![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
80 Cards in this Set
- Front
- Back
|
what are the three white cell neoplastic disorders we will study in this course?
|
1. leukemias
2. lymphomas 3. myeloproliferative disorders |
|
|
cancers of the blood or bone marrow are called?
|
leukemias
|
|
|
a cancer causing overproliferation of progenitor cells in the bone marrow is termed?
|
leukemia
|
|
|
what are the two cell lineages which can be effected by a leukemia?
|
1. myeloid
2. lymphoid |
|
|
does this describe acute or chronic leukemia?
Block in differentiation results in accumulation of stem/progenitor cells which eventually fill the bone marrow cavity and spill out into the blood stream as blasts? |
acute
|
|
|
when suppression of normal hematopoietic stem cells occurs, what is the term for the resulting in a decrease of all types of normal blood cells?
|
Pancytopenia
|
|
|
why are acute leukemias aslo called clonal disorders?
|
all the abnormal cells came from one original cell thus all have the same characteristic mutations
|
|
|
what are the 2 types of acute leukemias?
|
1. Acute lymphoblastic Leukemia (ALL)
2. Acute myeloid leukemia (AML) |
|
|
Is chronic or acute leukemia described below?
No clear blockage of progenitor cell maturation, but there are a heterogeneous collection of mature and immature WBC's in the blood. |
Chronic leukemia
|
|
|
why are chronic leukemias watched very closely even if it appears they are progressing very slowly?
|
they can change into acute leukemia over a very short period of time
|
|
|
what are the two classifications of chronic leukemias?
|
1. Chronic myelogenous leukemia (CML)
2. Chronic lymphocytic leukemia (CLL) |
|
|
Cancers of the lymph tissue are called?
|
Lymphomas
|
|
|
an over production of lymphoid cells in the lymph nodes, spleen, or other lymphoid organ is known as?
|
lymphoma
|
|
|
what are the 2 types of lymphomas?
|
1. Hodgkin's disease
2. Non-Hodgkin Lymphomas (NHL) |
|
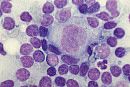
What is this cell and what white cell neoplastis disorder might cause its formation?
|
Reed-Sternberg Giant Cell caused by Hodgkins lymphoma
|
|
|
what are the 3 techniques for typing a lymphoma?
|
1. Morphology
2. Molecular Genetics 3. Immunophenotype determination |
|
|
what typing test for lymphomas relies on determining to B or T cell clonality and chromosomal translocations?
|
Molecular genetics
|
|
|
detection of surface membrane antigens (the CD type) is characterized by what technique used to determine the typing of a lymphoma?
|
Immunophenotype determination
|
|
|
Cancer of myeloid cells is called?
|
a myeloproliferative disorder
|
|
|
an excesss production of granulocytes is called?
|
Chronic myelogenous leukemia (is a myeloproliferative disorder)
|
|
|
an excess production of erythrocytes is called?
|
Polycythemia vera (is a myeloproliferative disorder)
|
|
|
an excess production of platelets is called?
|
Essential thrombocytopenia or ET (is a myeloproliferative disorder)
|
|
|
an excess production of fibroblasts of the bone marrow is called?
|
Agnogenic myeloid metaplasia (AMM) or idiopathic myelofibrosis (is a myeloproliferative disorder)
|
|
|
what is a good way to distinguish between a reactive process and a myeloproliferative disorder?
|
myeloproliferative disorders are usually monoclonal where as a reactive process would not be
|
|
|
cytogenic abnormalities, marrow hyperplasia or hypermetabolism, and abnormal maturation of WBC's indicate what type of white cell neoplastic disorder?
|
Myeloproliferative disorder
|
|
|
what percentage of caners in the U.S. are a type of leukemia?
|
5%
|
|
|
what percentage of malignant diseases in children are leukemias?
|
50%
|
|
|
what are the 3 common molecular characteristics of leukemias?
|
1. subtractions, additions, translocations on various chromosomes
2. Absent, increased, or decreased cellular proteins 3. Ability to proliferate inside the marrow even when producing faulty copies |
|
|
basic diagnosis of leukemia can only be made when what number of cancer cells is present?
|
10^9 for chronic phase
10^12 for acute phase |
|
|
what is the most common cause of acute and chronic leukemias is which of the following?
1. translocations 2. subtractions 3. additions |
translocations
|
|
|
interchromosomal gene rearrangements are called?
|
translocations
|
|
|
what is the translocation found in CML?
|
t(9;22)(Ph1)
|
|
|
what is the translocation found in AML?
|
t(8;21)
|
|
|
what is the translocation found in APL?
|
t(15;17)
|
|
|
what is the translocation found in Burkitt's Lymphoma?
|
t(8;14)
|
|
|
what is the abnormal protein resulting in CML?
|
bcr/able
|
|
|
what is the abnormal protein resulting in AML?
|
CBF/AML1
|
|
|
what is the abnormal protein resulting in APL?
|
RAR-alpha
|
|
|
what is the abnormal protein resulting in Burkitt's Lymphoma?
|
Ig/myc
|
|
|
what percentage of patients with CML have the t(9;22)(Ph1) translocation?
|
95%
|
|
|
describe the effect of the bcr/able protein mutation causes?
|
bcr/abl become fused resulting in a constitutively active abl tyrosin kinase
|
|
|
how is CML diagnosed?
|
karyotyping that reveals the presence of the philadelphia chromosome (chromosome 22) which is shorter than it should be
|
|
|
describe the pathogenesis of the CBF-AML1 protein mutations that lead to AML?
|
CBF-AML1 normally enhances transcription of hematopoietic genes that are essential for differentiation. In the mutation, ABL1 can bind an additional co-repressor complex which completely stops transcription of the genes responsible for progenitor cell differentiation causing a build up of "blasts"
|
|
|
what happens in the inv(16) mutation with CBF/ABL1?
|
CBF is mutated and when it binds to ABL1 they are ejected from the nucleus and cannot enhance transcription of differentiation genes
|
|
|
what is RAR?
|
retinoic acid receptor
|
|
|
what does vitamin A deficiency cause?
|
abnormal hematopoiesis
|
|
|
what is the cure for APL cancer?
|
all-trans retinoic acid (ATRA) because it can induce terminal differentiation in cell lines where RAR has been mutated to form a chimeric RAR
|
|
|
what are c-myc genes invloved in?
|
regulation of cell cycle
|
|
|
what are IgG genes involved in?
|
code for immunoglobulins
|
|
|
describe the pathogenesis of Burkitt's Lymphoma?
|
The promotor IgG gene gets placed next to the c-myc gene resulting in a oncogene which causes increased B-cell proliferation
|
|
|
what are the 3 types of changes that are seen in the HL-60 cancer cell model?
|
1. A growth factor receptor becomes constitutively activated
2. A tumor suppressor gene is deleted 3. A oncogene is overactive |
|
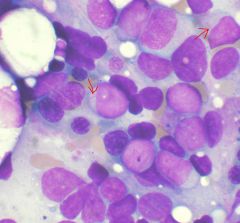
What are the structures indicated by the arrows?
|
Auer Rods seen in AML
|
|
|
what are 2 examples of enzyme assays that can be used to diagnose leukemia?
|
1. Myeloperoxidase activity (is it present?)
2. Alkaline phosphatase activity (low in leukemia) |
|
|
what are the two tools which can be used for genetic analysis in leukemia?
|
1. FISH
2. PCR |
|
|
Describe how FISH analysis works?
|
Probes for specific sequences of DNA such as BCR and ABL1 can be created to fluoresce specific colors when they are bound to target sites. If the target genes are together such as BCR/ABL1 then there colors will merge to make a new color
|
|
|
what stage of mitosis should cells be in for use of FISH?
|
metaphase
|
|
|
what stage of mitosis can lead to false positives?
|
interphase because around 4% of cells show an overlap of BCR/ABL1 in interphase
|
|
|
how is PCR used to find cancerous cells?
|
primers are created which will bind to specific points of DNA near protoncogenes. When these genes are brought together the polymerase reaction is able to occure and the cancerous DNA will be amplified so that it can be identified. If no cancerous cells are present then PCR reaction is not succesful.
|
|
|
Describe the technique of flow cytometry?
|
Monoclonal antibodies to specific CD (cluster differentiation = glycoprotein fingerprint of luekocyte) that are labeled with a flourescent tag. Different leukocytes or mutated leukocytes can be identified by this method
|
|
|
what key factor allows flow cytometery to identify cells in varrying stages of development?
|
as cells develope their CD fingerprint changes thus not only do cells of differing lineages have unique CD fingerprints, but also cells within the same lineage with differing degrees of maturity
|
|
|
what is the important CD domain which must be evaluated before a marrow transplant?
|
the MHC domain
|
|
|
what is the role of the MHC domain on cells?
|
to identify the cell as belonging to the organism
|
|
|
what are the two types of MHC?
|
Class I and Class II
|
|
|
how many MHC class I does each human have?
|
6
|
|
|
how many MHC class II does each human have?
|
8
|
|
|
which cells in the body have MHC class I?
|
all
|
|
|
which cells in the body have MHC class II
|
APC's
|
|
|
what T cells recognize MHC class I and destroy non-self cells?
|
Cytotoxic killer T cells
|
|
|
which T cells recognize MHC class II?
|
helper-T-cells
|
|
|
what is the target for antibodies conjugated with drugs or radioactive isotopes in AML?
|
CD33
|
|
|
what is the target of the engineered chimeric antibody against NHL?
|
CD21
|
|
|
what superfamily to GM-CSF, IL-3 and IL-5 belong to?
|
cytokine superfamily
|
|
|
describe the GM-CSF receptor?
|
an alpha and beta subunit which when complexed allows GM-CSF to bind to its receptor and transduce a signal
|
|
|
the signal tranduced by binding og GM-CSF to its receptor can start which 3 molecular intracellular pathways?
|
1. Ras/MAPk
2. JAK-STAT 3. PI3k/P70S6k |
|
|
Ras/MAPk pathway results in what?
|
initiation of gene transcription by the activation of c-fos, c-jun, and c-myc
|
|
|
the JAK/STAT pathway results in what?
|
proliferation and differentiation
|
|
|
the PI3k/P70S6k pathway results in what?
|
protein synthesis leading to cell growth
|
|
|
which of the singal tranduction pathways are associated with leukemia and what type?
|
1. Ras and Abl are associated with CML [t(9;22)]
2. Myc malfunction is associated with Burkitt's Lymphoma [t(8;14)] |
|
|
Explain the role of Ras in developing cancer?
|
it normally sends out a proliferative signal when grwoth factors are present. In its mutated form in is on constantly.
|
|
|
what is the new treatment being investigated to treat Ras mediated cancer?
|
drugs blocking the enzyme farnesyl transferase which activates Ras thus stopping ras's ability to cause cell proliferation
|

