviruses |
most are rep defective except Rous sarcoma introduce v-onc gene under LTR transcriptional control into host genome |
|
|
Cis activating retroviruses |
have no v-onc, but may insert at particular location upstream of oncogene - its LTR may allow for transcription of downstream oncogene - provirus may contain enhancer that amplifies production of nearby genes |
|
|
Trans activating retroviruses |
In humans - aboriginal populations HTLV-1 associated with adult T cell leukemia-lymphoma (ATLL), high persistence. spread across placenta, breastfeeding, sex |
|
|
HTLV-1 |
infects CD4 Tc exp gag, pol, env, tax and rex tax = TF upreg exp of other genes as well as c-onc IL2/IL2R = uncontrolled Tc prolif. prolif maintained by HBZ when tax downreg |
|
|
DNA tumour viruses |
lytic integration non essential v-onc are unique - inactivate inhibitors requires S phase enzymes to replicate DNA, therefore exp early prot to stimulate entry into S phase |
|
|
DNA tumours |
limites viral protein expression show integration of viral genome/episome |
|
|
Rb protein |
tumour suppressor gene controls transition of G1 to S gets hypophos, bind E2F TF to stop cell cycle |
|
|
P53 |
tumour suppressor gene TF, promotes expression of genes that stop cell growth and initiates apoptosis Hep C binds p53 |
|
|
Papilloma virus tumour virus |
DNA tumour virus -cutaneous skin (warts) - episome -cervix mucosa (cervical carcinoma) - random integration, loss of E2 gene that controls E6 and E7 E6 and E7 suppress p53 and Rb |
|
|
Hep B tumour virus |
genome randomly integrates into cell chromo
protein X encodes transactivating gene that deregulate c-onc. destroys regeneration of cells leading to chromo mt |
|
|
Herpes tumour virus |
solid tumours. EBV (herpes) transforms B cells causing clonal expansion of a single B cell Cofactors - malaria expands B cells |
|
|
Flavivirus |
ssRNA arthopod vector, no HH nonsp entry w promiscuous receptor |
|
|
Dengue - mosquitos |
transmission req moq w inf salivary glands period of asymp viremia - still spread to mosq females feed a lot during embryogenesis extrinsic inc period: 7-14 days to be inf |
|
|
Dengue 2nd infection
|
increased chance of complications = ab dependent enhancement non neautralising ab from 1st inf bind, Dengue uses Fc to infect macro/mono |
|
|
Dengue vaccine |
4 types, ag Xreactive, vacc based on YF vacc, uses Prm and env for each of the 4 types pretty meh efficacy, ~55% in Asia |
|
|
Yellow Fever |
20% fatality, due to spillover evens from tropical sylvatic cycle Aedes aegypti - SE Asia, US, upper AUS |
|
|
Yellow Fever vaccine |
well characterised 1 dose gives 10y immunity low production, low dist |
|
|
Issues for detecting emergence |
silent incubation asymptomatic/mild infections nonsp syndromes e.g. flu low frequency devastating complications |
|
|
Ebola outbreaks and host |
70s - outbreak outside of known geographic range, poor detection 2014 - Guinea, 80% mortality, spread to HCW maintained by bats, spillo ver to apes and deer things |
|
|
Ebola infection and shed |
enters via muc mem or broken barriers high blood titre low ID shed in blood, saliva, tears, milk, semen, faeces, urine |
|
|
Ebola diagnosis |
acute viremia culture, RTPCR, sequence serology - cannot do, people die before detectable IR, access to samples is limited |
|
|
Zika outbreaks |
Africa via sylvatic cycle, Aedes africans Asia, spread from Africa, Aedes aegypti |
|
|
Zika and Wolbachia |
coinfection with Wolbachia in mosq = compete for resources and replication, prevents growth of Zika |
|
|
Zika transmission and shed |
transmission mosquito, perinatal, sexual, transfusion, direct contact shed blood, saliva, urine, semen |
|
|
Zika diagnosis |
short viremia PCR, serology limited to expert labs |
|
|
surveillance and outcomes |
outcomes det by surveillance, diagnosis, contact tracing and isolation incidences in smaller pop harder to detect than larger pop, even if its at the same freq |
|
|
What is an emerging Infectious disease? |
disease whose incidence has increased in the past 20 years and threatens to increase in the near future |
|
|
How do you get emerging viruses? |
emerging viruses are the causative agent of new or previously unrecognised infection expanded host range, zoonoses, cross species infection, globalisation, climate change, viral mt |
|
|
Emerging virus contributing factors |
-access to susceptible/permissive cells -population density and health -human demographic change (expansion to new areas means new sources of inf) -economic development -behaviour change (child car facilities, sex, drugs) -zoonotic transsmission |
|
|
Climate change and emerging viruses |
- less predators, more food supply - temp can affect rodent breeding patterns - prolong the time vector can transmit disease - drought = resurgence of mosq disease as they move towards urban areas of water storage |
|
|
Evolution and emerging viruses |
biodiversity of pathogens produces quasispecies
|
|
|
zoonosis |
enzootic = endemic - virus continually circulated between animal hosts
epizootic - epidemic - occurs in animal host sparodically to cause epidemic disease in humans |

