![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
13 Cards in this Set
- Front
- Back
|
Oxidising power of a halogen |
The oxidising power of a halogen is a measure of the strength with which a halogen atom is able to attract and capture an electron to form a halide ion |
|
|
Half equations of a halogen |
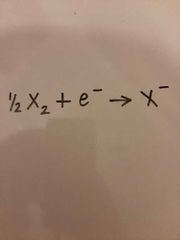
|
|
|
Trends in reactivity of Group 7 |
Reactivity DECREASES DOWN the group: More shells, atomic radius increases Greater shielding effect Weaker nuclear attraction Less able to attract and capture another electron Fluorine is most reactive |
|
|
Displacement reactions |
A displacement reaction is a reaction in which a more reactive element displaces a less-reactive element from an aqueous solution of its halide ions |
|
|
Colours of displacement reactions for halogens |
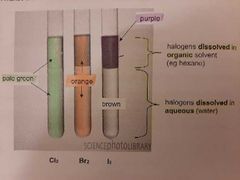
|
|
|
For each displacement reaction you must write : |
Overall redox reaction Ionic equation Observations |
|
|
Displacement reactions |

A more reactive halogen will displace a less reactive halogen. |
|
|
Disproportionation reaction definition |
A disproportionation reaction is a reaction in which the same element is both oxidised and reduced |
|
|
Disproportionation of chlorine in water |

|
|
|
Disproportionation of chlorine in aqueous sodium hydroxide |

|
|
|
Test for a halogen - chlorine |

|
|
|
Halogen test - bromine |
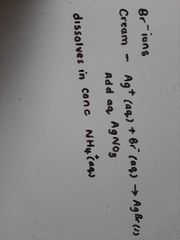
|
|
|
Halogen test - iodine ions |

|

