![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
9 Cards in this Set
- Front
- Back
|
What is the scientific name AND chemical formula for limestone? |
Calcium Carbonate CaCO3 |
|
|
What is thermal decomposition? |
Breaking a compound down into other substances by heating |
|
|
What is the word and symbol equation for the thermal composition of limestone? |
Calcium Carbonate => Calcium Oxide + Carbon Dioxide CaCO3 => CaO + CO2 |
|
|
What is produced when a carbonate reacts with an acid? |
salt + water + carbon dioxide |
|
|
What is cement? |
Powdered limestone (calcium carbonate) and clay, heated together |
|
|
What is concrete made of? |
Cement, gravel, sand and water |
|
|
What are the advantages of limestone quarrying? |
- Limestone is valuable as it is used to make glass and concrete - It provides employment which helps the economy |
|
|
What are the disadvantages of limestone quarrying? |
- They're visible from long distances - Heavy industry results in noise and traffic, which disturbs the people living nearby - Disturbs the environment and local habitats |
|
|
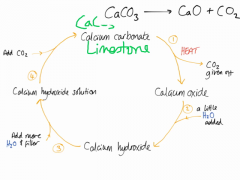
Draw the limestone reaction cycle |
|

