![]()
![]()
![]()
Use LEFT and RIGHT arrow keys to navigate between flashcards;
Use UP and DOWN arrow keys to flip the card;
H to show hint;
A reads text to speech;
24 Cards in this Set
- Front
- Back
|
Name five factors that influence drug release
|
1. Solute 2. Formulation 3. Particle size 4. Solubility/saturation 5. Methodology |
|
|
What is the Noyes-Whitney equation? |
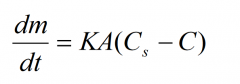
|
|
|
Name five factors that influence drug release in dissolution experiments in the lab |
1. Solvent 2. Agitation 3. Volume 4. Diameters of vessels 5. Temperature |
|
|
Name three BP dissolution apparatuses |
1. Basket apparatus
2. Paddle apparatus 3. Flow through apparatus |
|
|
What materials is the basket dissolution apparatus useful for? |
Large microparticles. Floating dosage forms. |
|
|
What materials is the paddle dissolution apparatus useful for? |
Tablets/capsules. 'Sinking' dosage forms. |
|
|
What materials is the flow-through dissolution apparatus useful for? |
Tablets/capsules/particles. Sinking OR floating dosage forms. |
|
|
Name one non-compendial dissolution apparatus |
1. Franz cell system |
|
|
What materials is the Franz cell system dissolution apparatus useful for? |
Semi-solids/suspensions. |
|
|
What are the conditions for simulated stomach dissolution fluid? |
- Simulated gastric fluid: 2.0g NaCl, 3.2g pepsin powder, 80mL 0.1M HCl and dilute to 1L with water.
|
|
|
What are the conditions for simulated intestine dissolution fluid? |
Simulated intestinal fluid: 6.8g monobasic potassium phosphate in 250mL water, 10g pancreatin, adjust to pH 6.8 and dilute with water to 1L. |
|
|
What are the seven stages of tablet dissolution? |
1. Mechanical lag 2. Wetting 3. Penetration of fluid 4. Disintegration 5. Disaggregation 6. Dissolution 7. Occlusion |
|
|
What is the Hixson & Crowell equation? |
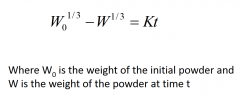
|
|
|
What is a Wagner plot? |
Log(undissolved drug %) vs Time |
|
|
What is the Higuchi equation? |
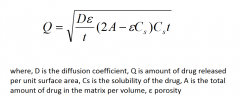
|
|
|
What is plotted on a Higuchi plot? |
Q vs sqrt(t) |
|
|
What is IVIVC? |
In vitro, in vivo correlation |
|
|
Purpose of IVIVC? |
- Dissolution originally for QC and reproducibility - Bioequivalence only needs to be in-vitro if referenced to a bioequivalence standard - Bioequivalence standard is an in-vitro test that has proven IVIVC - Dissolution may have good IVIVC - Once IVIVC is established, replaced PK requirements - Cost, ethically better |
|
|
What is the intercept method in regard to IVIVC? |

|
|
|
Give an example of poor IVIVC |
Ibuprofen dissolution |
|
|
Give an example of good IVIVC |
Aspirin excretion |
|
|
State three things that may cause a poor IVIVC
|
1. Poor study design 2. Drug 3. Gastric emptying dependant |
|
|
Give an example drug for each of the BCS classes |
Class 1 - Propanol
Class 2 - Carbamazepine Class 3 - Atenolol Class 4 - Frusemide |
|
|
What are the four BCS classes? |
Class 1 - high permeability, high solubility Class 2 - high permeability, low solubility Class 3 - low permeability, high solubility Class 4 - low permeability, low solubility |

