![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
30 Cards in this Set
- Front
- Back
- 3rd side (hint)
|
Mineral
|
Homogenous, Naturally Occurring, Inorganic, Solid, Orderly arrangement of atoms, Defineable chemical composition.
|
There are 6
|
|
|
Crystal Form
|
The shape formed by the natural growth of a mineral. Crystal faces smooth and flat.
|
|
|
|
Cube
|
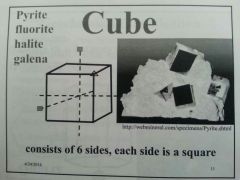
|
P, H, F
|
|
|
Hexagonal Prism
|

|
|
|
|
Rhombohedron
|
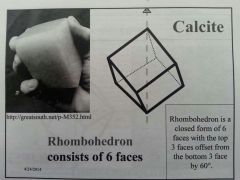
|
|
|
|
dodecahedron
|

|
|
|
|
Octahedron
|

|
|
|
|
Luster
|
How light is reflected off surface of mineral. Metallic , non-metallic
|
|
|
|
Color
|
What you see.
|
|
|
|
Streak
|
color of powered mineral
|
|
|
|
Hardness
|
Resistance to surface being scratched. (Finger nail 2.5) (talc 1) (glass = Iron nail = 5.5) (Diamond10)
|
|
|
|
Cleavage
|
breaks along weakness in structures and produces flat smooth surface.
|
|
|
|
Specific Gravity
|
Density/Density of water ranges from 2-15
|
|
|
|
Acid reaction
|
10% HCI : Bubbles (Calcite) Stinks (Sulfide) Dissolves (Salt)
|
|
|
|
Natural Magnets
|
Magnetite, Fe3O4, arrangement of electrons causes ot to behave like an Iron magnet.
|
|
|
|
Siderophile
|
Iron liquid affinity for Fe into core
|
|
|
|
Chalcophile
|
sulfide liquid affinity for S
|
|
|
|
Lithophile
|
silicate liquid affinity for SI and O into mantle and crust.
|
|
|
|
atmophile
|
gas phase volatiles into atmosphere, N2
|
|
|
|
Biophile
|
affinity for life
|
|
|
|
Hydrophile
|
affinity for water dissolves in water
|
|
|
|
cosmosphile
|
affinity for space gas that remains in space or escapes from earth
|
|
|
|
Native elements
|
elements in native state
|
|
|
|
Halides
|
cation bonded with CI or F
|
|
|
|
Oxides
|
cation bonded with O2
|
|
|
|
sulfides
|
cation bonded with SO42
|
|
|
|
carbonates
|
cation bonded with CO32
|
|
|
|
silicates
|
Cations bonded with SiO44- groups
|
|
|
|
cations are
|
positive Na+, SI4+, Fe2+ etc....
|
|
|
|
Native minerals
|
Diamond, Graphite, gold, silver, Iron, Platinum, Sulfur.
|
|

